General Topics
KEY POINTS
- The prevalence of obesity and obstructive sleep apnoea (OSA) are increasing. OSA is still underdiagnosed.
- Moderate to severe OSA is suspected when patients score 5-8 on the STOP-BANG screening tool.
- Demand is higher than ever for ambulatory surgery and enhanced recovery after major surgery, even in complex, co-morbid patient groups. Critical care resources are scarce and should be reserved for those most in need.
- Patients with established or suspected OSA need a practical risk assessment to guide perioperative management.
INTRODUCTION
10-20% of surgical patients are identified as being at high risk of obstructive sleep apnoea (OSA) at preoperative assessment and around 80% of these patients are not previously diagnosed1. The prevalence of obesity (a closely related risk factor) and OSA are increasing.
Patients with OSA have more than twice the incidence of post-operative oxygen desaturations, respiratory failure, cardiac events and unplanned intensive care admissions2.
Capacity for providing level two care (see Figure 1) or extended monitoring is limited. There is also increasing pressure to manage complex surgical patients as day cases or on enhanced recovery programmes despite co-morbidities. There are restrictions on who can practically be referred for polysomnography due to limited resources or the timely nature of surgery required. Risk stratification of the ‘at-risk’ patient ensures that patients are managed safely, in an appropriate environment, and that scarce resources are reserved for the most in need.

Figure 1: UK Department of Health Levels of Care 3
This article is a comprehensive review of up to date evidence regarding management of both the elective patient with known or suspected sleep apnoea and the emergency patient. It will build on previous tutorials entitled, “Obstructive Sleep Apnoea and Anaesthesia”4 and “Anaesthesia for Children with Obstructive Sleep Apnoea5, and considers guidelines produced by the Association of Anaesthetists of Great Britain and Ireland (AAGBI)6 and the UK Society for Obesity and Bariatric Anaesthesia (SOBA)7. Together with a practical approach to risk stratification and management algorithms,8 this tutorial hopes to provide a holistic approach to perioperative management of these patients.
OBSTRUCTIVE SLEEP APNOEA
Definition
Obstructive sleep apnoea (OSA) is defined as repeated episodes of complete airway obstruction leading to apnoea (defined as 10s of absence of flow) and/or partial obstruction leading to hypopnoea (defined as flow halved over 10s period), both causing a fall in oxygen saturation. Oxygen desaturation is defined as ≥4% drop from baseline or last recorded value for >10s, or Sp02 levels of <90%.
Central recognition of reduced partial pressure of oxygen leads to repeated partial arousal from sleep. This causes increased sympathomimetic activity, leading to tachycardia and hypertension (paradoxically higher than day time blood pressure). This in turn leads to day-time somnolence and a sustained increase in adreno-cortical tone9.
Epidemiology
The prevalence of diagnosed mild OSA is 25% in males and 10% in females. Moderate sleep apnoea occurs in 11% of males and 5% of females1. Prevalence increases to 30-50% in the bariatric surgical population10.
Obesity is a major risk factor for the development of sleep-disordered breathing, and currently affects approximately 25% of all men and women. Trends suggest that this could reach 50% by 205011.
Risk Factors
OSA can be caused by physical characteristics of the face, neck and airway reducing airway patency and/ or central causes leading to respiratory muscle weakness or reduced respiratory drive12 (Figure 2).
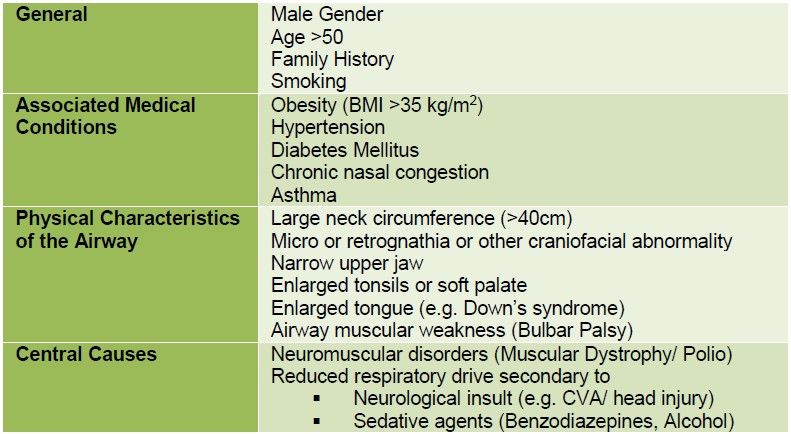
Figure 2: Table showing the risk factors and associated conditions of Obstructive Sleep Apnoea3, 13
Diagnosis
The gold standard method for diagnosis of OSA is polysomnography or sleep study, resulting in the apnoea-hypopnoea index (AHI). Patients are referred based on clinical suspicion of OSA triggering the use of screening tests. A common screening tool used in perioperative care is the STOP-BANG questionnaire (Figure 3). A score of 5-8 identifies patients with a high probability of moderate to severe OSA. Anyone with risk factors (see Figure 2) should be screened for OSA using the tool below.

