Pain
KEY POINTS
- Illicit opiate use is a growing global problem, with 0.4% of the world using heroin or opium.
- Buprenorphine is used in the treatment of opioid use disorder, and has a high affinity but low intrinsic activity for the μ-opioid receptor.
- The naloxone component is used to prevent parenteral diversion, and has no significant clinical effect when taken sublingually or enterally.
- Continuing, reducing, or discontinuing buprenorphine/naloxone perioperatively requires careful consideration of acute postoperative pain expectations, possible multimodal non-opioid analgesic options, and psychosocial factors like relapse risk.
- A multidisciplinary team of surgeons, anesthesiologists, and addiction medicine specialists, is recommended for perioperative management of opioid use disorder patients who are taking buprenorphine/naloxone.
INTRODUCTION– OPIOID USE DISORDER
An estimated 2.1% of the United States population may have opioid use disorder (OUD), while a global estimate is currently unknown (1). The diagnosis of OUD, per the Diagnostics Statistical Manual V, requires the patient to meet a least two out of eleven criteria within twelve months, with severity of disease defined by the number of criteria fulfilled (Table 1) (2). Multiple management options are available for OUD patients, with opioid agonist therapy – including buprenorphine/naloxone, methadone, slow-release morphine, and injectable hydromorphone or diacetylmorphine (heroin) – being the mainstay of treatment (3).
Buprenorphine/naloxone (Suboxone®, Zubsolv®, or Bunavail®), typically dosed between 8~24 mg of buprenorphine daily for OUD, is an evidence-based therapy for OUD that is becoming increasingly common. A 2014 Cochrane Review on buprenorphine’s effectiveness for OUD found that buprenorphine is more effective than placebo at all dose levels at retaining OUD participants in treatment, while high doses (≥16 mg) help suppress illicit opioid use when detected by urine drug screen(4). Compared to methadone, buprenorphine at medium (7~15 mg) and high doses were equivalent to methadone’s medium (40~85 mg) and high (≥85 mg) doses respectively regarding treatment retention, self-reported opioid use, and detection of illicit opioid use by urine drug screen.
Though similar in effectiveness to methadone, buprenorphine/naloxone (BUP/N) has been recommended as the first-line treatment for OUD over methadone in some jurisdictions due to its relative safety profile (3). Buprenorphine is six-fold safer than methadone for overdose risk due to a lower respiratory depression potential (3,5,6). Other advantages including more cost-effectiveness than methadone, and easier and faster titration of BUP/N over days, in contrast to weeks for methadone, which may help decrease relapse rates. With the growing opioid and fentanyl overdose crises affecting much of North America, efforts to expand access to OUD treatment are underway. Thus, anesthesiologists will likely see more patients on buprenorphine/naloxone prior to elective or emergency surgery.

Table 1. Opioid Use Disorder (OUD) criteria, from the Diagnostic and Statistical Manual of Mental Disorders V1. Severity of OUD depends on the number of criteria fulfilled (2~3 = mild, 4~5 = moderate, ≥6 = severe).
BUPRENORPHINE AND NALOXONE – PHARMACOLOGY
Pharmacodynamics of buprenorphine
Buprenorphine is a thebaine-based opioid with partial μ (mu)-receptor agonism (7). What makes buprenorphine unique among opioids are its low intrinsic activity, high affinity, and slow dissociation for the μ-receptor (8). It is 30 times more potent than morphine, but purportedly has as a ceiling μ-agonist effect somewhere between 8 and 16 mg. Buprenorphine’s onset after sublingual administration is between 30 and 60 minutes, peaking at 100 minutes (9). Higher doses result in prolonged duration: 4~12 hours for 4~8 mg, 24 hours for 8~12mg, and 2~3 days for ≥12 mg. Because buprenorphine binds so strongly to the μ-receptor (Ki = 0.22, with a lower Ki meaning higher binding affinity), it can displace other full opioid agonists with higher Ki (causing opioid withdrawal symptoms) or prevent them from binding to μ- receptors (leading to ineffective analgesia) (Table 2).
