Pain
QUESTIONS
Answer true or false
- Phantom pain
- Occurs in around 50% of patients after amputation
- Reduces in incidence with time
- Is usually a constant pain
- Is commonly experienced by people with congenitally absent limbs
- Is more common in patients with persistent stump pain
- Recognised treatments include
- Non-steroidal anti-inflammatories
- Paracetamol
- NMDA Antagonists
- Sodium channel blockers
- Tricyclic antidepressants
INTRODUCTION
Sensation experienced in an amputated limb was first described in 1551 by Ambroise Paré, a French military surgeon. Subsequently, in 1871 during the American Civil War, the term ‘phantom limb’ was first recorded by the neurologist Silas Weir Mitchell. Despite increased clinical recognition of phantom pain since that time, the mechanisms underlying the phenomenon remain poorly understood. The pathogenesis of phantom pain is complex, including both peripheral and central neural processes and is still the subject of on-going study. Phantom pain is a form of neuropathic pain and, once established, can be extremely difficult to treat.
DEFINITIONS
Following amputation, patients experience a number of different forms of pain. Nociceptive pain in the amputated stump is a normal and predictable response to the surgical insult. Early post-operative perception of non-painful sensations in the amputated limb are common (phantom sensation) and should be regarded as normal following amputation. However, phantom pain is the perception of pain in the amputated limb.
These different forms of pain must be distinguished by taking a careful history from the patient as treatment varies depending on the predominant nature of the pain described.
- Phantom sensation: Any sensation in the absent body part, except pain.
- Phantom pain: Painful sensations referred to the absent body part.
- Stump pain: Pain localized in the stump.
FEATURES
Phantom sensations
Phantom sensation and phantom pain commonly co-exist. Phantom sensation occurs in most amputees, and is experienced as resembling the pre-amputation limb in shape and size and may include feelings of posture and movement. Patients may describe feelings of warmth, cold, itching, tingling or electric sensations. Phantom sensation usually appears soon after amputation and can last from weeks to years, but is not experienced as being painful.
Some patients also describe the phenomenon of ‘telescoping’. This is where the distal part of the phantom limb is felt to be closer to the stump or within the stump itself. For example, forearm amputees may describe feeling that their amputated hand is attached to their elbow stump. This probably occurs because the cortical magnification of the hand is proportionally over represented on the somatosensory cortex.
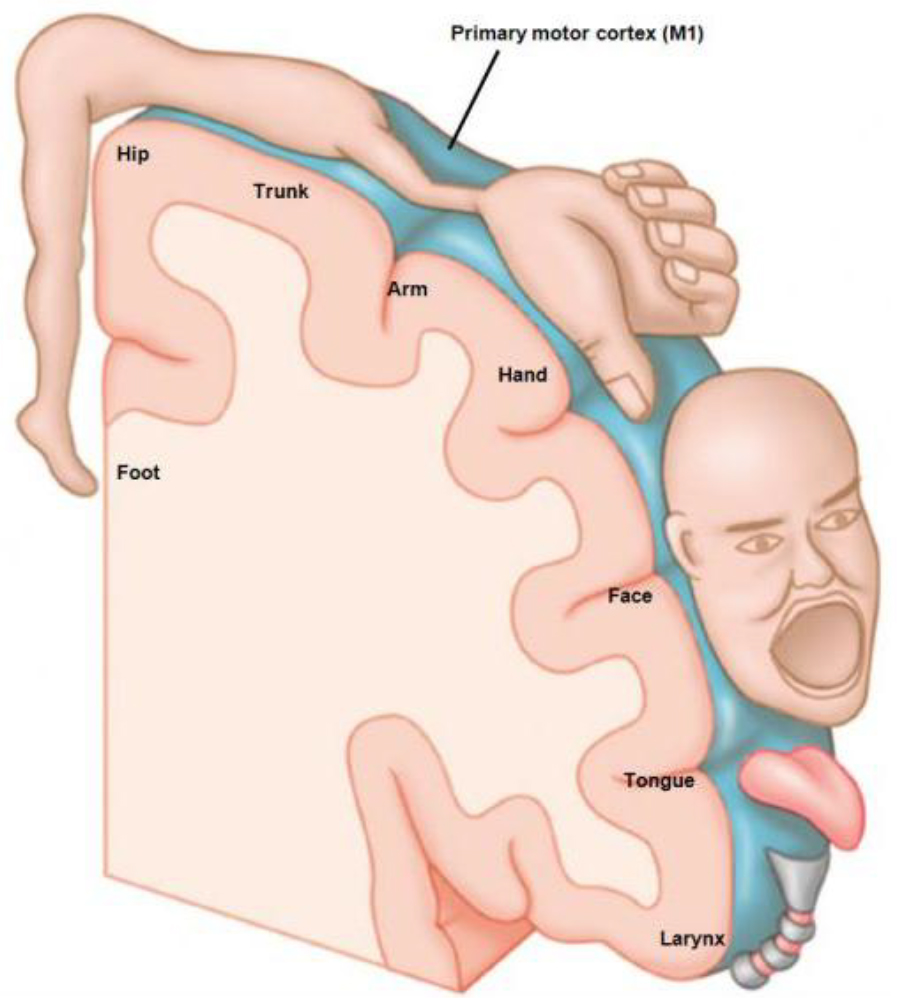
Figure 1. The Homunculus; A pictoral view of the somatosensory map with body parts scaled in sizes proportional to their cortical representation (reproduced with permission from Posit Science Corporation)