Figure 3. The STOP-BANG questionnaire14. 1 point is received for each positive answer. 0-2 (Low Risk), 3-4 (Intermediate Risk), 5-8 (High Risk)
Full polysomnography is labour intensive, expensive and often has a long waiting list. Home respiratory studies are more accessible and less costly and include overnight pulse oximetry (giving oxygen desaturation index, ODI) or pulse oximetry plus airflow and abdominal effort monitoring (to give ODI and apnoea-hypopnoea indices, AHI). The AHI is the number of apnoeas or hypopnoeas recorded per hour of sleep during a sleep study.
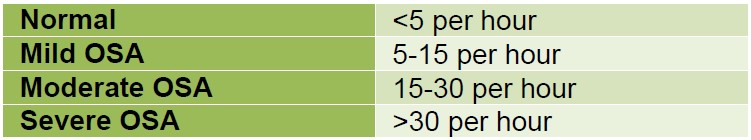
Figure 4. Apnoea Hypopnoea Indices (AHI)
Using ODI, a 4% reduction in SpO2 is considered a significant event. Reductions in SpO2 that stay at or above 90% are considered mild, 80-89% are moderate and <80% are severe. 30 events per hour is diagnostic of sleep-disordered breathing, suggesting increased risk of post-operative complications. ODI is a good predictor of AHI.
Implications for Anaesthesia
Anaesthetic agents such as sedatives, hypnotics, opioids and muscle relaxants all worsen airway instability, central sensitivity to hypoxia and hypercapnia, and respiratory muscle function, leading to more frequent and severe apnoeas. Combined with the metabolic and cardiovascular response to surgical stress, there is significant risk to the patient undergoing major surgery.
Patients with OSA have over double the incidence of post-operative oxygen desaturation, respiratory failure, cardiac events and unplanned ICU stay2.
PERIOPERATIVE MANAGEMENT
PREOPERATIVE ASSESSMENT
For elective surgery, a thorough approach well in advance of surgery is ideal so that investigations can be performed to assess risk and plan appropriate care. For the emergency patient, more rapid assessment tools can be utilised to risk stratify the patient and formulate a safe management plan.
History
Establish associated co-morbidities common to both obese patients and those with obstructive sleep apnoea. Cardiovascular problems include hypertension, ischaemic heart disease, hypercholesterolaemia, heart failure, arrhythmias, cor-pulmonale and cerebrovascular disease. The metabolic syndrome causing insulin resistance or diabetes, gastro-oesophageal reflux and osteoarthritis are also common.
A history for symptoms such as shortness of breath, snoring, chest pain, syncope and fatigue should be taken. Exercise tolerance should be assessed and measured using the New York Heart Association score and explored further by quantitative tests where there are concerns.
Examination
Examination may reveal increased BMI (30-34.9 kg/m2 Obese 1, 35-39.9 kg/m2 Obese 2, >40 kg/m2 Obese 3, previously ‘morbid obesity’15), shortness of breath on exertion or at rest, resting hypertension and tachycardia or signs of right heart failure (increased jugular venous pressure, parasternal heave, tricuspid regurgitation murmur, hepatomegaly and peripheral oedema). A full airway assessment including neck circumference (>40cm is significant), Mallampati score, neck (atlanto-occipital) mobility, assessment of dentition, tongue size and mouth opening should be performed.
Investigation
Blood tests should include full blood count (for polycythaemia), urea and electrolytes (for renal impairment secondary to hypertension), liver function (for hepatic congestion, non-alcoholic steatohepatitis) and blood glucose (+/- HbA1c). An ECG may reveal arrhythmias such as atrial fibrillation, right ventricular hypertrophy, right axis deviation, tall R waves in V1 and right ventricular strain.
