Covid-19
KEY POINTS
- COVID-19 is a potentially life-threatening infection caused by the SARS-CoV-2 virus.
- The clinical features of COVID-19 are generally non-specific and may include cough, fever and shortness of breath.
- Patients with proven or suspected infection must be isolated and treated by staff in appropriate personal protective equipment.
- Some estimates suggest that 15% of patients with COVID-19 who require hospitalization will become critically unwell with respiratory and other organ failures.
- Pulmonary microvascular thrombosis and pulmonary embolus may be prominent feature of severe disease.
- Patients requiring intensive care admission due to severe COVID-19 are at high risk of prolonged mechanical ventilation, failed extubation and acute kidney injury, 20-25% require renal replacement therapy
INTRODUCTION
SARS-CoV-2 is the virus responsible for the coronavirus disease 2019 (COVID-19) pandemic. Transmission is predominantly via inhalation of droplets containing the live virus. COVID-19 represents an unprecedented global healthcare challenge. As of 1st June 2020 there have been 6,189,560 confirmed cases worldwide and it is estimated that 372,469 patients have died as a result of the infection.1 However due to current limited global capability to perform wide-spread testing, these figures may represent significant underestimates of the true impact of the disease. In some case series up to 12% of hospital cases require additional support beyond that which can be provided for in a normal ward environment.2
The aim of this article is to provide practical advice on the management of critically ill patients with this disease. We have used an organ systems approach and in particular have highlighted those areas where management of these patients differs from the management of other critically ill patients.
BACKGROUND
Since the initial description of a new respiratory viral infection in Wuhan, China in late 2019, COVID-19 has become a global pandemic. In the vast majority of affected individuals the infection causes mild flu-like symptoms, and whilst asymptomatic infection has also been described, some of those infected will develop severe, life-threatening disease.
The key feature of severe COVID-19 is hypoxic respiratory failure. Patients with severe disease may also develop progressive multiple organ failure. The current mainstay of treatment is supportive care and currently there are no specific treatments for patients with COVID-19 that have been demonstrated to improve outcomes. This article will discuss the principal elements of supportive care for these patients.
CLINICAL MANAGEMENT
Airway
Summary of recommendations for emergency intubation of adult COVID-19 patients
The predominant route for transmission of SARS-CoV-2 is via inhalation of droplets containing the live virus. The highest viral loads are found in the respiratory tract and these correlate with severity of illness.3,4In some case series, 2.3% of hospitalised COVID-19 patients and up to 71.1% of patients admitted to ICU required invasive mechanical ventilation (IMV).5,6Intubation represents a high-risk procedure for both patients and staff. As all aerosol-generating procedures (AGP), including tracheal intubation, risk spreading the infection to health care workers (HCW), protection of staff is an important aspect of airway management.
The airway team should be kept to the minimum number required. All team members should wear ‘aerosol generating’ personal protective equipment (AGPPE), having previously been trained how to put on and taking off PPE (donning and doffing) (7). Suitable AGPPE includes gloves, fluid resistant long-sleeved gown, suitable face mask (N95 or FFP3 equivalent) and eye protection, such as a visor or goggles. If gowns are not fluid resistant, a fluid resistant apron should be worn over the top. AGPPE should be worn whenever an aerosol generating procedure is performed and when providing direct care to intubated and ventilated patients, due to the risk of accidental breathing circuit disconnection.
The risk of contamination to staff and the requirement for staff to wear full PPE during intubation undoubtedly complicates performing this procedure. However the same principles apply as for any other emergency intubation. Many clinical guidelines and checklists for emergency tracheal intubation of COVID-19 patients have been produced and the use of a checklist is advised. A checklist produced by the Royal College of Anaesthetists of the United Kingdom (UK) has been reproduced below. The team should consist of intubator, intubation assistant, drug administrator and a runner. The runner should ideally remain outside the room in which the procedure is taking place or failing that as far away as possible, yet still able to communicate clearly with the rest of the team.8 When available, a COVID-19 specific intubation trolley and a negative pressure isolation room should be used in order to decrease cross-contamination risk. The individual with the greatest intubation experience should manage the airway to ensure highest first pass intubation success and use of videolayngoscopy is advised. Figure 1 illustrates an example cognitive checklist for emergency intubation.
The sequence suggested in the checklist below may need to be adapted, based on availability of equipment and usual practice. Particular points to highlight are:
- Pay special attention to procedure planning and assigning of roles; it is hard to communicate once in PPE.
- Preoxygenate with a Water’s (Mapleson C) or anaesthetic circuit for 3-5 minutes and use a 2 handed technique to ensure tight fit of mask (Figure 2).
- Perform a rapid sequence induction. Ketamine 1-2 mg/kg is a suitable induction agent. If required, it may be co-administered with an opioid such as fentanyl 1 mcg/kg. Rocuronium 1mg/kg or suxamethonium 1.5 mg/kg are suitable muscle relaxants.
- Avoid hand mask ventilation if safe for the patient, but not if severe hypoxia is present. If needed, use a 2 handed, 2-person technique (Figure 2) with low flow and low pressure.
- Inflate the cuff prior to ventilation.
- To avoid accidental circuit disconnection, push and twist all connections.
- Always place a heat and moisture exchange filter (HMEF), which incorporates a microbiological filter, between the catheter mount and circuit, unless using a wet circuit with humidification, when it should be placed at the end of expiratory limb.
- After stabilization, wet circuits should be used for COVID-19 patients receiving IMV. HMEFs should be checked at least 12 hourly for fluid collection.
- Use in-line suction to minimise circuit disconnections. If disconnection necessary consider clamping endotracheal tube. This should be done after an inspiratory pause in patients with profound hypoxia but beware damaging the tube; this has caused airway obstruction.
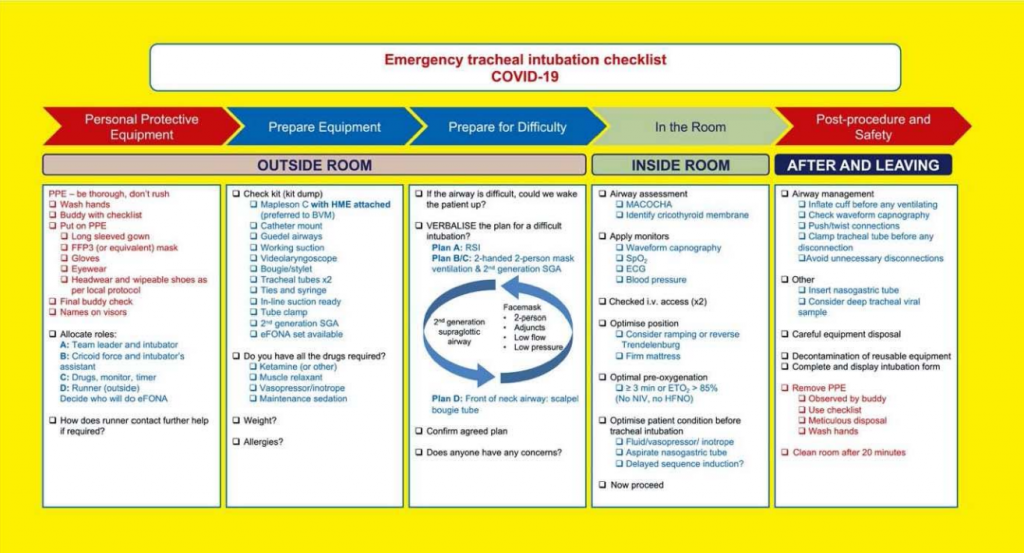
Fig 1. Cognitive aid for the emergency intubation of COVID-19 patients. eFONA – emergency front of neck access, BVM – bag valve mask, RSI -rapid sequence intubation, SGA – supra-glottic airway, MACOCHA score – Mallampati 3-4 (5 points), obstructive sleep apnoea (2 points), reduced cervical spine mobility (1 point), limited mouth opening ,3cm (1 point), coma (1point), hypoxia ,80% (1point), non-anaesthetist (1 point). Reproduced from (8).
