Basic Sciences
KEY POINTS
- Pulmonary hypertension (PH) is defined as a mean pulmonary artery pressure 20 mm Hg at rest.
- Perioperative management aims to minimise the effects of anaesthesia and surgery on right ventricular function in patients at risk of such dysfunction as a result of chronic PH.
- Right ventricular function can be compromised if there is (1) a reduction in right ventricular perfusion if systemic vascular resistance (SVR) falls and/or (2) a reduction in right ventricular stroke volume if pulmonary vascular resistance (PVR) increases.
- Falls in SVR can be minimised with the judicious use of vasopressors and intravenous fluids.
- Acute increases in PVR can be minimised by avoiding direct pulmonary vasoconstriction precipitants, namely, hypoxia, hypercarbia, acidosis and hypothermia.
- Regional anaesthetic techniques, where possible, are often preferable as they are less likely to compromise SVR and PVR in the manner described above
INTRODUCTION
Pulmonary hypertension (PH) is defined as a pulmonary artery catheterisation pressure of 20 mm Hg at rest and pulmonary vascular resistance .3 Wood units (WU).1 Severity is classified as mild (20-40 mm Hg), moderate (40-55 mm Hg) and severe (.55 mm Hg).
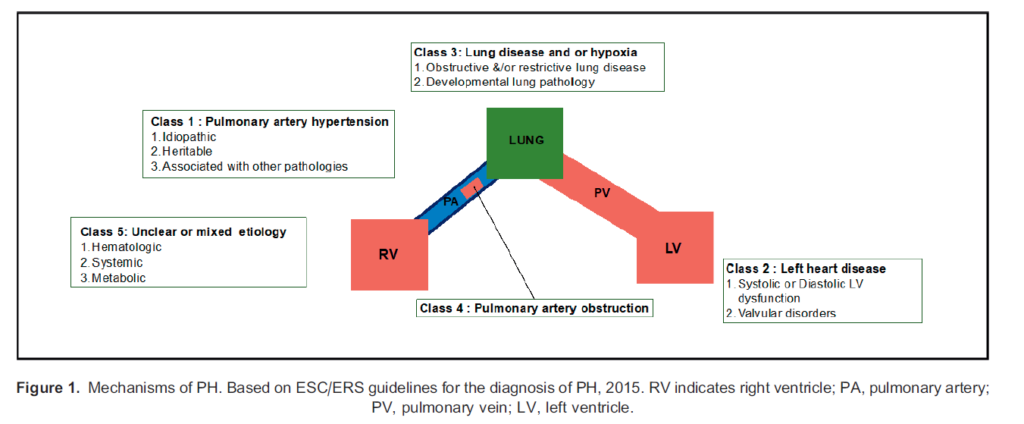
A Joint Task Force of the European Society of Cardiology and European Respiratory Society (ESC/ERS) provided a classification for the aetiology of PH (Figure 1).
- Class 1: disease of the pulmonary arterial vasculature
- Class 2: due to left heart disease
- Class 3: due to lung disease or hypoxia
- Class 4: due to pulmonary artery obstruction
- Class 5: multifactorial aetiologies
The European epidemic of PH in the mid-1960s, following widespread use of the amphetamine-like prescription appetite suppressant Aminorex, resulted in recognition by the World Health Organisation.2
The underlying causes vary between countries, with low-income countries burdened by infective aetiologies such as HIV, rheumatic heart disease and schistosomiasis, whilst the aetiology in resource-rich, high-income countries is largely secondary to chronic obstructive pulmonary disease (COPD) and left ventricular failure secondary to arterial disease. The annual incidence of PH is increasing in the United Kingdom; current estimates predict 8 cases per million people.
Demographically, patients diagnosed with PH are in the fifth decade of life (53 6 14 years) and are more likely to be female (25:1) with class 2 or 3 disease aetiology. The reason for the inequality between sexes remains unclear. Multicentre US-based registry data (REVEAL Registrye) highlight a racial disparity: 73% Caucasians, 13% African Americans and 9% Hispanic.3 Early disease recognition is often challenging because of nonspecific symptoms, leading to a delay in diagnosis of up to 2 years, with a clear influence on disease prognosis, especially in the case of systemic sclerosis.
