Basic Sciences
Before reading the tutorial try answering the following:
- What is an opioid and how do they work?
- How can opioid analgesics be classified?
- What are the adverse effects of opioids?
- Why is it useful to know the lipid solubility of an opioid?
- How does ionisation of opioids (at plasma pH) affects their onset of action.
- What are opioid antagonists, their uses and adverse effects?
DEFINITIONS
- Opium: A mixture of alkaloids from the poppy plant- Papaver Somniferum.
- Opioid: Any naturally occurring, semi-synthetic or synthetic compounds that bind specifically to opioid receptors (see below) and share the properties of one or more of the naturally occurring endogenous opioids.
- Opiate: Any naturally occurring opioid derived from opium (eg morphine).
- Narcotic: From the Greek meaning “to numb or deaden”. It is often used to denote an opioid but also widely used to describe drugs of addiction and hence includes non-opioid compounds.
MECHANISM OF ACTION
Opioids produce their actions at a cellular level by activating opioid receptors. These receptors are distributed throughout the central nervous system (CNS) with high concentrations in the nuclei of tractus solitarius, peri-aqueductal grey area (PAG), cerebral cortex, thalamus and substantia gelatinosa (SG) of the spinal cord. They have also been found on peripheral afferent nerve terminals and many other organs. The efficacy of centrally applied opioids is well recognised, but when applied peripherally, for example in post-traumatic and inflammatory states, their actions are less reliable.
Opioid receptors are coupled with inhibitory G-proteins and their activation has a number of actions including: closing of voltage sensitive calcium channels; stimulation of potassium efflux leading to hyperpolarization and reduced cyclic adenosine monophosphate production. Overall, the effect is a reduction in neuronal cell excitability that in turn results in reduced transmission of nociceptive impulses.
Pure opioid agonists (morphine, diamorphine, pethidine and fentanyl) bind to opioid receptors avidly and demonstrate high intrinsic activity at the cellular level as described above. Partial opioid agonists (buprenorphine, pentazocine), bind to opioid receptors but produce a sub-maximal effect compared to pure agonists. Opioid antagonists (naloxone, naltrexone), have receptor affinity but no intrinsic activity.
OPIOID RECEPTORS
Since their identification, opioid receptors have had a variety of names. The following is the current nomenclature approved by “International Union of Pharmacology” for identification of the opioid receptors:
- MOP (mu opioid peptide receptor)
- KOP (kappa opioid peptide receptor)
- DOP (delta opioid peptide receptor)
- NOP (nociceptin orphanin FQ peptide receptor)
The sigma receptor is no longer classified as an opioid receptor as it does not meet all the criteria for an opioid receptor. A number of different subtypes of each receptor exist; two MOP, three KOP, and two DOP.
OPIOIDS
Naturally occurring opioid compounds are found in plants (e.g. morphine) and produced in the body (endogenous opioids), where they are widely distributed throughout the central nervous system (CNS). These endogenous compounds are peptides that have variable potency and are preferentially bound by different opioid receptors. They have numerous actions including modulation of pain and control of the cardiovascular system, particularly in shock. Although of interest to pharmacologists, endogenous opioids currently have no clinical role. Synthetic and semi-synthetic opioids are widely used clinically, primarily for their analgesic actions. They exert their effect via the same receptors. Endogenous opioid peptides and commonly used opioid drugs, along with their selectivity for different types of opioid receptors, are shown in table 1.