- Use standard algorithms for failed intubations, including a second-generation supraglottic device for airway rescue if required.
- Initially cover single use equipment and dispose of it once the patient is in area where rest of care will take place. Disinfect reusable equipment according to manufacturer’s instructions.
- Take off PPE when leaving patient’s room. Pay careful attention to not contaminate yourself or others. There are many video resources providing guidance on how to don and doff PPE including that from the Center for Disease Control and Prevention.9
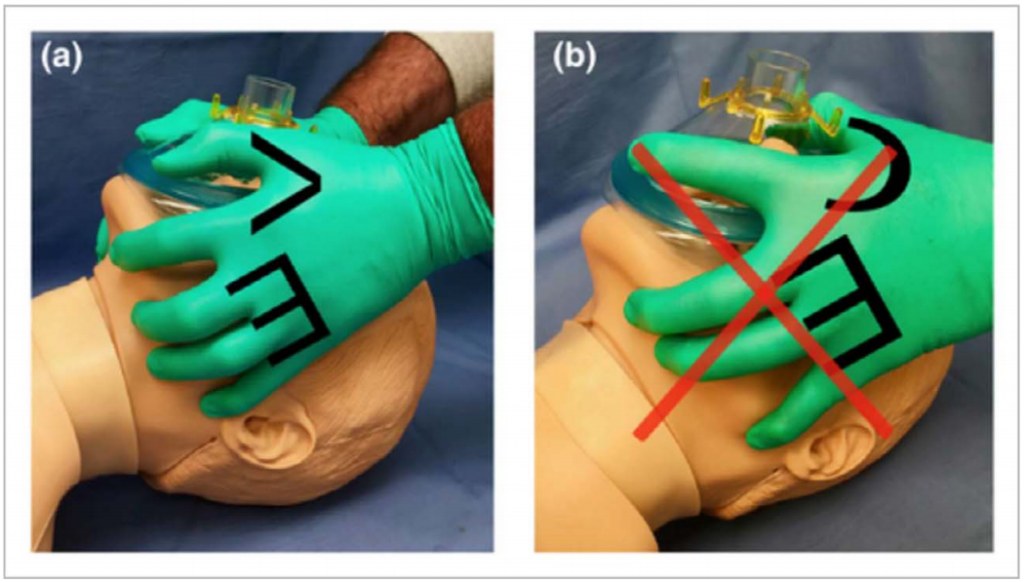
Fig 2: (a) two-person, two-handed technique with the VE hand position (b) The C hand position which should be avoided. Reproduced from (8), original image reproduced with permission from Dr A Matioc.
BREATHING
Respiratory
Respiratory failure is a hallmark of severe COVID-19 infection. The majority of hospitalised patients require oxygen. Globally there is widespread variation in the availability and use of respiratory support for those who develop severe respiratory failure from COVID-19. Of those admitted to ICU in the UK, 60.7% received non-invasive forms of respiratory support, such as high flow nasal oxygen (HFNO) and non-invasive ventilation (NIV), and 71.1% received IMV.6 Respiratory support for COVID-19 is an evolving area and advice on whether to use techniques such as high flow nasal oxygen (HFNO) and non-invasive ventilation (NIV) in particular has changed. Early in the pandemic it was suggested that both HFNO and NIV were contraindicated, because of the risk of aerosolization of SARS-CoV-2 and spread of infection associated with these techniques. More recently there has been increasing use of both techniques in many parts of the world. despite concerns over the high oxygen consumption of the high flow devices. This increasing use of HFNO and NIV has been driven in part by the difficulties associated with mechanical ventilation in this patient group, expanded upon later and also by reevaluation of the risk of HCW infection associated with HFNO in particular. There has also been an increasing use of prone positioning for both mechanically ventilated patients and those receiving noninvasive respiratory support. Prone ventilation, HFNO, NIV and IMV will all be discussed in turn.
Prone positioning
Prone positioning, in comparison to conventional supine positioning, has been demonstrated to improve oxygenation and decrease mortality in mechanically ventilated patients with moderate to severe ARDS.10 Recently, there has been increased use of prone positioning in COVID-19 patients receiving basic respiratory support. Importantly, prone positioning does not require any specialist or additional equipment and many non-sedated patients on face mask oxygen, HFNO or NIV may even be able to turn themselves prone. It is advised that suitable patients spend as much time as possible in the prone position. Mechanically ventilated patients who respond to prone positioning, as demonstrated by an improvement in oxygenation, should remain prone for up to 12-16 hours at a time. This should then be repeated on a daily basis until oxygenation improves. Awake patients may not tolerate spending this long prone. So as to minimize time spent in the supine position, patients on basic respiratory support can be advised to alternate supine positioning with prone and left and right lateral positioning.11
Suggested indications for awake proning:
- Confirmed or suspected COVID-19 and
- Requiring fraction inspired oxygen (FiO2) ≥ 0.28 to achieve SpO2 94-95% (or 88-92% if risk of hypercapnia)
Suggested indications for prone positioning for mechanically ventilated patients:
- Confirmed or suspected COVID-19 and
- Pa02: FiO2 ratio (P:F ratio) of <150 mmHg
Absolute contraindications for proning:
- Need for immediate intubation
- Significant haemodynamic compromise
- Agitation/altered mental state
- Chest injury/unstable spinal injury
Relative contraindications for proning:
- Facial injuries
- Seizures
- Morbid obesity
- 2nd or 3rd trimester pregnancy
- Anterior pressure sores
- Recent abdominal surgery
High flow nasal oxygen (HFNO)
HFNO has been demonstrated to reduce 90-day mortality in non-COVID-19 acute hypoxic respiratory failure including Acute Respiratory Distress Syndrome (ARDS) when compared face mask and NIV (12). Despite prior recommendations advising against the use of HFNO in patients with COVID-19, there has been a widespread change in opinion. HFNO is currently recommended as first-line non-invasive respiratory therapy by the National Institute of Health, European Society of Intensive Care Medicine and, for some patient groups, the World Health Organization.13–15
HFNO should be considered for patients who cannot, or are predicted not to, maintain adequate oxygen saturations whilst receiving maximum oxygen via a reservoir facemask or ‘non-re-breathing’ mask. However, a trial of HFNO will not be appropriate for all patients and careful triage is required. Palliation may be suitable in some patients. In addition, patients who are very hypoxic or who have a very high work of breathing, have a higher chance of failing a trial of HFNO therapy.16 These patients may require immediate mechanical ventilation instead. As for patients who are planned to undergo a trial of CPAP, the following considerations should be taken into account:
- Clarify ceiling of care and inform intensive care of patient if appropriate
- Comply with locally agreed infection control measures. Globally there is significant variation in this. Accepted practice ranges from the use of negative pressure rooms, standard side rooms, cohorting of infected patients and nursing suspected or confirmed cases in standard wards, as long as they are wearing a surgical mask over the top of their high flow cannulae.