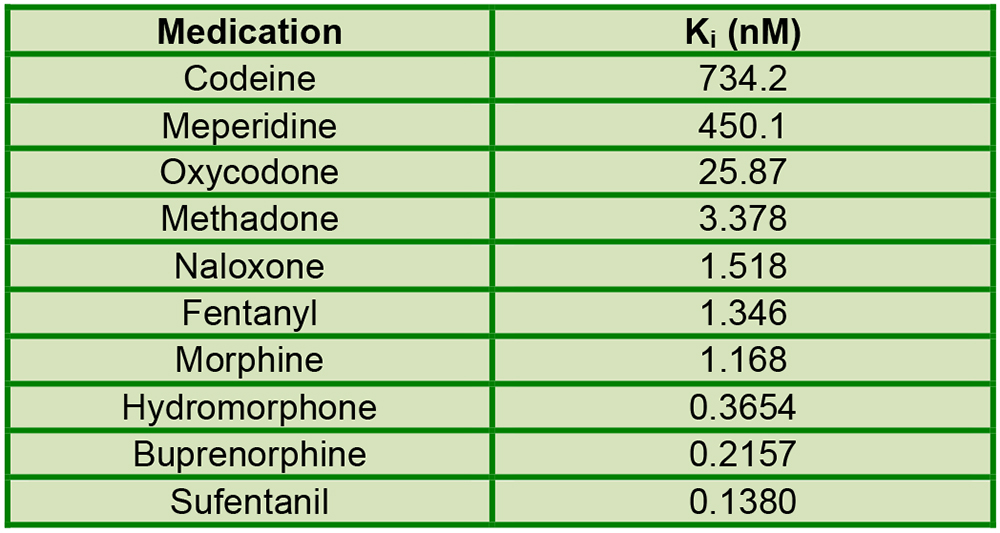
Table 2. Ki of commonly used opioids and naloxone to μ-opioid receptors (2). Lower Ki signifies higher binding affinity.
Buprenorphine also has κ (kappa)-receptor antagonism, which may be involved in not only decreasing the risk of respiratory depression related to opioid overdose, but also treating depression, stress, and addiction (9). However, respiratory depression may still occur when used in conjunction with non-opioid sedatives like benzodiazepines (7).
Pharmacokinetics of buprenorphine
Buprenorphine/naloxone exists in sublingual tablet, sublingual film and buccal film, with increasing oral mucosal absorption and bioavailability of buprenorphine respectively (30~52%) (7,10). Buprenorphine formulations without naloxone (i.e. Butrans® transdermal patch, Subutex® sublingual tablet) are prescribed for other indications such as chronic pain, but are generally not recommended for treating OUD due to diversion risk. Buprenorphine is highly lipophilic, distributing rapidly to tissues but releasing slowly from tissues. It therefore exhibits a depot-like effect, with higher doses resulting in longer duration. 96% of the drug binds to alpha- and beta-globulins. In the liver, buprenorphine utilizes the CYP3A4 system to metabolize via N-dealkylation into norbuprenorphine, with modification by glucuronidation. Norbuprenorphine binds to opioid receptors in vitro, but has not been studied clinically. Buprenorphine and its metabolites are eliminated and excreted via urine (30%) and feces (69%). Renal or hepatic impairment do not appear to significantly impact buprenorphine’s effects.
Pharmacology of naloxone
Naloxone is a μ-receptor antagonist. Naloxone is combined with buprenorphine to cause opioid withdrawal when buprenorphine is diverted parenterally for its euphoric effects. The 4:1 ratio with buprenorphine (i.e. buprenorphine: naloxone 2mg:0.5mg) produces intense opioid withdrawal symptoms when injected, while the 8:1 ratio is less effective (7). Given sublingually, naloxone only has 7~9% bioavailability, with no clinical significance (10). Naloxone undergoes direct hepatic glucuronidation to naloxone-3-glucuronide for urinary excretion.
PERIOPERATIVE MANAGEMENT OF PATIENT ON BUPRENORPHINE/NALOXONE
Deciding on continuing, reducing, or discontinuing buprenorphine/naloxone
Buprenorphine prescribed for chronic pain are at sufficiently low doses (typically 150~900 mcg sublingually twice a day or 5~20 mcg/hr transdermal patch) and the general recommendation is to continue buprenorphine perioperatively.
For OUD patients on BUP/N, there are currently no society or national guidelines nor trials to guide the perioperative management of BUP/N; only Anderson et al have published their institution’s protocol in managing this issue (9). While there are no evidence or trials, the decision to continue, reduce or discontinue BUP/N should be individualized to the patient. The advantages, disadvantages and suggested indications for continuing, reducing, or discontinuing BUP/N are described in Table 3.
