Obstetrics Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding peripartum cardiomyopathy (PPCM):
a. The aetiology is well established
b. Diastolic dysfunction is a common feature
c. 80% of women make a full recovery
d. Incidence is high
e. It is usually a diagnosis of exclusion - The following ECG findings are associated with pregnancy:
a. Right axis deviation
b. ST-segment depression in limb leads
c. Q waves in lead III
d. T wave inversion in inferior leads
e. Premature beats - Concerning PPCM:
a. It commonly presents in the first trimester of pregnancy
b. It may present up to five months post-partum
c. Those affected have a left ventricular ejection fraction of >45%
d. It is associated with mural thrombus
e. Mortality rate may be up to 50% - The following are risk factors for developing PPCM:
a. Advanced maternal age (>35 yrs)
b. Multiple pregnancy
c. Use of tocolytics
d. Primiparity
e. Ethnicity - With regard to management of PPCM:
a. Beta-blockers may improve outcome
b. Management should be multidisciplinary
c. PPCM is unlikely to occur in subsequent pregnancies
d. Anticoagulant therapy is contraindicated
e. Arrhythmias should be avoided
EXAMPLE CASE
A 37 year-old lady presents at 36 weeks gestation in her fourth pregnancy with a 6-day history of worsening shortness of breath, paroxysmal nocturnal dyspnoea, orthopnoea and peripheral oedema. Her three previous pregnancies were uneventful and she delivered vaginally each time.
Her past medical history includes hypothyroidism and morbid obesity (BMI 40). She has no known cardiac history. There is no familial history of note, and no background of drug or significant alcohol consumption.
On examination the patient is tachypnoeic and tachycardic at rest, with normal blood pressure. Oxygen saturations are 92% in room air. Bi-basal course crackles are audible on auscultation. Arterial blood gas analysis reveals a respiratory alkalosis with mild hypoxaemia, electrocardiogram (ECG) demonstrates sinus tachycardia with left bundle branch block, chest X-Ray shows cardiomegaly with fluid in the horizontal fissure and prominent upper-lobe vessels.
Transthoracic echocardiography reveals severe left ventricular dysfunction, with an ejection fraction of 15-17%, dilated cardiomyopathy (left ventricular end diastolic diameter 81mm), severe mitral and moderate tricuspid regurgitation with moderately elevated pulmonary artery pressure.
INTRODUCTION
Peripartum cardiomyopathy (PPCM) is a rare condition that affects women in late pregnancy and in the post-partum period. It is characterised by heart failure that presents in the absence of any other identifiable cause. It is associated with significant morbidity and mortality. Early recognition and aggressive management of the condition is crucial to improving outcome.
INCIDENCE
The true incidence of PPCM is unknown. Reported incidence varies between 1 in 3,000 to 1 in 10,000 pregnancies.
AETIOLOGY AND PATHOPHYSIOLOGY
The aetiology of PPCM is unknown. It is postulated that PPCM could be mediated through many mechanisms including: immunological factors, an abnormal hormone response, abnormal inflammatory changes, myocarditis, or a genetic predisposition.
PPCM manifests predominantly as ventricular systolic dysfunction from a global reduction in myocardial contraction and thus left ventricular ejection fraction. Compensation through the enlargement of the left ventricle occurs increasing end diastolic volume and stroke volume. As compensation fails, cardiac output falls and cardiac failure ensues.
RISK FACTORS
Risk factors for PPCM include:
- Maternal age >30 yrs
- Multiparity
- Ethnic group – e.g. African descent
- Obesity
- Multiple pregnancy
- Pregnancy associated hypertensive disorders
- Essential hypertension
- Tocolytic therapy with β-agonists
- Cocaine use
CLINICAL FEATURES AND DIAGNOSIS
Symptoms of shortness of breath on exertion and orthopnoea are common in the third trimester. Diagnosis of PPCM can be delayed if these symptoms are erroneously attributed to the normal physiological changes of pregnancy.
Symptoms of PPCM include: dyspnoea, fatigue, orthopnoea, paroxysmal nocturnal dyspnoea, palpitations, haemoptysis and peripheral oedema. These may present as acute or sub-acute episodes of left ventricular failure.
Tachyarrhythmias can occur in PPCM, including supraventricular tachycardia (SVT), atrial fibrillation (AF) and rarely ventricular tachycardia (VT). There is an increased risk of thromboembolism due to the potential development of mural thrombus.