- Staff must wear appropriate AGPPE at all times.
- Rapid escalation of care, including provision of RSI and transfer to intensive care must be available
- Assess response within 30-60 minutes of starting HFNO
- Unless there is a risk of hypercarbic type 2 respiratory failure, arterial line and/or arterial blood gas sampling may not be required, as long as SpO2 is continually monitored.
- Suggested initial settings:
- Flow 40 l/min
- FiO2 titrated to severity of hypoxia (suggest at least 0.6)
- Aim SpO2 94-96% unless risk of type 2 respiratory failure (if so consider IMV or BIPAP)
- Consider proning and check SpO2 after every change of position. Patients who remain supine, should be sat up 30-60 degrees
- Lack of improvement or deterioration should prompt consideration of immediate intensive care review
Non-invasive ventilatory support
Continuous positive airway pressure (CPAP)
Although in some countries CPAP has been recommended as a first line non-invasive support for hypoxic COVID-19 patients this remains controversial.17 CPAP does not replace the need for IMV, but early use may delay or prevent the need for IMV. Key points to consider for the use of CPAP in COVID-19 are very similar to those listed for HFNO. Suggested initial settings are CPAP 10 cm H2O and an FiO2 titrated to achieve target SpO2 of 94-96%, except in the presence of acute or acute on chronic type 2 respiratory failure, in which case target SpO2 between 88-92% is appropriate.
Bi-level positive airway pressure (BIPAP)
Non-invasive ventilation is usually not required in COVID-19 patients with previously normal lungs as compliance is usually preserved in the initial phase of disease. However, in patients who are awaiting intubation for high work of breathing, BIPAP can be considered as a bridging therapy. Otherwise, BIPAP should be reserved for those presenting with acute-on-chronic ventilatory failure with documented respiratory acidosis.17
Invasive mechanical ventilation
Phenotypes
It has been postulated that there are 2 distinct phenotypes in COVID-19 patients.18 This hypothesis, which remains controversial, may be useful in understanding why different patients with respiratory failure due to COVID-19 may respond to
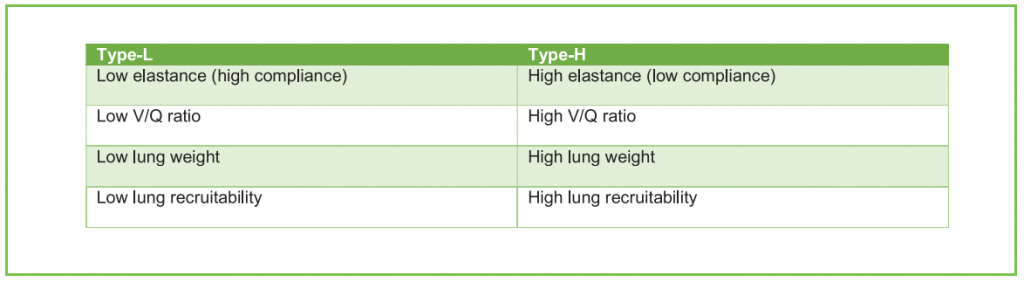
Table 1: Phenotype defining features adapted from (18)
different treatments, as do some individuals at different times in the course of their disease.18 The two phenotypes have been designated as ‘Type-L’ and ‘Type-H.’
It has been suggested that during the initial stages of COVID-19 disease, patients present with Type-L phenotype. They may then transition into Type-H as the disease progresses (18). It has been suggested that transition from Type-L to Type-H may be caused by the COVID-19 pneumonia itself or by patient self-inflicted lung injury (P-SILI). The latter is thought to be caused by high tidal volume (Vt) spontaneous breaths. Type-H behaves in a similar way to Adult Respiratory Distress Syndrome (ARDS).
Ventilatory strategies
Initial approaches for the provision of mechanical ventilation for COVID-19 patients reflected usual strategies employed in ARDS. However, it is has been suggested by some that this approach, utilising high levels of positive end expiratory pressure (PEEP), could be harmful during the early phase of illness (Type-L phenotype).19,20As early on, Type-L patients have compliant lungs it has been suggested that higher Vt values of up to 8-9 ml/kg in these patients may be appropriate, primarily to reduce the incidence of hypercapnia and absorption atelectasis associated with a lower Vt, although this remains very controversial and is counter to core ARDS management. This, along with lower PEEP levels (8-10 cm H2O) may reduce ventilator induced lung injury (VILI) and progression towards the Type-H phenotype. Patients who transition into Type-H phenotype behave much more like classical ARDS patients and require lower Vt (5-6 ml/kg) and higher PEEP levels (up to 15 cm H2O).20 Figure 3 contains a summary of clinical recommendations for mild and moderate to severe ARDS. Table 2 lists suggested physiological targets. Both are further expanded upon in the text.
Step 1
Initial ventilator settings 22,23
Step 2
Strategies to optimise oxygenation
- Wean FiO2 to maintain initial target SpO2 around 92-96%.14 Oxygen stauration target may be lowered further in presence of severe oxygenation problems.