Right Ventricular Physiology and Pathophysiology
Right Ventricular Structure and Blood Supply
The right ventricle (RV) pumps deoxygenated blood into the high-volume, low-pressure lung vasculature. This system requires less stroke work when compared with the left ventricle (LV), with a consequent 6-fold reduction in right ventricular mass.
Usually, the right coronary artery (RCA) supplies the right heart. In the absence of pathology, such as hypertrophy, the relatively low intraventricular pressure allows flow through the RCA throughout the cardiac cycle, although diastolic flow predominates in the more distal vessels. The RV perfusion pressure is therefore dependent on the mean arterial pressure rather than diastolic pressure alone; this differs from the situation with LV perfusion.4
Pulmonary Vascular Resistance
PH may or may not be associated with a raised pulmonary vascular resistance (PVR). Nevertheless, anaesthetic management is frequently based on avoiding acute increases in PVR that can exacerbate RV dysfunction. Understanding factors that alter PVR is therefore critical in the perioperative management of these patients.
The pulmonary vasculature is thin walled and easily distensible; resistance is low and dependent on passive physical factors such as gravity, posture and the pressure differential between alveoli and capillaries. The alveolar-capillary pressure differential is increased at times of low cardiac output (capillaries easily collapse), lung underexpansion (forces holding alveoli open are reduced), or lung overexpansion (pulmonary capillaries are compressed). Other factors actively alter PVR by affecting pulmonary vascular muscle tone (Table 1).
Pathophysiology
Chronic changes in RV afterload, as observed in PH, can negatively impair right coronary perfusion, because of the effect of myocardial adaptive changes on the coronary vessels, similar to what occurs with systemic hypertension impairing LV myocardial perfusion. This may make the RV of a patient with PH more susceptible to ischaemia if perfusion pressure drops.

In health, the RV can dilate and accommodate higher volumes more easily than the LV does. However, chronic elevation in pulmonary artery pressure can lead to hypertrophic changes with an increase in RV wall thickness and a reduction in internal radius. At this stage, the RV responds poorly to acute elevations in pulmonary artery pressure, which may precipitate acute RV failure secondary to its overdistention, both macroscopically into the LV (via intraventricular septal deviation) and microscopically due to sarcomere overstretching. Ultimately, both mechanisms lead to a reduction in output from both ventricles for a given preload, resulting in cardiogenic shock.
PREOPERATIVE ASSESSMENT AND MANAGEMENT
History
Symptoms of chronic PH are often nonspecific, leading to a delay in diagnosis. Symptoms can be classified into 4 categories:
- Systemic symptoms of reduced cardiac output (eg, dyspnoea and fatigue)
- Primary cardiac symptoms: RV failure leads to exertional chest pain and syncope with weight gain and fluid overload.
- Intrathoracic mechanical symptoms: Dilation of the pulmonary artery trunk can lead to compression of the left recurrent laryngeal nerve, presenting as hoarseness.
- Symptoms relating to the underlying aetiology (Figure 1)
- Symptoms suggestive of upper airway disease (obstructive sleep apnoea), chronic respiratory disease (COPD, venous thromboembolism), connective tissue diseases, endocrine and metabolic disorders
- History of illicit drug use, alcohol intake, cigarette smoking and HIV risk factors5
Examination
The clinical signs can reflect the following:
- RV failure: elevated jugular venous pressure and associated signs of fluid overload, such as tender hepatomegaly, ascites and peripheral oedema
- Tricuspid valve regurgitation causing a pansystolic murmur and prominent ‘v’ wave on the central venous pressure waveform
- Elevated pulmonary artery pressure leading to an increased intensity of the second heart sound (loud P2)
Investigations
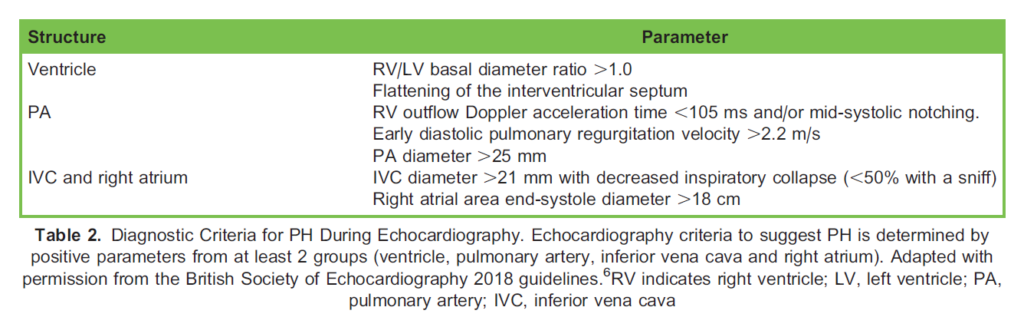
Clinical investigations can be used to both screen and diagnose patients with PH. Basic screening investigations should include an echocardiogram; the 2018 British Society of Echocardiography outlines criteria for diagnosis of PH (Table 2).6 Needless to say, this echocardiogram can also comment on the size and function of the RV to help delineate the end-organ consequences of a diagnosis of PH.