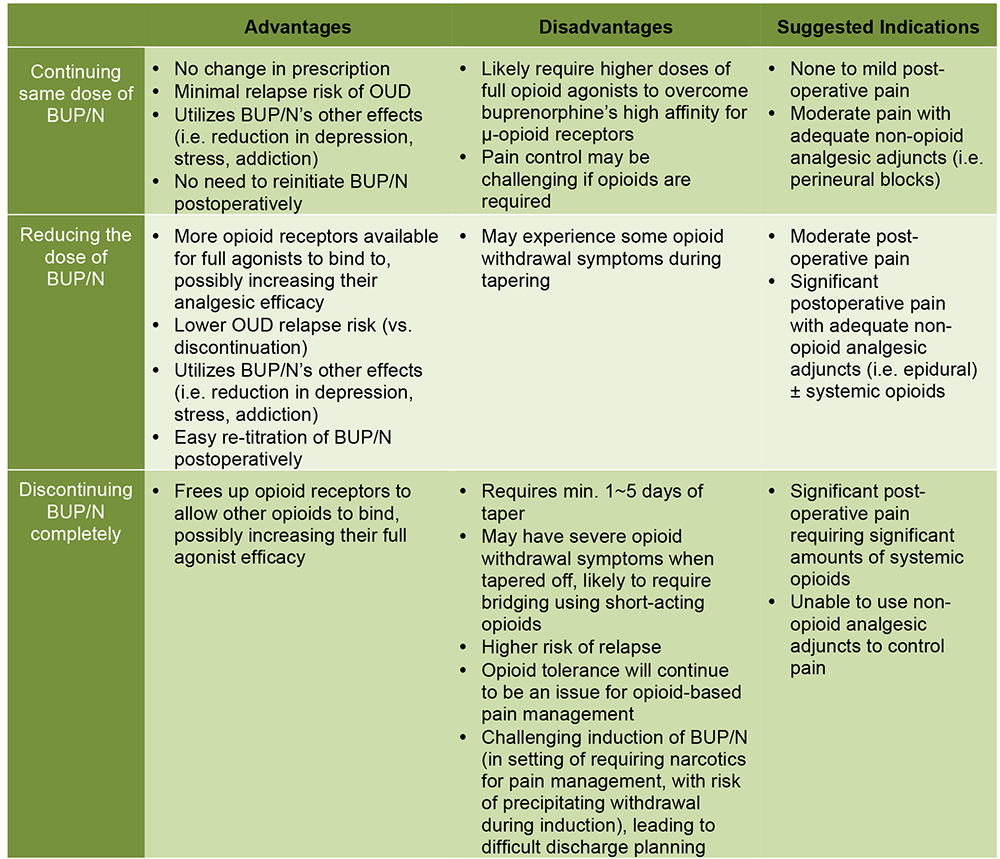
Table 3. Advantages, disadvantages and suggested indications for continuing, reducing, or discontinuing buprenorphine/naloxone perioperatively. (BUP/N = buprenorphine/naloxone, OUD = opioid use disorder)
What makes the perioperative management of BUP/N difficult to manage are the multiple considerations, including BUP/N itself and the patient population. Though complex, perioperative management of BUP/N can be summarized into two questions:
- Can this patient’s pain be managed perioperatively while on buprenorphine/naloxone (at the same or reduced dose)?
- If buprenorphine/naloxone is discontinued, is there a well-defined plan to reinitiate buprenorphine/naloxone postoperatively when appropriate to avoid relapse?
The high doses of BUP/N used for OUD pose a significant concern regarding buprenorphine’s interference with opioidbased analgesia. Buprenorphine’s high affinity and slow dissociation may prevent full opioid agonists with higher Ki from binding to μ-receptors and relieving pain, but buprenorphine’s low intrinsic activity may not provide sufficient analgesia by itself. Although unlikely to impact procedures with minimal postoperative pain, BUP/N may make pain control a challenge for major surgeries associated with significant pain requiring opioids. For example, a patient on BUP/N who underwent an emergency decompression and stabilization of C6-C7 fracture had higher anaesthetic and analgesic requirements, than during a subsequent C5-T11 arthrodesis with posterior instrumentation after abstaining from BUP/N for 5 days (11). Anderson et al recommend stopping BUP/N for surgeries known to have moderate to major postoperative pain to give full agonist opioids better analgesic efficacy (9).