PPCM is a diagnosis of exclusion. Parturients with recognised congenital or acquired heart disease can present with similar symptoms in late pregnancy or during labour. Similarly, pregnant women with undiagnosed ischaemic or structural heart disease have decreased functional reserve and are less able to tolerate the haemodynamic physiological changes of normal pregnancy.
Diagnosis of PPCM requires four criteria to be met:1
- Heart failure developing towards the end of pregnancy or up to five months post-partum
- Absence of another identifiable cause of cardiac failure
- Absence of cardiac symptoms or disease prior to late pregnancy
- Left ventricular dysfunction – defined as an ejection fraction less than 45% or reduced fractional shortening of less than 30%
Cardiomyopathy meeting criteria 2-4 but presenting earlier than the third trimester is referred to as pregnancy-associated cardiomyopathy (PACM). Underlying pathophysiology is probably similar to PPCM.2
DIFFERENTIAL DIAGNOSES
Differential diagnoses of PPCM includes:
- Pulmonary embolism
- Severe sepsis
- Amniotic fluid embolism
- Preeclampsia / pregnancy induced hypertensive disease
- Arrhythmias
- Severe anaemia
- Myocardial infarction
- Dilated cardiomyopathy of other aetiologies (see Table 1)
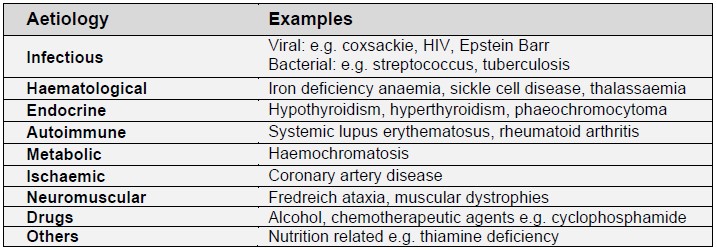
Table 1: Causes of dilated cardiomyopathy
INVESTIGATIONS
Investigations include:
- Electrocardiogram (ECG)
- Transthoracic echocardiogram including Doppler studies
- Cardiac MRI
- Coronary angiography
- Viral studies
- B-Type Natriuretic Peptide (BNP): is a screening marker but lacks specificity
PROGNOSIS
Prognosis is poor and PPCM is one of the leading causes of maternal death.3 A prognostic indicator is the degree of dysfunction at presentation, either defined by the New York Heart Association (NYHA) functional classification or by transthoracic echocardiography.
The mortality rate is between 15 and 50%. 30-50% will improve and recover a left ventricular ejection fraction of 50% or more. Death results from intractable heart failure, arrhythmias and thromboembolism.
MANAGEMENT
Early identification of PPCM enables optimal management.
Multidisciplinary management by senior staff is essential, and should include involvement from an obstetrician, a cardiologist with a special interest in obstetric disorders, and an anaesthetist. Should PPCM present during pregnancy, maternal optimisation is key, which may require expedited delivery. A neonatologist should be involved in discussions regarding timing of delivery and to address the potential impact of maternal disease on neonatal outcome. If critical care admission is likely, early involvement of a critical care specialist is important.
Where women with PPCM should be cared for will depend on the timing and presentation of the condition. Referral to a specialist obstetric care centre with the expertise and resources to manage such patients should be considered in all cases.
Pharmacological therapies
Management of PPCM is similar to that of other causes of cardiac failure and includes maintenance of adequate oxygenation, fluid and salt restriction and ventricular off-loading by vasodilation and diuresis.
Angiotensin converting enzyme inhibitors (ACE inhibitors) have been shown to improve mortality in cardiac failure and are recommended as first-line therapy for the management of PPCM post-partum. Angiotensin receptor blockers (ARBs) are an alternative for those intolerant to the side-effects of ACE inhibitors. ACE inhibitors and ARBs are contraindicated during pregnancy due to the risk of birth defects. Administration of ACE inhibitors, particularly in the second and third trimester is associated with increased fetal loss and fetal renal dysfunction, oligohydramnios and other congenital anomalies. ACE inhibitors and ARBs are not recommended for use in breastfeeding mothers initially due to the risk of neonatal hypotension.4
Post-partum ACE inhibitor therapy is indicated for as long as the left ventricle remains impaired. In the longer-term ACE inhibitor therapy may improve cardiac remodelling.
Hydralazine and nitrate therapy, such as isosorbide dinitrate, can be used safely during pregnancy. These drugs reduce afterload, preload and intracardiac filling pressures.