An arterial blood gas (ABG) is useful to assess arterial SpO2 and to assess for carbon dioxide retention, although not routinely performed in most pre-operative assessment clinics. A baseline oximetry reading of ≤ 94% without any other underlying condition is suggestive of severe OSA16. Full pulmonary function tests may be necessary where indicated by respiratory disease, poor exercise tolerance or ABG.
Echocardiography may be indicated to assess right heart function and identify pulmonary hypertension in patients with established OSA where the surgery is expected to be high risk or of lengthy duration16. Pulmonary hypertension is present when mean pulmonary artery pressure exceeds 25 mmHg at rest or 30 mmHg with exercise. In reality echocardiography can be difficult to perform in patients with increased BMI. Many patients are also limited physically and exercise tolerance is impossible to quantify.
Premedication
Premedicate with simple analgesics such as paracetamol and ibuprofen if not contraindicated. Avoid sedative premedication where possible. Consider preoperative proton pump inhibitor and/or antacids.
Optimisation of co-morbid conditions
Weight reduction can be encouraged at the preoperative assessment and surgery may be delayed to achieve this (if appropriate). Optimisation of diabetes and hypertension should be undertaken as per local pre-assessment guidelines.
Further preoperative planning depends on whether there is a suspicion of OSA or an established diagnosis.
Preoperative assessment in known OSA patients
Known OSA patients should be counselled regarding increased perioperative risk and the possible need for prolonged post-operative monitoring. Polysomnography results should be reviewed and the severity of the disease noted (see Figure 4).
In ‘Mild’ OSA (AHI 5-15 and pulse oximetry on air of >94%) patients can proceed with routine perioperative care. Day surgery for minor operations if co-morbidities are well controlled is not contraindicated and should be actively supported.
Where the AHI is >15 or pulse oximetry is ≤ 94% the patient should be referred or may already be established on continuous positive airway pressure (CPAP) non-invasive therapy or bilevel positive airway pressure (BIPAP) therapy with respiratory specialist support. Some patients may not be compliant with therapy and may benefit from encouragement and support in the preoperative period.
When the patient presents for urgent surgery, plans need to be made for prolonged postoperative monitoring in a Level 2 area (high dependency or recovery).
Patients on established treatment (CPAP) should continue therapy and bring their device with them to hospital. CPAP improves sleep quality and daytime function and has been proven to improve post-operative apnoea hypopnoea indices, reduce episodes of desaturation16 and postoperative cardiovascular complications,17 and lead to reduced postoperative stay1. The evidence for the exact duration of treatment required to reduce risk is unclear, however two to three months of therapy where possible is recommended.
Preoperative assessment in suspected OSA Patients
In the suspected OSA patient, preoperative management depends on the urgency of surgery. All patients who have risk factors (Figure 2) can be screened using the STOP-BANG tool (Figure 3) in both elective and emergency settings if they are conscious and able to answer questions. If not, the anaesthetist should make an appropriate plan, based on information available.
Where possible those with STOP-BANG scores ≥ 5 (moderate to severe OSA):
- WITH co-morbidities (uncontrolled hypertension, arrhythmias, cerebrovascular disease, morbid obesity, heart failure and metabolic syndrome) AND/OR undergoing major elective surgery should be referred for full polysomnography and a review by a sleep physician.
- WITHOUT co-morbidities and undergoing minor surgery can be referred for overnight pulse oximetry alone or treated as presumed moderate OSA and appropriate risk reduction steps taken.
Where sleep studies are impossible due to the urgency of surgery, a practical and safe perioperative approach should be undertaken with consideration of the risks from the surgery and of the patient’s own problems.
INTRAOPERATIVE MANAGEMENT
Staff
High risk patients should be managed by a senior anaesthetist and surgeon (morbidly obese, moderate to severe OSA, or via a validated risk scoring system such as P-POSSUM). Ensure adequate staff and appropriate theatre equipment to manage an obese patient if indicated.
Monitoring
Pulse oximetry, ECG, non-invasive blood pressure and respiratory monitoring should be supplemented by invasive arterial monitoring in severe sleep apnoea. This allows assessment of acid-base status and provides information about respiratory function when planning post-operative care.