Phantom pain
Around 60 – 80% of amputees will experience phantom pain in the early post-operative period with the incidence decreasing with time following amputation. The incidence of phantom limb pain appears to be independent of age, gender and level or side of amputation. 75% of patients will develop phantom pain within the first few days after amputation but the first emergence of phantom pain may be delayed and develop several years later. Phantom pain is often regarded as a chronic pain problem lasting for many years following amputation. Several studies, however, have shown a reduction in pain over periods of 2 – 5 years post amputation, although most continue to experience some pain beyond this.
A number of factors have been shown to be predictive of the onset of phantom limb pain post-operatively. Patients found to be most at risk are those who have severe pain in the amputated limb pre-operatively, patients undergoing bilateral amputation and patients with persisting stump pain. The incidence of phantom limb pain is however lower in paediatric amputees and very rare in those with congenitally absent limbs.
Phantom pain is most commonly thought of as occurring following amputation of a limb but it is also well recognised following amputation of other bodily parts including testis, penis, breast, eye or tongue. The incidence of phantom pain following mastectomy is quoted as high as 15% but is a poorly recognised and seldom acknowledged sequelae of this type of operation.
Phantom pain is usually felt as being located in the distal part of the amputated limb and is often described as being gripping, burning, shooting or cramping in character. Unlike many forms of neuropathic pain, phantom limb pain is commonly intermittent although some patients will experience constant pain. Once established, phantom limb pain can be very resistant to treatment; for instance dense regional anaesthetic blockade provides only limited benefit. Indeed, a number of cases have been reported of patients developing phantom limb pain for the first time while under spinal anaesthetic and also of patients experiencing exacerbations of pre-existing phantom pain with spinal and epidural anaesthesia. This reinforces the view that phantom limb pain is not solely a phenomenon of the peripheral nervous system but involves more widespread and complicated central processes.
Stump pain
Stump pain is common in the early post-operative period. This is an acute nociceptive pain that usually resolves as the wound heals. Stump pain may persist in 5 – 10% of patients due to on-going local pathology or an acute neuropathic process. Sensory examination of the stump at this time may demonstrate hyperalgesia and allodynia. Surgical revision should be avoided if at all possible and is only indicated for localised pathology such as osteomyelitis or abscess. Persistent stump pain may be a risk factor for phantom pain.