Additional tests that may provide further information include the following:
- Laboratory biochemistry (cardiac disease may cause associated renal dysfunction, either directly or as a consequence of pharmacologic treatment for heart failure)
- Elevated brain natriuretic peptide may be suggestive of RV 6 LV dysfunction
- Electrocardiography (right axis deviation, right bundle branch block and finally R wave/S wave ratio .1 in lead V1 are suggestive of RV strain)
- Chest x-ray (may show evidence of cardiorespiratory pathology and PH with enlargement of central pulmonary vessels and right atria and ventricular enlargement)
Pulmonary function tests (PFTs) are a noninvasive method of assessing lung function. Patients presenting with PH should undergo PFTs, especially those with multifactorial (class 5) or lung-specific disease (class 3). Attention should be focused on reversibility, common airway patterns (forced expiratory volume in 1 second/forced vital capacity ratios) and gas transfer (diffusing capacity for carbon monoxide) rates.
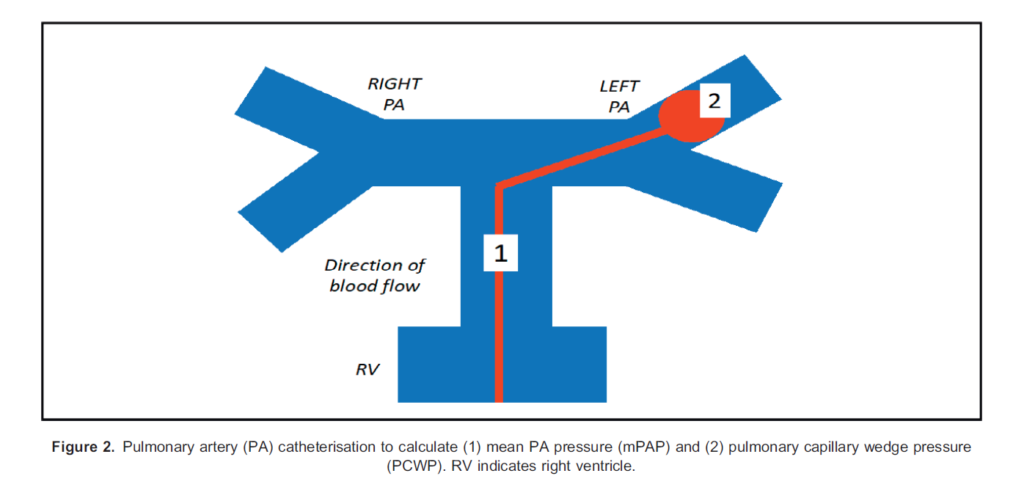
The gold standard for diagnosis remains right heart catheterisation (RHC; Figure 2).7 Parameters of note during RH are as follows:
- Pulmonary
- Pulmonary artery pressure
- PVR
- Pulmonary capillary wedge pressure (Figure 2)
- Cardiac
- Right ventricular pressure
- Right atrial pressure
- Cardiac output
- Cardiac index
Cardiopulmonary exercise testing (CPET) remains a fundamental noninvasive and dynamic tool to determine functional capacity and exercise limitation whilst attaining information regarding metabolic capacity and cardiorespiratory function.8 CPET can provide supporting information regarding disease patterns but, unlike RHC, will not diagnose PH.