This issue is complicated by the patients themselves: OUD patients are usually opioid tolerant, anxious, and more sensitive to pain, whether due to decreased pain threshold or hyperalgesia secondary to chronic opioid exposure (9,12). To illustrate this, Chern et al found similar high analgesic requirements in a patient who was on BUP/N for her first gynecological surgery versus off BUP/N prior to her second operation. MacIntyre et al found that patients on buprenorphine who did not receive their dose on the morning of their surgery required more analgesics in the first 24 hours postoperatively (13). Consequently, discontinuing BUP/N does not necessarily simplify pain management afterwards.
Managing postoperative pain while on BUP/N is still feasible. Kornfeld et al described seven such cases who underwent major surgeries (i.e. right-sided colectomy, knee arthroplasties, bilateral mastectomies, etc.) and treated the pain satisfactorily using perineural, neuraxial, and/or intravenous patient-controlled analgesia (IV-PCA) (14). Advantages of continuing BUP/N include avoiding the need and difficulty of re-initiating buprenorphine in a patient with acute pain, and utilizing the potential κ-antagonism effects to reduce depression and stress reduction.
The relapse risk can also be minimized if BUP/N is continued perioperatively. Relapse rates after tapering off BUP/N reaches above 90% (3). Discontinuing BUP/N several days pre-operatively will result in opioid withdrawal symptoms, often necessitating a bridge of short-acting opioids during the taper. Furthermore, the surgery, the postoperative pain, the fear of being judged, and the hospital setting are stressful and may trigger an OUD patient to leave against medical advice to seek illicit opiates (15). BUP/N continuation may help mitigate relapse risk while under psychosocial stress and therefore improve the patient’s long-term postsurgical success.
The current medical literature is focussed on continuing versus discontinuing BUP/N perioperatively (i.e. “To Stop or Not, That is the Question”(9)), but a third option exists: decreasing BUP/N to a smaller dose. Interestingly, this choice is not often mentioned in the literature, with one case report of a patient who developed severe refractory pain after a Clagett window closure and achieved adequate comfort once her BUP/N dose was reduced to 8 mg (16). Decreasing the BUP/N dose offers multiple advantages. A smaller dose of BUP/N will expose more μ-receptors for other opioids to bind to, potentially improving analgesic efficacy as suggested by the case report. Moreover, having some BUP/N present permits easy up-titration to pre-surgical doses, avoiding the risk of precipitating withdrawal. Patients may still experience opioid withdrawal symptoms while on a lower BUP/N dose preoperatively, but it is unlikely to be of the same severity as that from complete BUP/N discontinuation. Consequently, BUP/N reduction can be considered in lieu of discontinuation.
Lastly and most importantly, the value of a multidisciplinary team and a biopsychosocial approach to perioperative BUP/N and pain management cannot be understated. Consulting addiction medicine specialists early can help with the decision-making of continuing, reducing or discontinuing BUP/N preoperatively, and postoperatively during the management of postoperative pain and OUD. Collaborating with surgeons, patients’ family physicians, and clinical support staff like social workers and outreach nurses will assist with surgical planning to optimize postoperative analgesia and minimize emotional distress.
Continuing or reducing buprenorphine/naloxone in elective surgery
The standard anaesthetic history and physical exam, a brief history of illicit drug use (including types of drugs and last exposure), medical consequences (i.e. intravenous drug use: HIV, infective endocarditis, viral hepatitis, etc.), and buprenorphine/naloxone dose and duration of treatment should be obtained. Rapport building is particularly important with OUD patients. Avoiding words with judgemental connotations like “addict”, “habit” and “abuse”, and using objective medical terminology – “illicit drug use”, and “opiate use disorder” – will help achieve this. As emphasized above, involving relevant care providers (i.e. BUP/N prescriber) and addiction medicine specialists will assist with developing an individualized perioperative plan and addressing the patient’s concerns. The preanesthetic interview should inform the patient of realistic pain expectations, typical course of acute pain, and analgesic options, with reassurance that the pain will be treated. Lastly, one should consider prescribing preoperative acetaminophen, non-steroidal anti-inflammatory drugs (NSAIDs), and/or gabapentin when not contraindicated.
Intra-operatively, higher anaesthetic doses may be needed (11). For pain management, multimodal non opioid analgesics should be fully explored and administered if appropriate, including but not exclusively intravenous medications, perineural and neuraxial nerve blocks (Table 4). If opioids are required, then a higher opioid consumption should be expected than the typical non-OUD patient. Looking at binding affinities (Table 2), sufentanil and hydromorphone are the ideal opioids to compete with buprenorphine for μ-receptor binding sites.