Airway Management
Be prepared for a difficult airway with appropriate help and equipment available and a clear plan communicated to all team members. Airway difficulty occurs eight times more frequently in the presence of OSA7. Preoxygenate adequately, in a ramped position (ear level with sternum)18 with positive end-expiratory pressure (PEEP) if necessary. Beware of gastrooesophageal reflux disease due to hypotonia of the lower oesophageal sphincter, and consider a rapid sequence induction. Intubate and ventilate rather than relying on spontaneous ventilation via a laryngeal mask to control carbon dioxide levels throughout surgery.
Anaesthetic and Analgesic Technique
Regional or local anaesthesia will reduce the risk of anaesthetic agents, opioids and muscle relaxants and should be used preferentially where possible, especially in ambulatory surgery. If general anaesthesia is mandatory, use short acting anaesthetic agents (desflurane, remifentanyl, propofol) and minimise opioid use by using local or regional techniques and a multi-modal approach to analgesia (paracetamol, NSAIDS (if not contraindicated), ketamine, clonidine (beware of sedative effects)). Postoperative reduction in oximetry is 12-14 times more likely when post-operative opioids are used compared to non-opioids7.
In cases with a BMI >35 kg/m2 adjust doses according to the SOBA Guideline (see Figures 5 and 6).
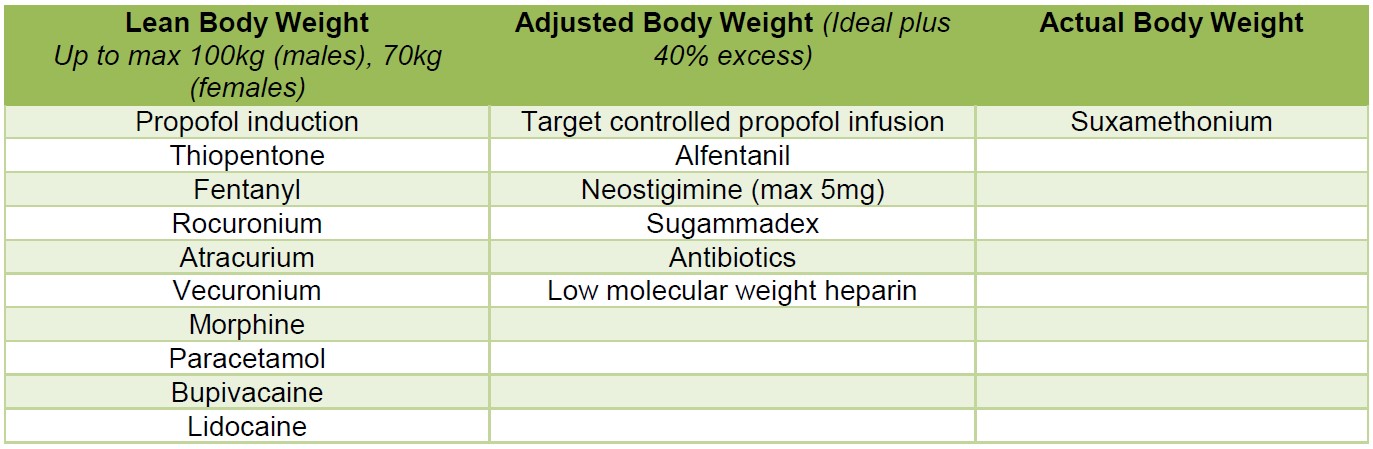
Figure 5: The SOBA Single Sheet Guideline: Suggested dosing regimes for anaesthetic drugs18

Figure 6: Lean Body Weight and Ideal Body Weight Calculations5, 19
Extubation
Check the arterial blood gas and ensure full reversal of neuromuscular blockade before extubation. Consider extubating directly on to the patients’ own CPAP machine. Extubate all patients fully conscious and non-supine (semi-upright or lateral).
POSTOPERATIVE MANAGEMENT
Patients with mild OSA, especially those having ambulatory or minor surgery can be managed routinely.
All patients with suspected or proven moderate to severe OSA should be nursed in a Level 2 or higher area following surgery, in an upright position, with appropriate monitoring and continuous supplemental oxygen therapy until they can maintain their oxygen saturation on room air.