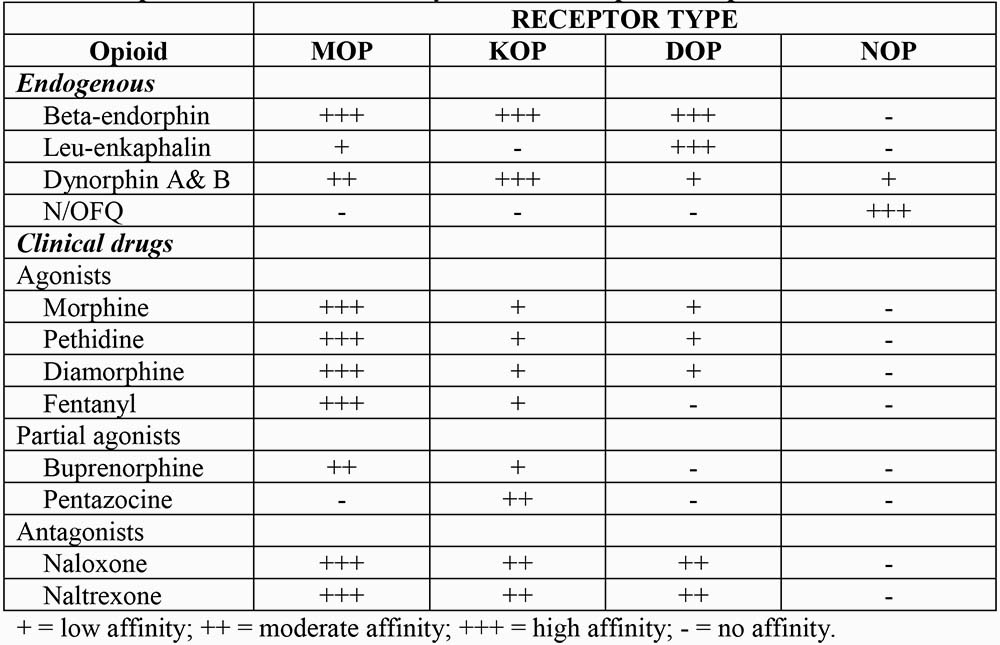
Table 1: Opioids with their selectivity for different opioid receptors
CLASSIFICATION OF OPIOIDS
Several classifications have been proposed (Table 2).
- Traditional: based upon analgesic potency
- Origin of drug: ie naturally occurring or manufactured
- Function: their action at the opioid receptor
In the traditional classification, the “strong” group includes drugs that are pure agonists, whereas intermediate group includes partial agonists.
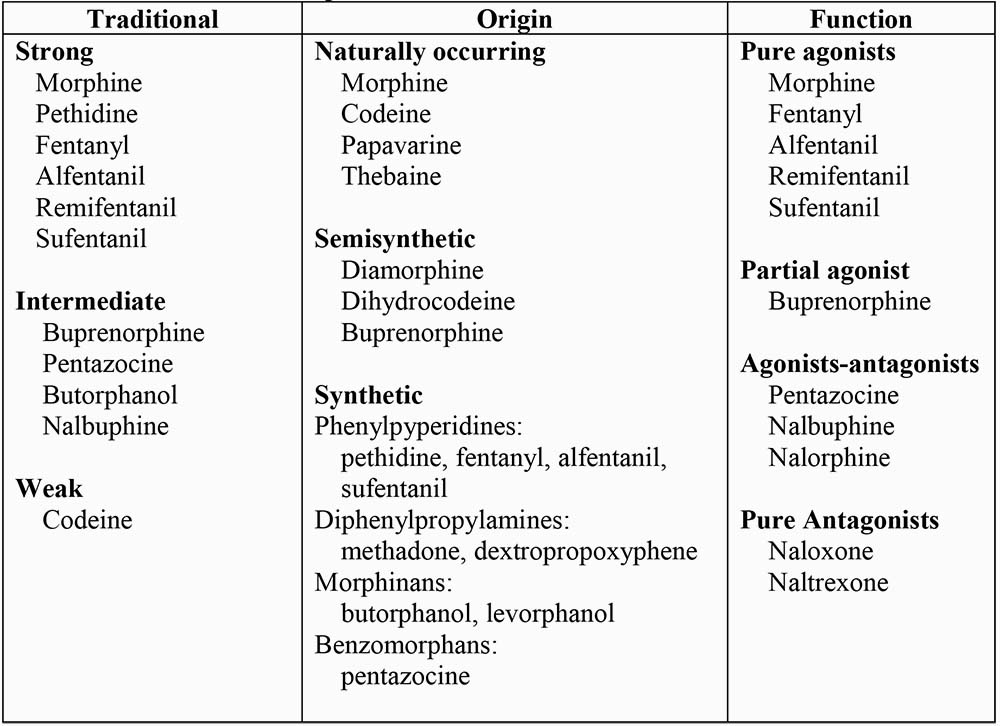
Table 2: Classification of opioids
PHARMACOLOGICAL ACTIONS OF OPIOID AGONISTS
Central Nervous System
- Analgesia: Most effective in relieving dull, continuous and poorly localised pain arising from deeper structures, for example the gut. Less effective against superficial and sharp pain. Neuropathic pain can be very resistant, but patients may report that pain is still present, but the intensity is decreased and it no longer bothers them as much.