Table 4. Non-opioid analgesic options.
Post-surgery, acute pain service and/or addiction medicine specialists should be recruited for postoperative pain management. One may consider splitting BUP/N into three to four times a day (q6~8 hours) dosing to maximize buprenorphine’s analgesic properties (9,12). Non-opioid analgesics should be prescribed if not contraindicated: regular acetaminophen and NSAIDs, intravenous lidocaine or ketamine infusion, and/or continuous perineural or epidural analgesia. Should a short period of postoperative ventilation be needed for other reasons, then consider dexmedetomidine for its sedative and analgesic effects.
If opioids are needed, then IV-PCA should be provided, with anticipation for higher doses than opioid naïve patients. Hydromorphone is preferred, and fentanyl is a suitable alternative given its high potency. Morphine at high doses, despite its higher Ki value, has also been used successfully(14,17). If multimodal analgesia is unsuccessful, then reducing or, as last resort, discontinuing the BUP/N dose should be considered(16). Once the acute pain phase is over, the next steps are tapering off opioids and returning to the original dose of BUP/N (if reduced).
Many healthcare providers raise concerns about the high opioid consumption of OUD patients and “feeding their habit” or addiction. Pain is a subjective perception. As stated above, not only are OUD patients opioid tolerant, but they also have lower pain thresholds or hyperalgesia. When patients are in pain, opioids provide pain relief and minimal euphoria (12). Our institution has taken the harm reduction approach: we would rather treat pain, than risk the patients leaving hospital against medical advice to find relief using illicit opiates and relapsing, or developing complications from lacking care. Measures to prevent diversion (i.e. opioids in oral solution, locked PCA pump) and frequent assessments for sedation from excessive opioids should be routine care. With this in mind, higher acuity monitoring early postoperatively (i.e. 6~12 hours) may be necessary to manage high opioid doses.
Discontinuing buprenorphine/naloxone for elective surgery
In this situation, it is even more crucial to liaise with the patient’s BUP/N provider and the addiction medicine service to safely taper off BUP/N preoperatively. The timing to stop BUP/N is directly related to the dose: 24 hours preoperatively if on 0~4 mg of buprenorphine, 48 hours for 4~8 mg, 72 hours for 8~12 mg, and 3~5 days if ≥12 mg (9). Whether to bridge with short-acting opioids during the taper should be discussed with the patient, provider and addiction medicine specialist, the alternatives include morphine, hydromorphone, and methadone(9,12).
Intraoperative and postoperative considerations are similar to those on BUP/N, with the expectation that OUD patients off BUP/N will still have high opioid requirements. The new complicating matter is restarting buprenorphine/naloxone. Re-introducing BUP/N too early may precipitate withdrawal and worsen acute pain (leading to relapse and distrust of the healthcare system), while discharging patients off BUP/N leaves them vulnerable to relapse and opioid overdose. Again, working with addiction medicine and/or the BUP/N provider will help manage this issue. Generally, BUP/N could be reinitiated once the pain is tolerable and requires minimal opioids. The patient should ideally be restarted on BUP/N before discharge, which may mean prolonging hospital admission. If BUP/N cannot be initiated in-hospital and the patient needs opioids, then safe opioid prescribing practices apply: limited number of oral narcotics prescribed to take home, daily witnessed ingestion by a pharmacist or trusted relative, and close community follow-up, ultimately aiming to restart BUP/N urgently before the patient relapses.
Buprenorphine/naloxone management in urgent or emergent surgery
Inquiring about the time and amount of the last BUP/N dose is crucial for assessing its impact on opioid withdrawal and anaesthetic requirements. Possible sources of this information include the patient, the patient’s pharmacy, and the patient’s computerized community medication list if available.
Management of BUP/N during urgent or emergency surgery is unclear. Anderson et al recommend stopping BUP/N and providing IV-PCA postoperatively (9). However, at our institution, we suggest continuing BUP/N perioperatively. Postoperative analgesic requirements will still be affected by buprenorphine due to its long duration of action. By the time the buprenorphine is eliminated several days later, postoperative acute pain should be improving anyway, with the added problem of re-introducing BUP/N later. The gradual elimination of buprenorphine becomes an unpredictable variable affecting opioid titration. If pain control is unmanageable, then the BUP/N dose reduction should be considered. Disposition to a monitored high acuity bed for opioid titration is suggested.