Patients with PH may be unable to achieve adequate perioperative oxygenation, due to impaired cardiac output in the presence of elevated mean pulmonary artery pressures (mPAPs) and possible RV dysfunction. The most recent European guideline (2015 ESC/ERS) uses peak VO2 (maximal oxygen consumption in mL/kg/min) and VE/V0CO2 (ventilatory equivalents for carbon dioxide) to categorise PH patients into low-, medium- or high-risk categories, although the document acknowledges that the incremental ramp protocol used for CPET is not standardised for PH patients.
Chronic Disease Management
In the United Kingdom, chronic disease management for PH is initiated by 1 of 8 tertiary centres when the mPAP is 25 mm Hg. There are effective drug treatments available specific to each aetiology.
Pulmonary endothelial cells release nitric oxide, which stimulates vascular smooth muscle cells to produce cyclic guanosine monophosphate (cGMP), an intracellular pathway, which then results in cyclic adenosine monophosphate–mediated pulmonary vascular smooth muscle cell relaxation.
Basic drug classes for the pharmacologic management of PH include the following:
- Phosphodiesterase-5 inhibitors (sildenafil): inhibition of this enzyme prevents the breakdown of intracellular cGMP
- Guanylate cyclase activators (riociguat): stimulates production of cGMP via the same pathway as nitric oxide
- Prostacyclin receptor agonists (selexipag): these selectively bind the prostaglandin I receptor, a G-protein–coupled receptor mediating vasodilation.
- Endothelin receptor antagonist (ERA; ambrisentan, bosentan): ERAs inhibit the action of endothelin-1, an amino acid produced by endothelial cells that mediates vascular constriction.
Active respiratory agents should be continued throughout the perioperative period, with conversion to inhaled or intravenous routes when enteral is unavailable.
Other therapies may include diuretics and supplemental oxygen. A suspected prothrombotic state in PH or a thromboembolic aetiology may mean patients are also treated with anticoagulants, but the heterogeneous nature of PH means this is often patient specific.
INTRAOPERATIVE CARE
Preoperative Decision Making
It is important that patients are assessed and optimised by a specialist in PH prior to elective surgery; the presence of PH increases perioperative mortality, so the risks and benefits of surgery need to be balanced against each other.
This may mean a multidisciplinary discussion to identify whether surgery is in the patient’s best interests or not. A condition that might normally be treated surgically may be treated conservatively if that is the correct decision for that patient. This balance of risks needs to consider the specialist’s opinion, the patient’s wishes, the surgical pathology and the site and magnitude of surgery; this decision making is probably as important as the intraoperative management of the patient’s physiology.
Cardiovascular Requirements
The two main intraoperative goals when delivering anaesthesia for patients with PH aim to maintain RV output.
First, to avoid compromising the RV perfusion that takes place more or less continuously during the cardiac cycle, it is essential to maintain systemic blood pressure. This means maintaining systemic vascular resistance (SVR) by counteracting the reduction that often occurs in the presence of vasodilating anaesthetic agents. This can be achieved with vasopressors administered peripherally (metaraminol or phenylephrine) or centrally (eg, noradrenaline or vasopressin).
Noradrenaline theoretically helps maintain RV function via b1 adrenoceptor agonism and b2-mediated PVR reduction, whilst vasopressin offers a similar potential benefit by appearing to not cause pulmonary vascular constriction. However, these benefits have not been proven to effect outcomes in clinical trials, and all of the agents have been used safely and effectively in clinical practice.9
Second, it is important to avoid acute elevations in PVR that will compromise RV ejection. This can occur with preserved RV function but will be more severe in an impaired ventricle. As mentioned above, if the RV dilates in response to an increased afterload, it may also lead to interventricular septum distortion, compromising LV ejection.10 PVR can be influenced both pharmacologically and nonpharmacologically.
Nonpharmacologic measures include avoiding physiological precipitants of pulmonary vasoconstriction such as hypoxia, hypercapnia (aim PaCO2 4-4.5 kpa), over- and underinflation of lungs (6-8 mL/kg ideal body weight), acidosis, pain and hypothermia.
Pharmacologic measures include the implementation of pulmonary vasodilator agents that can be administrated by both inhaled and intravenous routes. Agents include synthetic analogues of prostacyclin (iloprost and epoprostenol) and gases (nitric oxide); these agents and, more importantly, experience of their use may be available only in specialist centres.