- Consider assessing compliance:20
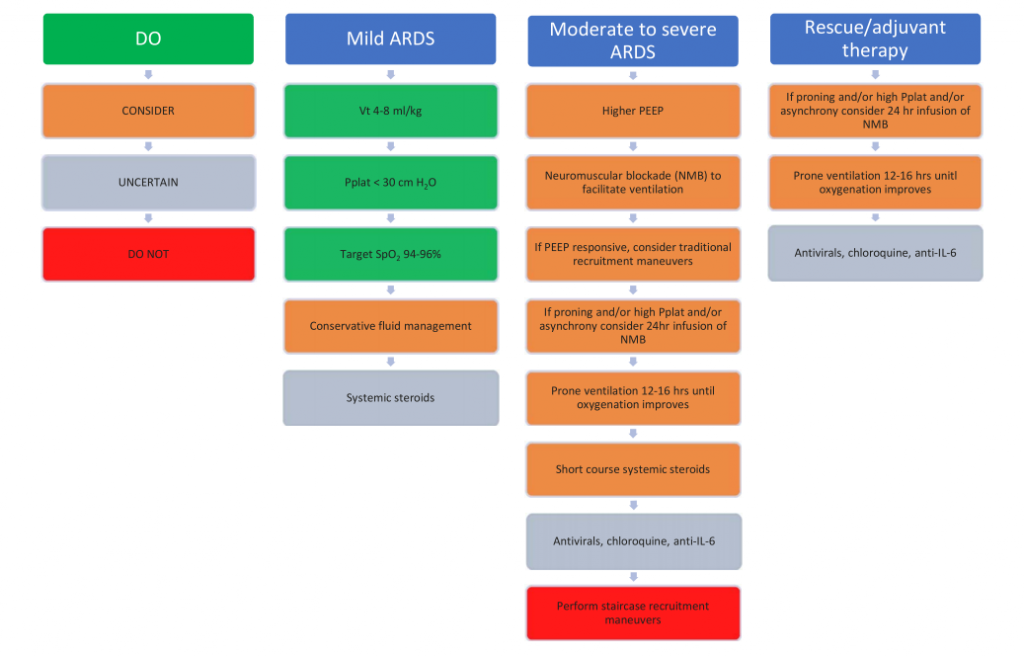
Figure 3. Summary of clinical recommendations for COVID-19 ventilation. Adapted from (13). Pplat – plateau pressure
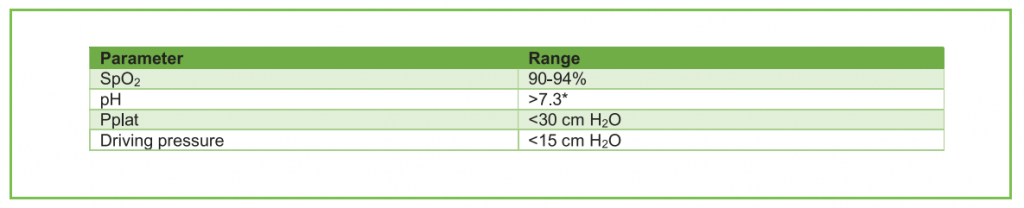
Table 2. Suggested targets when commencing invasive mechanical ventilation. *Permissive hypercapnia may be required to limit ventilator induced lung injury and it is usually well tolerated if the patient is hemodynamically stable down to a pH of 7.1 -7.15 (21)
-
- If >.50 ml/cm H2O consider lower PEEP strategy (< 10 cm H2O) and a Vt of up to 8 ml/kg (Type-L phenotype), although higher Vt remains controversial
- If <50 ml/cm H2O consider higher PEEP strategy (up to 15 cm H2O) and a lower Vt of up to 6 ml/kg (Type-H phenotype)
- If driving pressure > 14 cm H2O reduce Vt until <14 cm H2O (minimum Vt 4ml/kg) 14,24
- P:F ratio <200 mm Hg:
- Consider NMB (bolus preferred)
- Consider higher PEEP strategy (monitor for barotrauma)
- Consider prone ventilation for 12-16 hours
- If proned, asynchronous or Pplat > 30 cm H2O consider NMB infusion for 24 hours
Step 3
Rescue therapies
If P: F ratio does not improve, the following strategies should be considered for both groups :
- Prone positioning, if not done already
- Neuromuscular blockade infusion for 24hrs, especially if prone, dyssynchronous, or high inspiratory pressures. However there is conflicting trial data for the use of neuromuscular blockade; the 2010 ACURASYS trial demonstrated a 90-day mortality benefit with the use of cisatracurium but the more recent ROSE trial did not demonstrate a mortality benefit.25,26
- Referral for extra corporeal membrane oxygenation, if available.
- Adjuvant therapies such as prostacyclin or nitric oxide may be considered, although evidence in non-COVID-19 ARDS suggests there is no mortality benefit with routine use of these agents.14 Either inhaled nitric oxide or prostacyclin (epoprostenol) may improve V/Q matching and both may also offload a failing right ventricle, as may intravenous prostacyclin.
- Consider airway pressure release ventilation (APRV) if familiar with this mode (although there is no proven survival benefit associated with this mode of ventilation) as it may improve oxygenation and reduce number of ventilated days.27
Weaning from mechanical ventilation
This should be performed very cautiously. There is concern that trialing spontaneous breathing too early increases oxygen demand, promotes pulmonary oedema and puts the patient at risk of P-SILI due to excessive negative intrathoracic pressures. Reducing ventilatory support must be done cautiously and spontaneous breathing trials should only be attempted towards the end of the weaning process.20
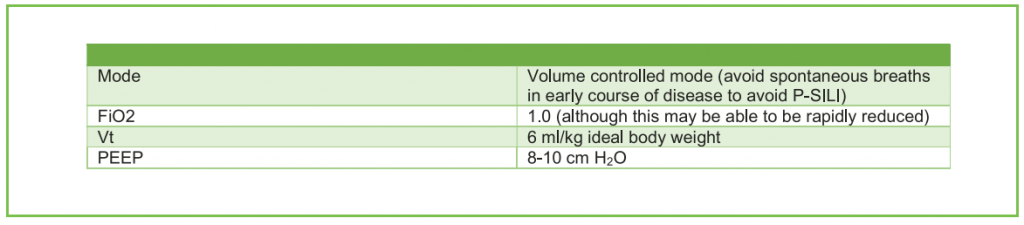
Table 3. Suggested ventilator settings when commencing invasive mechanical ventilation
Extubation
A trial of weaning with a view to extubation should be considered once patients have met the following criteria (or similar) and have a rapid breathing index of less than 100-105 whilst on a spontaneous mode of ventilation:28
- Cooperative cognitive state whilst off sedation
- Adequate cough
- Absence of excessive secretions
- SpO2 ≥ 90% on FiO2 ≤ 0.4
- Pressure support ≤10 cm H2O
- PEEP ≤ 8 cm H2O
- RR < 35/minute
- Vt > 5 ml/kg
- Vital capacity > 10 ml/kg
- Maximal inspiratory pressure of ≤ 20-25 cm H2O
Failed extubation rates in COVID-19 patients of up to 60% have been reported in the first 24-48 hours.24 One contributing factor is an increased incidence of clinically significant airway oedema. In order to reduce the chance of failed extubation, the following measures have been suggested:19
- Routinely check that there is an audible cuff leak when the cuff is temporarily deflated prior to any planned extubation. Do not proceed if no cuff leak. The oropharynx should be suctioned prior to cuff deflation so as to minimise the chance of microaspiration.
- Perform chest physio and tracheal suction as required
- Consider administration of intravenous dexamethasone (4mg every 6 hours) in the 24 hours prior to extubation
- Administer nebulized adrenaline (5 ml of 1:1000) if post-extubation stridor is present.
Following extubation, patients with proven COVID-19 should wear a surgical facemask on top of an oxygen mask or nasal cannulae. Current guidance suggests that unless they have had 2 negative reverse transcription polymerase chain reaction tests for coronavirus, 24 hours apart, they remain an infection risk.15 This is an area of controversy though and many centres are continuing to cohort these patients until discharge from hospital. Tracheostomy may be required, but this needs careful consideration.