- Sedation: Drowsiness, feeling of heaviness and difficulty in concentrating are common. Sleep may occur with relief of pain, although they are not true hypnotics.
- Euphoria and dysphoria: Morphine and other opioids cause a sense of contentment and well-being (euphoria). If there is no pain, morphine may cause restlessness and agitation (dysphoria).
- Hallucination: These are more common with KOP agonists, but morphine and other MOP agonists may also cause hallucinations.
- Tolerance and Dependence: Tolerance is the decrease in effect seen despite maintaining a given concentration of a drug. The mechanism is not fully understood but could involve down regulation of opioid receptors or decreased production of endogenous opioids. Dependence exists when the sudden withdrawn of an opioid, after repeated use over a prolonged period, results in various physical and psychological signs. These include; restlessness, irritability, increased salivation, lacrimation and sweating, muscle cramps, vomiting and diarrhoea.
Cardiovascular System
- Mild bradycardia is common as a result of decreased sympathetic drive and a direct effect on the sino-atrial (SA) node.
- Peripheral vasodilatation caused by histamine release and reduced sympathetic drive may result in a slight fall in blood pressure that may be significant in hypovolaemic patients.
Respiratory System
- Respiratory depression is mediated via MOP receptors at the respiratory centres in the brainstem. Respiratory rate falls more than the tidal volume and the sensitivity of the brain stem to carbon dioxide is reduced. Its response to hypoxia is less affected but if hypoxic stimulus is removed by supplemental oxygen then respiratory depression may be augmented. Concurrent use of other CNS depressants, for example benzodiazepines or halogenated anaesthetic, may cause marked respiratory depression.
- Opioids suppress cough. Codeine suppresses cough to a degree similar to morphine but has lesser analgesic activity. Morphine and diamorphine are used in paroxysmal nocturnal dyspnoea, as they produce sedation, reduce preload and depresses abnormal respiratory drive.
Gastrointestinal System
- (1) Stimulation of the chemoreceptor trigger zone causes nausea and vomiting. Smooth muscle tone is increased but motility is decreased resulting in delayed absorption, increased pressure in the biliary system (spasm of sphincter of Oddi) and constipation.
Endocrine System
- (2) The release of ACTH, prolactin and gonadotrophic hormone is inhibited. Secretion of ADH is increased.
Ocular effects
- (3) MOP and KOP receptors in Edinger-Westphal nucleus of occulomotor nerve are stimulated by opioids resulting in constriction of the pupils (meiosis).
Histamine release and itching
- (4) Some opioids cause histamine release from mast cells resulting in urticaria, itching, bronchospasm and hypotension. Itching occurs most often after intrathecal opioids and is more pronounced on the face, nose and torso. Mechanism is centrally mediated and may be reversed by naloxone.
Muscle rigidity
- (5) Large doses of opioids may occasionally produce generalised muscle rigidity especially of thoracic wall and interfere with ventilation.
Immunity
- (6) The immune system is depressed after long-term opioid abuse.
Effects on Pregnancy and Neonates
- (7) All opioids cross the placenta and if given during labour, can cause neonatal respiratory depression.
- (8) Chronic use by the mother may cause physical dependence in utero and lead to a withdrawal reaction in the neonate at birth that can be life threatening.
- (9) There are no known teratogenic effects.
PHARMACOKINETICS OF OPIOID AGONISTS
There is substantial variability (3-5 fold) in the clinical response to opioids due to their pharmacokinetics and pharmacodynamics. Pharmacokinetic properties of the opioids commonly used in anaesthesia are displayed in table 3.