It may also be necessary to directly support cardiac output with an inotropic agent. Calcium sensitisers (eg, levosimendan) and phosphodiesterase inhibitors (eg, milrinone) may be helpful as they can reduce PVR and therefore be more beneficial in right heart failure. However, no selective right heart inotrope exists. Note that inotrope use may require concurrent vasopressor administration to attenuate the vasodilation that occurs with some inotropic agents.
ANAESTHESIA TECHNIQUES
The anaesthetic technique most appropriate for each patient encounter will depend on patient, anaesthetic and surgical factors.
Patient factors obviously include relevant additional medical conditions and patient choice but will also need to take severity of PH and associated cardiac dysfunction into account.
Anaesthetic and surgical factors include patient positioning, duration of surgery, site of operation and risk of cardiovascular instability (eg, blood loss); these are clearly applicable with or without PH.
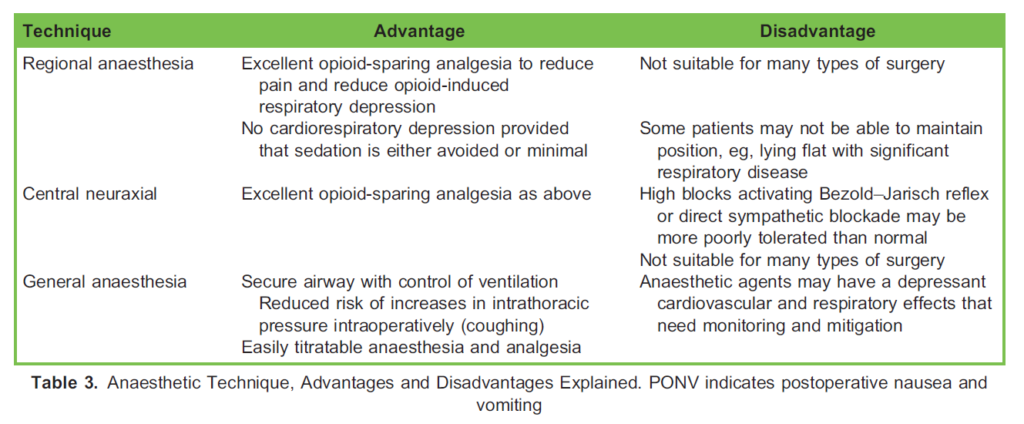
General and regional anaesthesia—be that central neuraxial, peripheral regional or local anaesthesia—are all potential options, and the key advantages and disadvantages are listed in Table 3. Whilst many of these advantages and disadvantages are generic, it is the avoidance of any unnecessary cardiac and respiratory depression that is of specific benefit in patients with PH, more so than in a fit and healthy patient, hence the role of opioid-sparing analgesia, avoidance of excessive sedation or good control of gas exchange.
Anticoagulation may be common and needs to be considered when opting for certain types of regional anaesthetic techniques.
Regional and Local Anaesthesia
Regional anaesthesia remains a safe and simple option to provide these intraoperative cardiovascular conditions with the added advantage of good, intra- and postoperative opioid-free analgesia. Clearly, some forms of surgery, such as orthopaedic or ophthalmic surgery, lend themselves to regional anaesthesia.
These advantages can be negated if regional anaesthesia is accompanied by any adverse effects of accompanying sedation. The use of sedation should therefore be considered very carefully.
Central Neuraxial Anaesthesia
Neuraxial anaesthesia can be applied safely to PH patients if attention is paid to achieving the cardiovascular goals described above. Relatively rapid systemic vasodilation can occur, which can compromise right-sided cardiac filling as well as RV coronary perfusion. If combined with inhibition of high thoracic cardioaccelerator autonomic nerve fibres, it can result in a profound negative chronotropic and inotropic response. In both instances, cardiovascular impairment in the presence of elevated mPAPs may lead to reduced respiratory perfusion, acute pulmonary hypertensive crisis and cardiovascular collapse, so care must be taken with the previously detailed cardiovascular targets.
General Anaesthesia
Regional or local anaesthesia is often a preferred option. However, there are circumstances in which general anaesthesia is the only choice, and it is still a safe option in PH patients if attention is paid to maintain the goals previously described.
Diligent control of CO2 and thus pH is important and is often more easily achieved via controlled ventilation. This usually necessitates endotracheal intubation, but for shorter cases in appropriate patients (eg, nonobese, low risk of aspiration, etc), a suitable second-generation supraglottic airway device (SAD) can be used.