Beta-blockers have been shown to improve survival and may be protective against tachyarrhythmias.
Digoxin is considered safe in pregnancy and may be used for its positive inotropic effect and for the treatment of atrial fibrillation should it occur. Digoxin plasma levels should be carefully monitored.
Loop diuretics, e.g. furosemide, may reduce pulmonary congestion and peripheral oedema and can be used safely in pregnancy and lactation.
Calcium channel blockers are usually avoided due to their negative inotropic properties. Aldosterone antagonists such as spironolactone are not recommended during pregnancy.
Endomyocardial biopsy can be performed to assess whether intravenous immunoglobulin may be of benefit. There is some evidence in small trials to suggest this targeted therapy and others such as bromocriptine and pentoxifylline, may be of benefit if biopsy shows an inflammatory component.5,6,7 Not all PPCM is associated with a myocarditis inflammatory state and immunoglobulin therapy is not recommended empirically.
Due to the risk of venous thromboembolism and mural thrombus associated with PPCM, prophylactic low molecular weight heparin (LMWH) therapy is indicated. If thromboembolic sequelae or mural thrombus have been identified, full anticoagulation is indicated.
Non-pharmacological therapies
Patients in the acute setting may require supportive therapy with non-invasive ventilation or intubation, ventilation and inotropic support.
In severe cases an intra-aortic balloon pump, left ventricular assist device (LVAD) or extracorporeal membrane oxygenation (ECMO) may be required. Heart transplantation may be indicated for patients with severe disease who do not respond to pharmacological treatments.
Implantable defibrillators and cardiac resynchronisation are possible interventions in those women who survive the acute phase but experience on-going significant functional impairment.
Pre-conceptual counselling
Pre-conceptual counselling should be offered to all women with a history of PPCM. There is a significant risk of reacquiring PPCM in subsequent pregnancies. Women with on-going cardiac impairment following PPCM i.e. NYHA class 3 or 4 cardiac failure, or those with a left ventricular ejection fraction of less than 30% are at highest risk from further pregnancies. Woman most at risk should be provided with information regarding the risks of future pregnancy and guidance on contraception, and potential need to consider termination should pregnancy occur.
Anaesthetic Considerations
PPCM poses many challenges for the anaesthetist. Anaesthetic technique will be influenced by the urgency of delivery and the physiological condition of the parturient. Women with suspected PPCM, or a past history of PPCM should be reviewed by an anaesthetist in a timely manner and an agreed plan made for labour, delivery and post-partum care.
Considerations regarding mode of delivery should include the patient’s past obstetric history, current haemodynamic status and their response to medical management. Vaginal delivery is an option for patients with compensated PPCM. Techniques to limit the increase in plasma catecholamines, systemic vascular resistance and myocardial workload associated with labour are advised.
Aorto-caval compression should be prevented at all times by avoiding supine positioning without adequate uterine displacement. Regular monitoring with ECG, oxygen saturations and blood pressure measurements should be initiated early with a low threshold for invasive arterial blood pressure monitoring. In order to limit cardiovascular stress, early labour epidural analgesia is recommended. This will not only reduce the sympathetic response caused by painful contractions, but also decrease afterload and provide a means of achieving surgical anaesthesia should operative intervention be required.
The second stage of labour can be managed with instrumental assistance which may reduce myocardial workload and the detrimental cardiovascular effects of prolonged valsalva manoeuvres during pushing.
Both general and regional anaesthesia have been described for caesarean delivery.8 Monitoring should include invasive arterial blood pressure. Cardiac output monitoring will provide additional information that may aid perioperative management.
The anaesthetic management aims are to:
- Maintain myocardial perfusion by avoiding:
- arrhythmias
- episodes of hypotension or tachycardia
- Optimise cardiac output
- maintain preload but prevent fluid overload
- maintain / increase myocardial contractility
- prevent increased afterload
Titrated neuraxial anaesthesia, by incremental top-up of an epidural or a combined epidural and low-dose spinal anaesthetic technique, may achieve these aims. Neuraxial anaesthesia reduces afterload, promoting forward flow and avoids the use of general anaesthetic agents that reduce myocardial contractility. Neuraxial opioids provide effective postoperative analgesia thereby reducing sympathetically mediated post-partum increases in afterload. Significant falls in systemic vascular resistance should be avoided so that coronary perfusion is maintained. If using a neuraxial anaesthetic technique, attention should be given to the timing of anticoagulation administration.