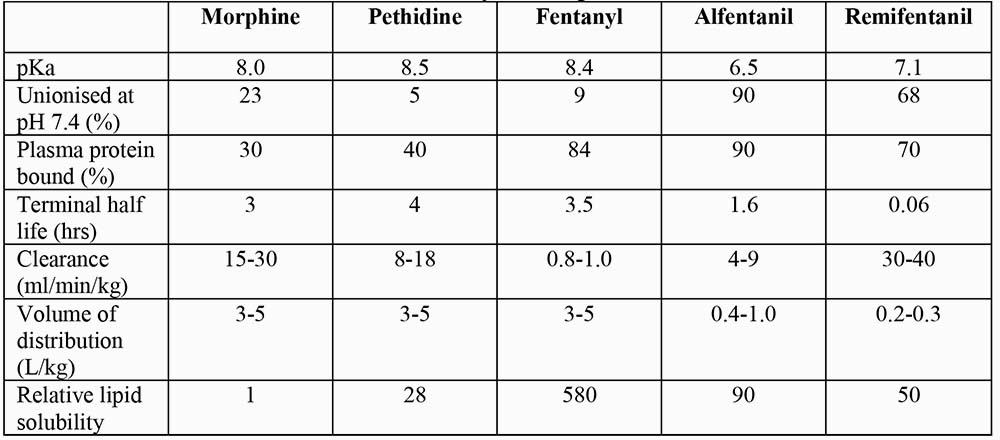
Table 3: Pharmacokinetics of commonly used opioids
Opioids are weak bases (pKa 6.5-8.7). In solution, they dissociate into ionised and unionised fractions, the relative proportions depend upon the pH of the solvent and their pKa. The unionised fraction is more diffusible than ionised form. In the acidic environment of stomach, opioids are highly ionised and therefore poorly absorbed. Conversely, in the alkaline small intestine, they are predominantly unionised and are readily absorbed. However, many opioids then undergo extensive first-pass metabolism in the intestinal wall and liver, resulting in low oral bioavailability. High lipid solubility facilitates opioid transport into the biophase or site of action. Consequently, high lipid solubility confers a more rapid onset of action.
Drugs with high lipid solubility, high unionised fraction or low protein binding in the plasma, demonstrate large volumes of distribution. Most opioids are extensively distributed in the body and their volumes of distribution exceed total body water. Small intravenous dose of short acting opioid (like alfentanil, sufentanil or fentanyl) produce short duration of action because plasma (and brain) concentration remain above the threshold for therapeutic action for only a brief period as the drug rapidly redistributes from the CNS to other tissues. Larger doses produce longer durations of action because plasma concentrations remain above the threshold at the completion of drug redistribution and depend upon the slower elimination process to be reduced below the threshold level.
Opioids are metabolised mainly in the liver to both active and inactive compounds that are excreted in urine and bile. Morphine and other opioids are excreted partly in the bile as water-soluble glucuronides. In the gut, these glucuronides are metabolised by the normal gut flora to the parent opioid compound and reabsorbed (entero-hepatic recirculation). Highly lipid soluble opioids, for example fentanyl, may diffuse from the circulation into the stomach mucosa and lumen, where they are ionised and concentrated because of the low pH. Later, gastric emptying and reabsorption from the small intestine may produce secondary peak effect (gastro-enteric recirculation). Extra-hepatic metabolism is important for some opioids; the kidneys play a vital role in conjugating morphine, whereas blood and tissue esterases are responsible for remifentanil metabolism.
Opioids differ substantially in their durations of action. Explanations for these differences are complex and not always evident from their clearance and terminal half-lives. For example, an analgesic dose of morphine lasts longer than a dose of fentanyl producing an equivalent degree of analgesia; yet the half-life for morphine is shorter than fentanyl. In the case of morphine, its relatively long duration of action is a reflection of its relatively low lipid solubility and slow diffusion out of CNS tissue. Once it enters blood it is effectively cleared from plasma.