CARDIOVASCULAR
Pre-existing cardiovascular disease is common among patients with COVID-19; the incidence of hypertension and ischaemic heart disease in this group is 17.1% and 16.4% respectively.29 COVID-19 patients with underlying cardiovascular disease are more likely to require intensive care, and once admitted to an intensive care unit, they have a higher mortality rate, up to 5- fold in some studies.4 Many patients are profoundly dehydrated on admission to hospital. This places them at increased risk of cardiac and renal complications, especially if an overly conservative fluid strategy is utilised, which does not take into account pre-existing losses. Cardiac failure has been seen in up to a quarter of COVID-19 patients with almost half having no prior history of heart disease. This, combined with potential right ventricular dysfunction seen in ARDS, increases the challenge of providing these patients with appropriate fluid resuscitation.30 In this section we will discuss specific cardiovascular complications reported in COVID-19 and detail appropriate heamodynamic support strategies.
Myocarditis
Like previous coronaviruses such as Middle Eastern Respiratory Syndrome, COVID-19 has an association with myocardial injury and myocarditis. Patients may present with chest pain, dyspnoea, left ventricular failure and arrhythmias.30 Both myocardial infarction and myocarditis will cause abnormally raised troponin levels and an electrocardiogram may not differentiate between these two pathologies. For this reason, echocardiography (ECHO) may be useful; regional wall motion abnormalities (RWMA) are often present in myocardial infarction, whilst patients with myocarditis will usually have global wall motion dysfunction but no RWMA.30 Due to the systemic inflammation and hypercoagulable states seen in COVID-19, patients are also at increased risk of acute coronary syndromes including ST-elevation and non-ST-elevation myocardial infarctions.
Vasoactive agents
Surviving sepsis campaign guidelines for the management of patients with COVID-19 suggest no alteration to the accepted mean arterial pressure (MAP) targets for patients with sepsis (60-65 mm Hg in patients with no previous history of hypertension). When vasoactive support is required, noradrenaline should be used as the 1st line agent. Adrenaline and vasopressin are suitable 2nd line agents if noradrenaline is not available.14 If target MAP is not achieved despite noradrenaline therapy, vasopressin should be considered as an additional agent.
Cardiac Failure and Shock
Patients with evidence of cardiac failure on echocardiography or clinical examination may require dobutamine in addition to noradrenaline when hypoperfusion persists despite appropriate fluid resuscitation.14 Refractory Shock Low dose corticosteroids (e.g. 50mg intravenous hydrocortisone 4 times daily) should be considered for patients with refractory shock.14
Refractory Shock
Low dose corticosteroids (e.g. 50mg intravenous hydrocortisone 4 times daily) should be considered for patients with refractory shock.14
Right Ventricular Dysfunction
Right ventricular (RV) dysfunction, including acute cor pulmonale, is a recognised complication of COVID-19. As seen in classical ARDS, RV dysfunction is commonly associated with an increase in mortality.31 It results in heamodynamic instability due to the combined effects of increased afterload, due to alveolar and pulmonary capillary inflammation and thrombosis, plus hypercapnia, hypoxia and mechanical ventilation. Further fluid administration in this setting may exacerbate RV dysfunction due to fluid overload.32 Monitoring NT-proBNP or cardiac troponin T may also be used to provide an indication of worsening myocardial dysfunction. Table 4 details some of the approaches that may be helpful to reduce right ventricular dysfunction.33 RV dysfunction can be diagnosed clinically as summarized in table 5, but when available, echocardiography is the investigation of choice.
Clearly some of the goals in table 4 may be impossible to achieve, particularly in patients with severe ARDS. In addition to prone positioning, pulmonary vasodilators (e.g. epoprostenol) or inodilators (e.g. levosimendan and milrinone) have been used to reduce afterload on the right ventricle by reducing hypoxic pulmonary vasoconstriction.33
Investigations for right ventricular dysfunction
Thromboprophylaxis and anticoagulation
A higher than expected rate of thrombosis has been observed in critically ill COVID-19 patients.34 Both spontaneous and provoked (e.g. line associated) venous thromboembolic (VTE) disease has been seen. It is therefore appropriate to adopt a lower threshold for imaging these patients in order to rule out VTE disease. Arterial involvement, such as stroke, has also been reported but not as commonly. In addition to large vessel clots, micro-thrombi have been reported in pulmonary microvasculature. These are thought to contribute to poor pulmonary perfusion in the absence of demonstrable pulmonary emboli (PE).34 Consensus International guidelines for all aspects of anticoagulation therapy are still awaited. In practice, both therapeutic low-molecular-weight-heparin (LMWH) and unfractionated heparin (UFH) infusions are being utilised. Dose modification of LMWH is required in patients with creatinine clearance < 30ml.min-1 . Where available anti-factor-Xa levels should be used to monitor effectiveness of LMWH therapy.
Where there is a high index of clinical suspicion that a patient has a PE and has signs of having a hypercoagulable state, such as high inflammatory markers, therapeutic anticoagulation should be considered even if imaging is delayed or unavailable, as long as there are no contraindications.34
DISABILITY
Deep sedation is often needed to facilitate mechanical ventilation for patients with very poor respiratory function. Anecdotally, we have found that many COVID-19 patients require higher doses of sedative medications than would be expected in similar mechanically ventilated patients with respiratory failure from other causes. In order to minimise the
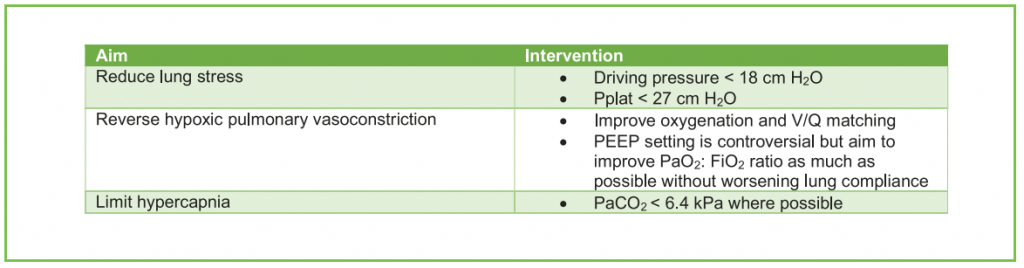
Table 4. Strategy for reducing right ventricular dysfunction
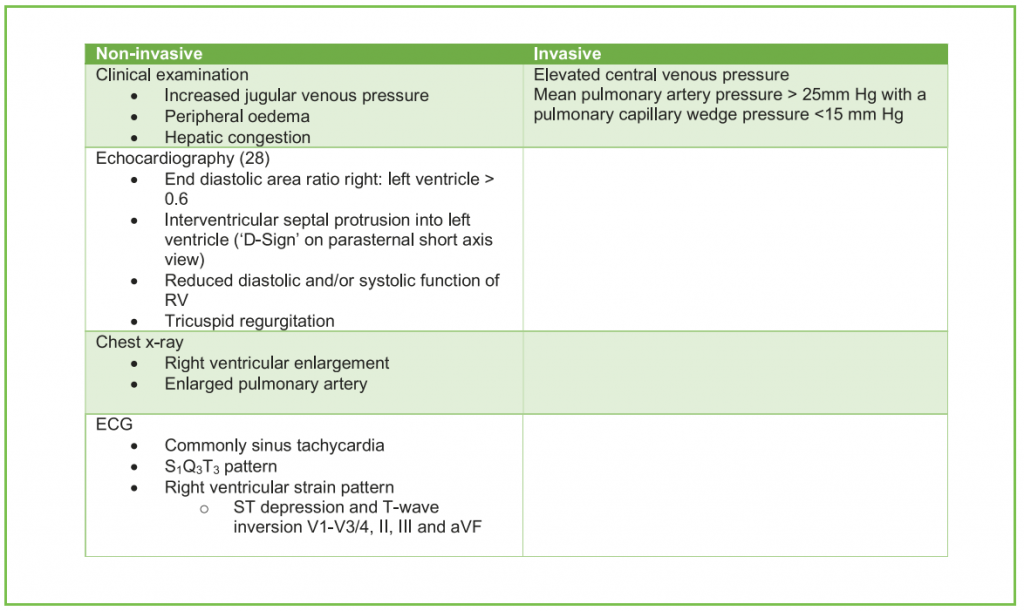
Table 5. Non-invasive and invasive investigations for right ventricular dysfunction
complications associated with deep sedation, which include delirium and prolonged mechanical ventilation, sedation level should be frequently assessed using a recognized method and always be tailored to clinical need. Infusions of an opioid and benzodiazepine or propofol is a common combination for analgesia and sedation of ventilated patients. There is very little evidence of superiority of one regimen over another. Additional agents such as clonidine and chloral hydrate may sometimes be required.