If general anaesthesia is undertaken, induction should be as smooth as possible to minimise both hypotension and hypertension. The pressor response to laryngoscopy and intubation should be attenuated by administration of an appropriate dose of a rapid-acting opioid, e.g. alfentanil or remifentanil. Inotropes, such as dobutamine, calcium sensitisers (e.g. levosimendan) or phosphodiesterase inhibitors (e.g. milrinone) may be necessary to offset the myocardial depression of intravenous and volatile anaesthetic agents. Vasopressors may also be required to counteract vasodilation and maintain coronary perfusion. Preload should be maintained and minimally invasive or invasive cardiac output monitoring may provide useful information to guide these therapies.
Uterotonic drugs should be used cautiously due to their associated side effects:
- Oxytocin can cause a marked decrease in systemic vascular resistance and in higher doses has an antidiuretic effect
- Ergometrine may cause significant increases in afterload
Following delivery the patient should be recovered in a critical care environment with ongoing invasive haemodynamic monitoring, careful fluid management and adequate analgesia.
SUMMARY
- Peripartum Cardiomyopathy (PPCM) is a rare condition with significant mortality
- PPCM has strict diagnostic criteria
- Early identification of PPCM improves outcome
- Management is multidisciplinary
- Preconceptual counselling should be offered to women who have previously suffered from PPCM
ANSWERS
- a. F
b. F
c. F
d. F
e. T- Aetiology is unknown. There could be immunological factors, inflammatory changes, myocarditis, or a genetic predisposition.
PPCM predominantly affects systolic function.
True incidence is unknown.
Morbidity and mortality is high. 30-50% will improve and recover a left ventricular ejection fraction of ≥50%.
Other causes should be excluded prior to diagnosis of PPCM.
- Aetiology is unknown. There could be immunological factors, inflammatory changes, myocarditis, or a genetic predisposition.
- a. F
b. T
c. F
d. T
e. T- Left axis deviation can be present in pregnancy due to displacement of the apex.
A Q wave in lead III is normal variant of the whole population, it is not specific to pregnancy.
- Left axis deviation can be present in pregnancy due to displacement of the apex.
- a. F
b. T
c. F
d. T
e. T- PPCM is defined as the development of heart failure toward the end of pregnancy or within the first five months of the postpartum period. Left ventricular dysfunction in PPCM is defined as an ejection fraction less than 45% or a reduced fractional shortening of less than 30%.
- a. T
b. T
c. T
d. F
e. T- Risks for PPCM also include multiparity and ethnic group e.g. African descent.
- a. T
b. T
c. F
d. F
e. T- Anticoagulation reduces the risk of thromboembolism.
Arrhythmias should be avoided to optimise coronary perfusion, atrial and ventricular contraction, stroke volume and ultimately cardiac output.
- Anticoagulation reduces the risk of thromboembolism.
REFERENCES AND FURTHER READING
- Sliwa K, Hilfiker-Kleiner D, Petrie MC, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of peripartum cardiomyopathy: a position statement from the Heart Failure Association of the European Society of Cardiology Working Group on peripartum cardiomyopathy. Eur J Heart Fail 2010;12:767-78
- Elkayam U, Akhter MW, Singh H, et al. Pregnancy-associated cardiomyopathy: clinical characteristics and a comparison between early and late presentation. Circulation 2005;111:2050-5
- Steer P, Gatzoulis M and Baker P. Heart Disease and Pregnancy. London: RCOG Press 2006
- Drug and safety undate May 2009, vol 2 issue 10:3 http://www.mhra.gov.uk/safetyinformation/drugsafetyupdate/CPN088003
- Mason JW, O’Connell JB, Herskowitz A, et al. A clinical trial of immunosuppressive therapy for myocarditis. The Myocarditis Treatment Trial Investigators. N Engl J Med 1995;333:269-75
- Sliwa K, Blauwet L, Tibazarwa K, et al. Evaluation of bromocriptine in the treatment of acute severe peripartum cardiomyopathy: a proof-of-concept pilot study. Circulation 2010;121:1465–73
- Sliwa K, Skudicky D, Candy G, Bergemann A, Hopley M, Sareli P. The addition of pentoxifylline to conventional therapy improves outcome in patients with peripartum cardiomyopathy. Eur J Heart Fail 2002;4:305–9
- Soni B, Gautam P L, Grewal A, Kaur H. Anaesthetic management of two cases of peripartum cardiomyopathy. J Obstet Anaesth Crit Care 2011;1:41-5