Conversely, spontaneous ventilation techniques, usually but not exclusively involving an SAD, are usually avoided in patients with PH unless the disease is mild and the procedure is short.
Both intravenous and volatile anaesthetic agents have cardiorespiratory depressant effects, but induction and maintenance of anaesthesia can be safely achieved with both. All intravenous agents and all inhalational agents given up to a concentration of 1 minimum alveolar concentration have negligible effects on pulmonary vascular resistance.11
All agents reduce ventilatory drive and minute ventilation, but this is prevented by control of ventilation. There is reduced hypoxic pulmonary vasoconstriction but little direct effect on PVR, apart from with nitrous oxide, which can directly increase PVR and is therefore best avoided in patients with PH.
Cardiovascular effects vary depending on the agent used, but all generally decrease myocardial contractility, SVR, stroke volume and blood pressure. There may be differing effects on heart rate depending on the agent and rate of administration. All of these effects are predictable; appropriate use of monitoring and cardiovascular pharmacological support can help to ameliorate them and maintain the target of a maintained SVR (for RV perfusion and RV filling from venous return).
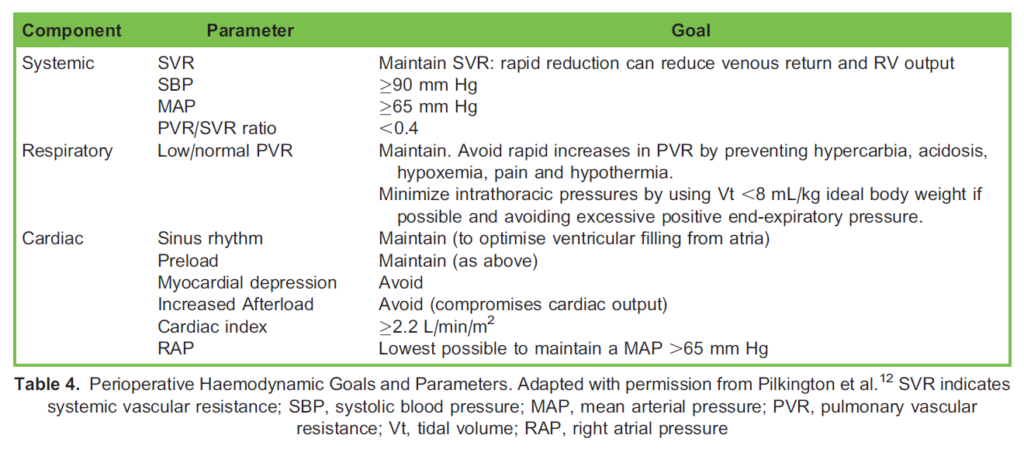
Patients should be monitored as per the standard recommended monitoring techniques described by the Association of Anaesthetists of Great Britain and Ireland. Additional monitoring will depend on the patient, and the surgery and may include invasive arterial and/or central venous pressure monitoring, pulmonary artery catheterisation or other cardiac output– monitoring techniques. Suggested perioperative haemodynamic parameters—when available from invasive monitoring—are suggested in Table 4.
POSTOPERATIVE CARE
PH carries significant perioperative mortality and morbidity. Patients should be monitored postoperatively in high-dependence areas. Postoperative goals reflect the cardiorespiratory parameters aimed at preventing elevations in PVR and maintaining SVR, as described (Table 4).
Good analgesia, ideally using opioid-sparing approaches including nonsedating analgesics (paracetamol, nonsteroidal antiinflammatory drugs), and local or regional analgesia should be applied where possible, as this assists in minimising PVR increases associated with hypoventilation as well as improving overall recovery.
There should also be minimal interruption of existing PH therapies.
Tachyarrhythmias are a common problem in PH patients. Unlike left heart diseases, arrhythmias are usually supraventricular, with ventricular rhythms being rare. Supraventricular arrhythmias can compromise both long- and short-term prognosis, with a potential for deterioration into right heart failure. Restoration and maintenance of sinus rhythm is highly desirable, with prophylactic antiarrhythmics being frequently used. Data regarding individual drugs are lacking, but using an agent with minimal negative inotropy should be considered.
Rhythms associated with haemodynamic instability should be treated as per resuscitation algorithms, using synchronised electrical cardioversion as appropriate.