FLUID BALANCE
At presentation to hospital many patients are significantly dehydrated. This may contribute to the higher than expected rate of acute kidney injury, increased thrombotic risk and V/Q mismatching. Balanced crystalloid solution should be used for the acute resuscitation of shocked COVID-19 patients. Once patients have been adequately resuscitated, the aim is to maintain euvolaemia. A conservative fluid strategy aiming for euvolaemia, has previously been demonstrated to improve lung function, reduce duration of invasive ventilation and length of ICU stay without increasing extra-pulmonary organ failure in acute lung injury, when compared to a liberal strategy.35 It is important to recognise that acute severe RV dysfunction may mimic severe hypovolaemia (e.g. tachycardia, low cardiac output, high pulse pressure variation). Fluid responsiveness should be assessed utilizing standard available methods. Administration of diuretics may be suitable to reduce oedema and excessive positive fluid balance in the absence of evidence of cardiovascular instability, poor perfusion and shock.
Renal replacement therapy
Acute kidney injury is common in COVID-19; it has been reported in up to 35% of cases on ICUs in the UK and over 20% of COVID-19 admissions to UK ICUs received renal replacement therapy.6,24 There are many potential causes for this high incidence of renal impairment including too conservative a fluid strategy, renal microthrombi or even direct viral injury to renal tubules. Patients should be managed following established evidence-based guidelines. Hypercoagulability may be responsible for the observed high incidence of filter circuit clotting. To prevent this, it may be necessary to combine regional citrate anticoagulation of the circuit with therapeutic anticoagulation, using either an infusion of unfractionated heparin or low molecular weight heparin (LMWH).19 Many units have instituted monitoring of LMWH therapy using factor Xa assays because of concerns regarding acquired heparin resistance.
As well as changes to anticoagulation protocols, the COVID-19 pandemic has required changes in the delivery of renal replacement therapy itself. Shortages of equipment to deliver renal support as well as shortages of replacement fluids have been reported. For these reasons, some centers have developed protocols for the delivery of intermittent haemodialysis and peritoneal dialysis, as well as utilising higher rates of renal replacement for a shorter period of time, so as to allow sharing of haemofilters between patients.19
IMMUNE SYSTEM
Secondary Haemophagocytic Lymphohistiocytosis (sHLH), Macrocyte Activation Syndrome (MAS) and Catastrophic Antiphospholipid Syndrome (CAPS)
These are uncommon hyperinflammatory syndromes that may be triggered by severe viral pneumonias.36 The syndromes are associated with multiorgan failure, an elevated cytokine profile, and may be associated with thrombosis (e.g CAPS). For patients with thrombocytopenia and thrombosis, heparin induced thrombocytopenia (HIT) should also be considered.
Features include:
- Unremitting fever
- Cytopenias
- Elevated ferritin
- ARDS
All patients with severe COVID-19 are at risk of hyperinflammatory syndromes and accurate diagnosis may allow specific treatment options such as methylprednisolone when appropriate. Specialist advice from Haematology or Rheumatology is advised.
SPECIFIC TREATMENTS AND ONGOING TRIALS
As detailed in this article, the mainstay of treatment for COVID-19 is supportive and in keeping with the management of other patients with viral pneumonia causing respiratory failure. The main principles of care include use of evidence- based guidelines for the management of patients with ARDS.23,37At the time of publication of this article there is no randomized control trial (RCT) evidence in favour of any specific treatment for COVID-19. However, there are 331 trials looking at pharmacological interventions.38
ETHICAL CONSIDERATIONS
A pandemic presents very difficult moral, logistical and medical challenges to the workforce. Decisions should be guided by the following ethical principles:39
- Fairness
- Duty to care
- Duty to preserve resources
- Transparent decision-making
- Constancy
- Proportionality
- Accountability
To protect against moral injury and to provide support for individuals, difficult decisions regarding admission, resource allocation and withdrawal of life sustaining treatment should ideally be made by more than one clinician. Where admission to ICU is being considered, discussions should involve ICU clinicians. On the ICU, important decisions, such as potential withdrawal of life-sustaining treatment, should be made within a multi-disciplinary team. These decisions should also be discussed with the patient and their representatives where possible.
The accountability of individual clinicians for their decision-making remains the same during a pandemic. Decisions should be aligned to local and/or national guidelines and should be documented in a clear and contemporaneous manner. Medical ethicists and hospital ethics committees can help with particularly difficult decisions, especially where there is disagreement between the clinical team and patient or their representatives. Where capacity exists, admission to ICU should proceed as usual. Where resources such as beds are limited, they should be allocated based upon an appropriate triage system. The specific triage system being used should be kept under review so that as knowledge and understanding of the condition grows, those with the greatest capacity to benefit are prioritised and cared for.