At a later stage, once the patient begins rehabilitation and mobilisation, stump pain may develop or be exacerbated due to a poorly fitting prosthesis.
AETIOLOGY OF PHANTOM PAIN
The exact mechanism of phantom limb pain is unknown but it is believed to involve both peripheral and central changes to the nervous system that occur following nerve injury during amputation.
Peripheral mechanisms
Amputation results in the severing of peripheral nerve axons and the formation of neuromas, which are enlarged, disorganised endings of C-fibres and demyelinated A fibres. Neuromas have been shown to demonstrate abnormal spontaneous and evoked activity that is thought to be due to fundamental changes in ion channel function. Altered sodium channel expression has been particularly implicated in this process. There are also changes to dorsal root ganglion cells that display abnormal spontaneous activity and increased sensitivity to mechanical and chemical stimulation. The sympathetic nervous system is also thought to be involved in the pathogenesis and persistence of phantom pain.
Spinal mechanisms
After nerve injury there is an increase in excitability of spinal cord neurons characterised by abnormal spontaneous activity and exaggerated response to mechanical and thermal stimuli. Sensitisation of dorsal horn neurons occurs in response to increased painful stimulus from the amputation site and is mediated by the release of pro-nociceptive agents such as glutamate and neurokinins. This hyperexcitability may result in the clinical picture of mechanical hyperalgesia.
Key to the phenomenon of spinal sensitisation is increased activity in N-methyl D-aspartate (NMDA) receptor mediated systems. These systems are important in the phenomenon of neuronal plasticity whereby the receptive field of a particular neurone is widened by forming new cross-links with adjacent afferent neurones in different laminae within the spinal cord. Other neurotransmitters such as substance P and calcitonin gene-related peptide have also been implicated in this process
Cerebral mechanisms
Soon after amputation there is a reorganisation of primary somatosensory and motor cortices and subcortical structures. Areas of somatosensory cortex which previously corresponded to the missing limb subsequently receive sensory information from other areas of the body that synapse at adjacent areas on the somatosensory cortex. It is thought that phantom pain may be a consequence of errors occurring in this remapping process.
The degree of reorganisation is related to perceived pain intensity and reduces with effective treatment of pain. This can be demonstrated by clinical examination of sensation, which may show a somatotropic representation of the phantom limb on the chest wall, in the stump or on the face.
PREVENTION
Many studies have been undertaken investigating methods of preventing phantom limb pain. A popular theory has been the use of pre-emptive analgesia. The constant, supra-normal nociceptive input from an amputated limb is thought to contribute to the neural re-organisation which may lead to phantom pain phenomena. It was hoped that by initiating intensive pre-operative analgesia, particularly by afferent nerve blockade, abnormal neural reorganisation may be prevented. However, in patients who have experienced a period of severe pain pre-operatively these changes may have already taken place.
Much of the early work in this area examined the impact of prolonged pre-operative epidural analgesia on the incidence of phantom pain. Despite showing promising early results, it was later realised that some studies had significant methodological problems and subsequent larger studies found no significant benefit on the overall incidence of phantom pain with this technique. Pre-operative epidural anaesthesia may however decrease the incidence of severe phantom pain but not the overall incidence of phantom pain.
An alternative, and increasingly popular technique to prevent phantom pain involves the placement of peri-neural catheters either pre or intra-operatively. Administration of local anaesthetic at the time of amputation and as a continuous infusion for 72 hrs post operatively has been shown to be of some benefit in helping prevent phantom pain. The addition of clonidine to a peri-neural local anaesthetic infusion may confer an additional benefit.
TREATMENT
In common with many areas in pain medicine, there is no strong evidence base to guide treatment of phantom pain. Many studies are of small size with differing end-points and investigate diverse patient groups with different peri-operative anaesthetic management. Consequently, much available evidence is based on small case-series, cohort studies and expert opinion making any firm recommendations difficult. However, some common treatment strategies are detailed below.