When using patient controlled analgesia devices avoid using background infusions where possible.
Those who undergo high risk surgery (see Figure 7) or have significant co-morbidities or excessive opioid requirements in recovery should automatically receive overnight continuous oximetry (in Level 2 area or higher). Consideration should be made for continuous invasive arterial pressure monitoring to allow assessment of pCO2.
Those with suspected or confirmed mild to moderate OSA but without co-morbidities, having low risk surgery with no significant opioid requirement can be observed for 30-60 minutes unstimulated in recovery to assess respiratory risk.
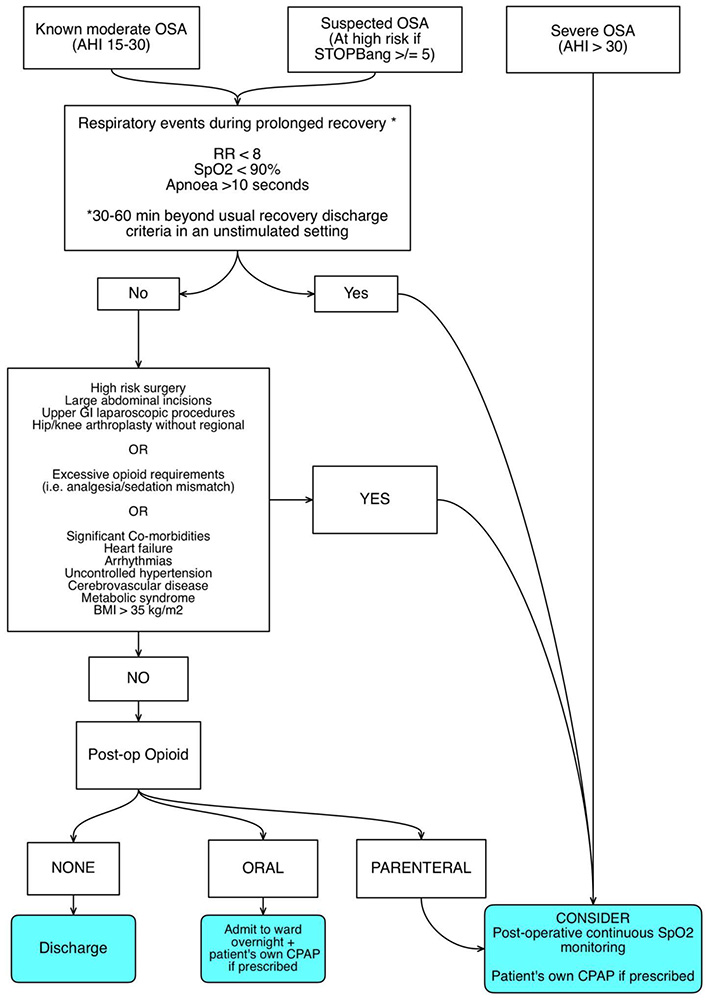
Figure 7: Algorithm for post-operative management of patients with suspected or proven sleep apnoea.
Consideration should be made to commence CPAP or Non-Invasive Positive Pressure Ventilation (NIPPV) and admit the patient for overnight pulse oximetry if recurrent adverse respiratory events occur (see Figure 7), they are non-compliant with CPAP therapy, have known severe OSA or pain sedation mismatch.
If patients have no opiate requirement and have had ambulatory surgery with no adverse respiratory events in recovery they can be considered for discharge. Those that have had non-ambulatory surgery, no opiate requirement and have an easy airway can be managed in a Level 1 or ward based setting.
Patients who are already established on CPAP or NIPPV (non-invasive positive pressure ventilation) should have this therapy available in recovery unless it is contraindicated by their operation.
SUMMARY
- Obesity and obstructive sleep apnoea are becoming increasingly common. Perioperative care of these patients places a demand on critical care resources.
- In the elective setting, ‘at risk’ patients should be identified and referred for investigation and treatment with adequate time to establish CPAP therapy, and undergo weight loss if indicated. Co-morbidities should be identified and optimised, and plans should be made for postoperative care.
- Appropriate intraoperative care that reduces risk to the patient will result in reduced postoperative adverse respiratory events and reduces postoperative stay.
- Patients who are undiagnosed may present for emergency surgery. Patients should be risk stratified using the STOP-BANG scoring system and high risk patients either automatically managed in a Level 2 setting or assessed for adverse respiratory events in recovery and then directed appropriately to Level 1 or 2 care.