PULMONARY HYPERTENSIVE CRISIS
A pulmonary hypertensive crisis (PHC) is characterised by acute elevation in PVR that overwhelms the ability of the RV to maintain output against the increased afterload and is associated with significant mortality (.50%).13 This can go on to negatively affect LV preload, LV cardiac output and diastolic coronary perfusion pressure, leading to myocardial collapse and cardiogenic shock.
Under normal physiological conditions, RV adaptation and remodelling help maintain RV output. However, intraoperative physiological changes secondary to physical (eg, patient positioning and pneumoperitoneum) or pharmacologic (anaesthetic or other drugs) factors can lead to decompensation. Rapid recognition of PHC is essential; whilst signs of cardiogenic shock are characteristically observed, confirmation generally requires invasive pressure measurements and/or bedside echocardiography (elevated right atrial and pulmonary pressures with decreasing cardiac output).
Treatment options are supportive or pharmacologic and reflect those previously discussed to control PVR:
- Supportive
- Treatment for a specific precipitating cause if possible (eg, pain, hypothermia)
- Supplemental oxygen
- Appropriate fluid administration (if hypovolaemia suspected)
- Ventilatory support to maintain CO2 and control pH if necessary; postoperative respiratory depression can be multifactorial
- Pharmacologic
- Cardiac output can be augmented with inotropic and chronotropic agents
- Systemic vascular resistance can be augmented with vasopressors
- Pulmonary vascular vasodilators can be given via the inhaled or intravenous route
SUMMARY
Patients with PH present for both elective and urgent cardiac and noncardiac surgery. Not all patients may have a preoperative confirmed diagnosis, so clinical assessment may be needed to aid diagnosis and determine the degree of cardiorespiratory compromise.
Intraoperative management should involve input from a physician experienced in the management of patients with PH. Postoperative care should be performed in an area of high acuity, with regional opioid-sparing techniques forming the mainstay of treatment.
REFERENCES
- Condon DF, Nickel NP, Anderson R, et al. The 6th World Symposium on Pulmonary Hypertension: what’s old is new. F1000Research. 2019;8(F1000 Faculty Rev-888).
- Montani D, Seferian A, Savale L et al. Drug-induced pulmonary arterial hypertension: a recent outbreak. Eur Respir Rev. 2013;22(129):244-250.
- Frost AE, Badesch DB, Barst RJ, et al. The changing picture of patients with pulmonary arterial hypertension in the United States: how REVEAL differs from historic and non-US contemporary registries. Chest. 2011;139(1):128-137.
- Crystal GJ, Pagel PS. Right ventricular perfusion: physiology and clinical implications. Anesthesiology. 2018;128(1):202218.
- Elliot CA, Kiely DG. Pulmonary hypertension. Contin Educ Anaesth Crit Care Pain. 2006;6(1):17-22.
- Augustine DX, Coates-Bradshaw LD, Willis J, et al. Echocardiographic assessment of pulmonary hypertension: a guideline protocol from the British Society of Echocardiography. Echo Res Pract. 2018;5(3):G11-G24.
- Rosenkranz S, Preston IR. Right heart catheterisation: best practice and pitfalls in pulmonary hypertension. Eur Respir Rev. 2015;24(138):642-652.
- Farina S, Correale M, Bruno N, et al. The role of cardiopulmonary exercise tests in pulmonary arterial hypertension. Eur Respir Rev. 2018;27(148):170134.
- Currigan DA, Hughes RJA, Wright CE, et al. Vasoconstrictor responses to vasopressor agents in human pulmonary and radial arteries: an in vitro study. Anesthesiology. 2014;121:930-936.
- Condliffe R, Kiely DG. Critical care management of pulmonary hypertension. BJA Educ. 2017;7(17):228-234.
- Sarkar MS, Desai PM. Pulmonary hypertension and cardiac anaesthesia: anesthesiologist’s perspective. Ann Card Anaesth. 2018;21(2):116-122.
- Pilkington SA, Taboada D, Martinez G. Pulmonary hypertension and its management in patients undergoing non-cardiac surgery. Anaesthesia. 2015;7(1):56-70.
- Simon M. Perioperative management of pulmonary hypertensive crisis. Adv Pulm Hypertens. 2013;12(1):38-39.