Pharmacological
Antidepressants
Tricyclic antidepressants (TCA’s) such as amitriptyline and nortriptyline act by inhibiting the re-uptake of noradrenaline and serotonin thereby potentiating the action of two important central anti-nociceptive pathways. TCA’s have been shown in a number of randomised controlled trials to be effective in the management of a variety of neuropathic pain conditions. Although there are no studies specifically examining the use of TCAs in phantom limb pain, they are commonly used and thought to have modest efficacy. TCAs have a number of side-effects including excessive sedation and anticholinergic side effects such as dry mouth, urinary retention and constipation. These may limit their use. However, when started at low dose, these effects can be minimised.
Duloxetine is a relatively new agent which has shown promise as an analgesic in other types of neuropathic pain (particularly in painful diabetic neuropathy). Duloxetine also acts on serotinergic and noradrenergic pathways similar to TCA’s. Clinical experience with this agent in phantom limb pain is limited; however, one early case report suggests that duloxetine may be beneficial in phantom limb pain.
Serotonin specific re-uptake inhibitors (SSRIs) are less effective in the management of neuropathic pain and consequently are seldom used for this purpose. This lack of efficacy is likely to be a reflection on the much narrower spectrum of action compared to TCA’s.
Sodium channel blockers and anticonvulsants
Gabapentin and pregabalin bind to voltage gated calcium channels and have been shown to be effective in a variety of neuropathic pain problems. Evidence for their use in phantom pain is unclear, with conflicting results from RCTs. Although generally well tolerated, the main dose-limiting side effects of gabapentin are somnolence and dizziness which can be minimised by gradual dose titration.
Second line agents include carbamazepine and lamotrigine. These should be considered after conventional mono and combination therapy with TCA’s and anticonvulsants has failed.
Intravenous lidocaine has been extensively reported as having good efficacy in neuropathic pain and its oral analogue, mexiletine, has also been shown in one small study to produce pain relief in phantom pain. This class of drug can be particularly useful as the analgesic effects are of faster onset in comparison to standard anti-neuropathic agents with the clinical effect often significantly outlasting the pharmacological action of the drug. These agents have a good safety profile and should be considered at an early stage after other first-line agents have been tried.
Tramadol
Tramadol is a synthetically produced drug that has both monoaminergic and opioid activity with less adverse effects than strong opioids. This extended action is particularly useful for neuropathic pain that can involve multiple receptor processes. Tolerance and dependence are uncommon. Tramadol has been shown in a number of studies to reduce neuropathic pain including post-amputation pain.
Other drugs
One double-blind crossover study has shown calcitonin to be effective in treating phantom pain if given early in the post-operative period. This is a clinically well tolerated agent that can be administered as a subcutaneous injection, intravenous infusion or intra-nasal spray. It does not, however, have any proven efficacy in treating established phantom pain.
Intravenous ketamine has been shown to reduce pain, hyperalgesia and ‘wind up’ in those with stump pain and phantom limb pain. However, ketamine administered via the epidural route peri-operatively does not decrease the incidence of phantom limb pain. The use of ketamine is frequently limited by unpleasant neuro-psychiatric side-effects such as hallucinations. An alternative NMDA antagonist is Memantine that has been shown in some small studies to be helpful in managing phantom limb pain if used during the acute or sub-acute phase. This drug has a good pharmacokinetic profile, well suited to pain management, with a better side-effect profile than ketamine.
Non medical treatments
Mirror box
The mirror box is a device containing a vertically placed mirror into which the amputated limb is placed and is positioned so that a reflection of the patient’s intact limb is ‘superimposed’ onto the perceived position of the phantom limb. It has been shown that mirrored movements activate the contralateral sensorimotor cortex, and this is associated with a reduction in pain.
The mirror box is particularly helpful in patients who experience spasms of their phantom upper limb perceived as involuntary clenching of the missing hand. The mirror box allows the patient to visualise the unclenching of this limb which can help relieve the pain of these spasms.