- Some patients with OSA have an unremarkable postoperative course and with appropriate assessment and care can be discharged to Level 1 care or even home, leaving high dependency beds available for higher risk patients.
REFERENCES AND FURTHER READING
- Raveendran R, Chung F. Ambulatory anesthesia for patients with sleep apnea. Ambul Anesth 2015; 2: 143-51
- Kaw R, Chung F, Pasupuleti V et al. Meta-analysis of the association between obstructive sleep apnoea and post-operative outcome. Br J Anaesth 2012; 109: 897-906
- Department of Health, UK. Comprehensive Critical Care: A Review of Adult Critical Care Services. Available from: http://webarchive.nationalarchives.gov.uk/20130107105354/http:/www.dh.gov.uk/prod_consum_ dh/groups/dh_digitalassets/@dh/@en/documents/digitalasset/dh_4082872.pdf. Accessed 29/04/2017.
- Bright S. Obstructive Sleep Apnoea and Anaesthesia. Anaesthesia Tutorial of the Week 2009; https://www.aagbi.org/sites/default/files/152-Obstructive-Sleep-Apnoea-and-anaesthesia1.pdf (Accessed 15/03/2016)
- Walker I. Anaesthesia for children with obstructive sleep apnoea. Anaesthesia Tutorial of the Week. 2009; http://www.frca.co.uk/Documents/159%20Anaesthesia%20for%20children%20with%20
obstructive%20sleep%20apnoea.pdf (Accessed 15/03/2016) - Association of Anaesthetists of Great Britain and Ireland. Peri-operative management of the obese surgical patient 2015. Br J Anaesth 2015; 70: 859–76
- Margarson M, Shearer E, Cousins J. Recommendations for screening and management of Sleep Disordered Breathing (SDB) in patients undergoing bariatric surgery. SOBAUK [website]. 2016, http://www.sobauk.co.uk/downloads/osa-guideline. (Accessed 11/03/2016)
- Seet E, Chung F. Management of sleep apnea in adults- functional algorithms for the perioperative period: Continuing Professional Development. Can J Anesth 2010; 57: 849-64
- Hall A. Sleep Physiology and the perioperative care of patients with sleep disorders. BJA Education 2015; 15 (4): 167-172
- OSA/OHS Consensus Document. SOBAUK [website]. Feb 2016. Available from: http://www.sobauk.co.uk/downloads/osa-guideline. (Accessed 11/03/2016)
- National Obesity Forum. State of the nation’s waistline–obesity in the UK: analysis and expectations. 2014. http://www.nationalobesityforum.org.uk/media/PDFs/StateOfTheNationsWaistline ObesityintheUKAnalysis andExpectations.pdf. (Accessed 09/02/2016)
- Shneerson JM. Central Sleep Apnoeas. Available from: http://www.britishsnoring.co.uk/central_sleep_apnoeas.php. (Accessed 11/03/2016)
- Mayo Clinic. Obstructive Sleep Apnoea: Symptoms and Causes. Available from: www.mayoclinic.org/diseases-conditions/obstructive-sleep-apnoe/symptoms-causes/dxc-20205871. (Accessed 30/08/2016)
- STOP-Bang.ca. OSA Screening. Available from: www.stopbang.ca/osa/screening.php. (Accessed 30/08/16)
- Public Health England. Measurement of Obesity. 2014. Available from: http://www.noo.org.uk/NOO_about_obesity/measurement (Accessed 09/02/2016).
- Abdullah HR, Chung F. Perioperative Management of Obstructive Sleep Apnoea. Current Anaesthesiology Reports 2014; 4: 19-27
- Mutter TC, Chateau D, Moffatt M, et al. A matched cohort study of post-operative outcomes in obstructive sleep apnea. Anesthesiology 2014; 121: 707-18.
- Anaesthesia for the Obese Patient, BMI >35kg/m2. Single sheet guideline. SOBA. Available from: http://www.sobauk.co.uk/downloads/single-sheet-guideline. (Accessed 11/03/2016)
- Carron M, Guzzinati S, Ori C. Simplified estimation of ideal and lean body weights in morbidly obese patients. Br J Anaesth. 2012; 109(5): 829-30