CONCLUSION
With the unique pharmacology of buprenorphine/naloxone and the complicated psychosocial, opioid tolerant nature of patients with opioid use disorder, having a multidisciplinary team approach comprising of surgeons, anaesthesiologists, buprenorphine/naloxone providers and addiction medicine specialists can help achieve satisfactory pain control and a smooth perioperative course.
SUMMARY
- Illicit opiate use is a growing global problem and there are several described opioid agonist therapies.
- Buprenorphine is a partial μ-opioid agonist with long duration of action and a higher binding affinity
- When not contraindicated maximise non-opioid analgesic options described in Table 4.
REFERENCES AND FURTHER READING
- Grant BF, Saha TD, Ruan WJ, Goldstein RB, Chou SP, Jung J, et al. Epidemiology of DSM-5 Drug Use Disorder: Results From the National Epidemiologic Survey on Alcohol and Related Conditions-III. JAMA psychiatry 2016;73:39-47.
- Substance-Related and Addictive Disorders. Diagnostic and Statistical Manual of Mental Disorders 5th ed. American Psychiatric Association; 2013. https://doi.org/10.1176/appi.books.9780890425596.dsm16 (accessed on August 22, 2017)
- Provincial Opioid Use Disorder Treatment Guideline Committee. A Guideline for the Clinical Management of Opioid Use Disorder. 2017. http://www.bccsu.ca/care-guidance-publications/ (accessed on July 15, 2017)
- Mattick RP, Breen C, Kimber J, Davoli M, Rp M, Breen C, et al. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane database Syst Rev 2014;2:CD002207.
- Marteau D, McDonald R, Patel K. The relative risk of fatal poisoning by methadone or buprenorphine within the wider population of England and Wales. BMJ Open 2015;5:e007629.
- Bell JR, Butler B, Lawrance A, Batey R, Salmelainen P. Comparing overdose mortality associated with methadone and buprenorphine treatment. Drug Alcohol Depend 2009;104:73-7.
- Suboxone monograph. 2017. https://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafety InformationforPatientsandProviders/UCM227949.pdf (accessed on August 20, 2017)
- Volpe DA, Tobin GAM, Mellon RD, Katki AG, Parker RJ, Colatsky T, et al. Uniform assessment and ranking of opioid Mu receptor binding constants for selected opioid drugs. Regul Toxicol Pharmacol Elsevier Inc.;2011;59:385-90.
- T. Anderson T, Quaye A, Ward N, Wilens T, Hilliard P BC. To Stop or Not, That Is the Question.Acute Pain Management for the Patient on Chronic Buprenorphine. Anesthesiology 2017;126:1180-6.
- Chiang CN, Hawks RL. Pharmacokinetics of the combination tablet of buprenorphine and naloxone. Drug Alcohol Depend 2003;70:39-47.
- Khelemsky Y, Schauer J, Loo N. Effect of buprenorphine on total intravenous anesthetic requirements during spine surgery. Pain Physician 2015;18:E261-4.
- Alford DP, Compton P, Samet JH. Annals of Internal Medicine Perspective Acute Pain Management for Patients Receiving Maintenance Methadone or Buprenorphine Therapy. Ann Intern Med 2006;149:698-707.
- MacIntyre PE, Rusel RA, Usher KAN, Gaughwin M, Huxtable CA. Pain relief and opioid requirements in the first 24 hours after surgery in patients taking buprenorphine and methadone opioid substitution therapy. Anaesth Intensive Care 2013;41:222-30.
- Kornfeld H, Manfredi L. Effectiveness of full agonist opioids in patients stabilized on buprenorphine undergoing major surgery: a case series. Am J Ther 2010;17:523-8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19918165
- Onukwugha E, Saunders E, Mullins CD, Pradel FG, Zuckerman M, Weir MR. Reasons for discharges against medical advice: a qualitative study. Qual Saf Health Care 2010;19:420-4.
- Huang A, Katznelson R, de Perrot M, Clarke H. Perioperative management of a patient undergoing Clagett window closure stabilized on Suboxone(®) for chronic pain: a case report. Can J Anaesth 2014;61:826-31.
- Mercadante S, Villari P, Ferrera P, Porzio G, Aielli F, Verna L, et al. Safety and Effectiveness of Intravenous Morphine for Episodic Breakthrough Pain in Patients Receiving Transdermal Buprenorphine. J Pain Symptom Manage 2006;32:175-9.