Cardiopulmonary resuscitation (CPR)
A discussion addressing goals of care and do not resuscitate (DNR) status should be held as soon as is feasible. During these discussions it should be acknowledged that certain resources such as ICU beds may be in short supply should the health system in question should enter ‘crisis standards of care,’ where some aspects of ordinary care may be not possible due to severe resource restraints.40 Lack of resources and the need to maximize the number of lives saved may lead to CPR not being attempted in specific circumstances. This may be because the patient has been deemed, through a transparent and fair triage process, to be ineligible for ICU care or deteriorating despite a period of optimal intensive care.40 When performing CPR, it is essential that HCW are protected. This requires teams to wear full and adequate AGPPE before attempting resuscitation and ensuring that only clinicians that are experienced in emergency intubation should attempt to secure the airway.40
WORKFORCE AND LOGISTICS
Staffing
Part of protecting patients is to ensure that healthcare workers are protected from harm so that they can continue to provide care. Staff who are either over 70 years old, or have significant co-morbidities which place them at increased risk of harm during this pandemic, should consider withdrawing from patient-facing care. In this case it may be possible to assign these staff roles away from direct patient contact, such as telemedicine and logistic or planning roles.
Increased bed occupancy, coupled with high levels of staff sickness, has resulted in many hospitals developing new models of working with the aim of ensuring that care is provided to all affected patients as safely as possible throughout the pandemic. This has involved redeployment of staff from other areas to acute wards and ICUs. This presents a challenge in terms of skill mix as some hospital staff may have not been involved in direct patient care of these types of patients for some time. Providing training is an immense task. Areas that need particular focus include:
- Safe donning and doffing of personal protective equipment
- Orientation to the ICU or other new environment
- Development of standardized operating procedures for medications, therapies and procedures, such as prone positioning
- Acute management of common medical emergencies
- Interpretation of common bedside investigations
- How to undertake simple administrative tasks such as completing a discharge letter or death certificate
Working in an unfamiliar setting can provoke anxiety amongst staff. Adequate face-to-face training may help address this. Where this is insufficient, advice and sign-posting to support services should be given. Wellbeing as a whole is extremely important to combat workforce fatigue and any long-lasting psychological morbidity. There should be a visible and accessible lead for staff wellbeing, so provision of timely support can be offered.
Supply chains
The COVID-19 pandemic has placed an unprecedented strain on healthcare systems including many hospital supply chains. Particular areas of concern include the provision of ventilators, oxygen, drugs for sedation, renal replacement therapy fluids, sets and machines. Many national bodies have provided advice for hospitals on these issues. Particular points to highlight include:
- Oxygen
- CPAP and HFNO require high oxygen flow rates. Discussions should be held early with medical engineering representatives to assess whether the available supply of oxygen will be sufficient to deal with demand. There are online calculation tools which allow hospitals to quantify the supply and demand of oxygen in order to predict where issues in supply may arise.41 In some hospitals, new vacuum-insulated evaporators have had to be installed in order to meet projected demands. It may also be prudent to supply oxygen where possible via concentrators to ease the burden on the piped supply. One solution, specifically designed for use in low resource environments are anaesthetic machines such as the Diamedica Glostavent® Helix, which avoids the need for piped oxygen supply by having an integral oxygen concentrator allowing delivery of up to 10 l/min.42
- Anaesthetic drugs
- Anaesthetic departments should consider providing regional and neuraxial anaesthesia where possible in order to preserve stock levels of drugs, such as propofol and ketamine, that may be required for induction of anaesthesia or sedation on critical care. Where general anaesthesia is required, alternative induction agents such as thiopentone, should be considered.43 Volatile anaesthetic agents, rather than total intravenous methods should be used to provide maintenance anaesthesia, unless there are specific indications for the latter. There may also be supply issues with Cis/ atracurium and rocuronium. Vecuronium, pancuronium and suxemethonium have been suggested as alternative agents. Alternatives to morphine may also need to be considered. Possible options include alternative opioids (oxycodone, tramadol, pethidine), local anaesthetic blocks and infusions, non-steroidal anti-inflammatory drugs and clonidine.
- For a full list of alternative drugs for anaesthetic practice, please see appendix 1.
- Equipment
- Demand for volumetric pumps, syringe drivers, enteral feed pumps and renal replacement machines will all be high during the pandemic. Strategies such as intermittent haemodialysis, bolus feeding or use of appropriate mixed syringes of drugs may be required.
REHABILITATION
Critically ill patients with COVID-19 who survive are expected to have a high incidence of significant physical and psychological morbidity. Rehabilitation should ideally start on the ICU, with early involvement of therapy staff such as physiotherapy, occupational therapy, psychology and speech and language therapy when required. It is hoped that this will reduce the overall individual and societal burden of this pandemic
REFERENCES
- Johns Hopkins University of Medicine. Coronavirus Resource Centre. 2020. https://coronavirus.jhu.edu/map.html. (Accessed 14/05/2020)
- Grasselli G, Presenti A, Cecconi M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy. JAMA. 2020;323(16):1545-6.
- Wang W, Xu Y, Gao R et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020. Epub. https://jamanetwork.com/journals/jama/fullarticle/2762997. (Accessed 12/04/2020)
- Wu Z MJ. Characteristics of and important lessons from the Coronavirus disease 2019 (COVID-19) outbreak in China. Summary of a report of 72,314 Cases from the Chinese Center for Disease Control and Prevention JAMA. 2020. Epub. https://jamanetwork.com/journals/jama/fullarticle/2762130. (Accessed 12/04/2020)
- Guan W, Ni Z, Hu Y, Liang W, Ou C, He J, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. NEJM. 2020. Epub. https://www.nejm.org/doi/full/10.1056/NEJMoa2002032. (Accessed 11/05/2020)
- Intensive Care National Audit and Research Centre (ICNARC). ICNARC report on COVID-19 in critical care 01/05/2020. 2020. https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports. (Accessed 05/05/2020)
- World Health Organization. Advice on the use of masks in the context of COVID-19. 2020. https://apps.who.int/iris/bitstream/handle/10665/331693/WHO-2019-nCov-IPC_Masks-2020.3-eng.pdf?sequence=1&isAllowed=y. (Accessed 12/04/2020)
- Cook TM, El-Boghdadly K, McGuire B, et al. Consensus guideline for managing the airway in patients with COVID-19. Guidelines from the Difficult Airway Society, the Association of Anaesthetists the Intensive Care Society, the Faculty of Intensive Care Medicine and the Royal College of Anaesthetists. Anaesthesia. 2020. Epub. https://onlinelibrary.wiley.com/doi/full/10.1111/anae.15054. (Accessed 12/04/2020)
- Centers for Disease Control and Prevention. Using personal protective equipment (PPE). Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/using-ppe.html (Accessed 05/05/2020)
- Cochrane Systematic Review. Prone position for acute respiratory failure in adults. 2015. https://doi.org/10.1002/14651858.CD008095.pub2. (Accessed 05/05/2020)
- Bamford P, Bentley A, Dean J, et al. ICS Guidance for Prone Positioning of the Conscious COVID Patient. Intensive Care Society. 2020. Epub. https://emcrit.org/wp-content/uploads/2020/04/2020-04-12-Guidance-for-conscious-proning.pdf. (Accessed 25/04/2020)
- Frat J-P, Thile A, Mercat A et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. NEJM. 2015;372:2185-96.