Mental imagery
Alternative methods of stimulating the motor or sensory cortices can also achieve a reduction in phantom limb pain. Mental imagery is the process of imagining motor sequences and is thought to work in a similar way to the mirror box. This follows a program of imagery exercises and can be completed by the patient at home. Studies looking at functional MRI (fMRI) in amputees before and after training in mental imagery have shown a significant reduction in pain which is associated with a corresponding change in cortical MRI signal. This is seen only in patients with amputation pain, and not in amputees with no pain or controls.
Acupuncture and TENS
Other treatments which have been used include acupuncture and TENS. It is not clear where the optimum site for placement of TENS electrodes. One approach is to apply stimulation to the contralateral limb. There may also be benefit in placing electrodes on the chest wall or flank in patients who display clear somatotropic representation of the limb on these areas.
Stump care
Stump care and prosthetics also have an important role. Immediate fitting of stump prosthesis can be helpful in the management of phantom pain.
Psychology
Patients may also benefit from explanation and reassurance or therapies such as hypnosis, psychotherapy and cognitive behavioural therapy.
Electrical stimulation
Electrical stimulation of the spinal cord, deep brain structures and motor cortex may relieve pain but its effectiveness may decrease over time. These invasive techniques should be reserved for the most refractory of cases.
CONCLUSION
Phantom limb pain occurs in 60 – 80% of patients following amputation as result of a combination of peripheral, spinal and cortical changes and is often difficult to treat. Current treatments are predominantly based on evidence from small studies and evidence of benefit in different types of neuropathic pain. Further research is required to examine methods of preventing phantom pain developing and to guide treatment choices in the future.
Summary
- Phantom limb sensation, stump pain and phantom limb pain often co-exist
- Phantom limb pain occurs as result of a combination of peripheral, spinal and cortical changes.
- Phantom limb pain is difficult to treat
- Pharmacological therapies include tricyclic antidepressants, sodium channel blockers, anticonvulsants, NMDA antagonists and calcitonin.
- Non-drug treatments such as mirror therapy, acupuncture, TENS, psychology and prosthetics are also important
- Epidural anaesthesia may have a role in preventing phantom limb pain but further studies are required.
ANSWERS TO MCQS
- FTFFT
- FFTTT
REFERENCES and FURTHER READING
- Nikolajsen L, Jensen TS. Phantom limb pain. Br. J. Anaesth. 2001; 87 (1): 107-116.
- Nathanson M. Phantom Limbs as reported by S.Weir Mitchell. Neurology 1988; 38:504-505
- Spiegel DR et al. A presumed case of phantom limb pain treated successfully with duloxetine and pregabalin. Gen Hosp Psychiatry. 2010; 32(2):228
- Dworkin RH et al. Pharmacologic management of neuropathic pain: Evidence-based recommendations. Pain 2007; 132 (3): 237-251
- Jaeger H, Maier C. Calcitonin in phantom limb pain: a double‐blind study. Pain 1992; 48: 21–27
- Diers M et al. Mirrored, imagined and executed movements differentially activate sensorimotor cortex in amputees with and without phantom limb pain. Pain 2010; 149: 296-304
- MacIver D, Lloyd M et al. Phantom limb pain, cortical reorganization and the therapeutic effect of mental imagery. Brain 2008; 131 (8): 2181-2191
- Fisher A, Meller Y. Continuous postoperative regional analgesia by nerve sheath block for amputation surgery—a pilot study. Anesth Analg 1991; 72: 300–3
- Churchill Pocketbook of Pain Second Edition 2004 Stannard C, Booth S
- Jackson MA, Simpson KH. Pain after amputation. BJA: Contin Educ Anaesth Crit Care Pain 2004; 4 (1): 20-23.
- Flor H. Phantom-limb pain: characteristics, causes, and treatment. The Lancet 2002 (1); 3:182-189