- National Institutes of Health. COVID-19 Treatment Guidelines. 2020. https://covid19treatmentguidelines.nih.gov/introduction/. (Accessed 27/04/2020)
- Alhazzani W, Møller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). 2020. Intensive Care Med. Epub. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7101866/. (Accessed 14/04/2020)
- World Health Organization. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected: interim guidance. 2020. https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected. (Accessed 25/04/2020)
- Wang K, Zhao W, Li J, et al. The experience of high-flow nasal cannula in hospitalized patients with 2019 novel coronavirus-infected pneumonia in two hospitals of Chongqing, China. Ann Intensive Care. 2020;10:(37)
- NHS Engalnd and NHS Improvement. Guidance for the role and use of non-invasive respiratory support in adult patients with COVID19 (confirmed or suspected) Version 3. 2020. https://www.england.nhs.uk/coronavirus/wp-content/uploads/sites/52/2020/03/specialty-guide-NIV-respiratory-support-and-coronavirus-v3.pdf. (Accessed 12/04/2020)
- Gattinoni L, Chiumello D, Caironi P, et al. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020. Epub. http://link.springer.com/10.1007/s00134-020-06033-2. (Accessed 14/04/2020)
- Rehill, N. COVID-19: a synthesis of clinical experience in UK intensive care settings. 2020. https://renal.org/wp-content/uploads/2020/04/COVID-19_synthesis-of-clinical-experience-in-UK-intensive-care_04.04.2020_FINAL.pdf. (Accessed 12/04/2020)
- Marini J, Gattinoni L. Management of COVID-19 respiratory distress. JAMA. 2020. Epub. https://jamanetwork.com/journals/jama/fullarticle/2765302?guestAccessKey=6961e867-c557-44f2-b747-a046cf53c780&utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jama&utm_content=olf&utm_term=042420. (Accessed 25/04/2020)
- Maharaj R. King’s Critical Care – Evidence summary clinical management of COVID-19. King’s College Hospital NHS Foundation Trust. 2020. https://www.nwpgmd.nhs.uk/sites/default/files/KCC COVID19 Evidence Summary.pdf. (Accessed 15/04/2020)
- National Institutes of Health and National Heart Lung and Blood Institute Acute Respiratory Distress Syndrome clinical network mechanical ventilation protocol summary. 2008. http://www.ardsnet.org/files/ventilator_protocol_2008-07.pdf. (Accessed 15/04/2020)
- The Faculty of Intensive Care Medicine. Guidelines on the management of acute respiratory distress syndrome. 2018. https://www.ficm.ac.uk/sites/default/files/ficm_ics_ards_guideline_-_july_2018.pdf. (Accessed 15/04/2020)
- Amato M, Meade M, Slutsky A, et al. Driving pressure and survival in the acute respiratory distress syndrome. NEJM. 2015;372:747-55.
- Papazian L, Forel J-M, Gacouin A, et al. Neuromuscular Blockers in Early Acute Respiratory Distress Syndrome. NEJM. 2010;363(1107–1116)
- The National Heart Lung and Blood Institute PETAL Clinical Trials Network. Early neuromuscular blockade in the acute respiratory distress syndrome. NEJM. 2019;380(1997–2008)
- Zhou Y, Jin X, Lv Y, Al E. Early application of airway pressure release ventilation may reduce the duration of mechanical ventilation in acute respiratory distress syndrome. Intensive Care Med2. 2017;43(11):1648-59.
- Boles J-M, Bion J, Connors M, et al. Weaning from mechanical ventilation. Eur Respir J. 2007;29:1033-56.
- Li B, Yang J, Zhao F. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. Res Cardiol. 2020. Epub. https://link.springer.com/article/10.1007/s00392-020-01626-9. (Accessed 25/04/2020)
- Long B, Brady W, Koyfman A, et al. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020. Epub. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7165109/. (Accessed 25/04/2020)
- Vieillard-Baron A, Matthay M, Teboul J et al. Experts’ opinion on management of hemodynamics in ARDS patients: focus on the effects of mechanical ventilation. Intensive Care Med. 2016;42(5):739-49.
- Peng Q, Wang X, Zhang L. Using echocardiography to guide the treatment of novel coronavirus pneumonia. Critical Care. 2020. Epub. https://ccforum.biomedcentral.com/articles/10.1186/s13054-020-02856-z. (Accessed 26/04/2020).
- Morelli A, De Castro S, Teboul JL, et al. Effects of levosimendan on systemic and regional hemodynamics in septic myocardial depression. Intensive Care Med. 2005;31:638-44.
- Rehill N. COVID-19, thromboprophylaxis and anticoagulation of ICU patients: shared clinical experience. 2020. https://s31836.pcdn.co/wp-content/uploads/COVID-19_Thromboprophylaxis-and-Anticoagulation-of.pdf. (Accessed 28/04/2020)
- The National Heart Lung and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network. Comparison of Two Fluid-Management Strategies in Acute Lung Injury. NEJM. 2006;354:2564-75.
- Rosa´rio C, Zandman-Goddard G, Meyron-Holtz EG, et al. The Hyperferritinemic Syndrome: macrophage activation syndrome, Still’s disease, septic shock and catastrophic antiphospholipid syndrome. BMC Med. 2013;22(11):185.
- Matthay MA, Aldrich JM, Gotts JE. Treatment for severe acute respiratory distress syndrome from COVID-19. Lancet Respir Med. 2020. Epub. https://www.thelancet.com/action/showPdf?pii=S2213-2600%2820%2930127-2. (Accessed 15/04/2020)
- National Institutes of Health. US National Library of Medicine. 2020. https://clinicaltrials.gov. (Accessed 06/05/2020)
- National Academies of Sciences, Medicine and Engineering. Rapid expert consultation on crisis standards of care for the COVID-19 pandemic. https://www.nap.edu/catalog/25765/rapid-expert-consultation-on-crisis-standards-of-care-for-thecovid-19-pandemic-march-28-2020. (Accessed 14/05/2020)
- Kramer D, Lo B, Dickert N. Perspective: CPR in the Covid-19 Era – An Ethical Framework. NEJM. 2020. https://www.nejm.org/doi/full/10.1056/NEJMp2010758. (Accessed 14/05/2020)
- UCSF Anaesthesia Division of Global Health and Association of Asthesiologists of Uganda. Oxygen supply and demand calculator. https://fervent-carson-c81c94.netlify.app/about. (Accessed 14/05/2020)
- Diamedica. Glostavent Helix. 2015. http://www.diamedica.co.uk/english/product_details.cfm?id=1487. (Accessed 26/04/2020)
- Association of anaesthetists and Royal College of Anaesthetists. COVID-19 potential anaesthetic drug list. 2020. https://icmanaesthesiacovid-19.org/drug-demand-supply-covid19-potential-anaesthetic-drug-list. (Accessed 21/04/2020)
Appendix 1

Appendix 1



