General Topics
KEY POINTS
- The presurgical workup of a patient with epilepsy requires multidisciplinary input.
- The Wada test is primarily used for assessing hemispheric language and memory dominance to determine the feasibility of surgical resection.
- Understanding the principles of the Wada test enables anaesthesiologists to assist in the investigation and to anticipate possible perioperative challenges that the patient may encounter
- There is no standardised protocol for the Wada test.
- The shortage of amobarbital has led to the use of other anaesthetics, such as propofol, etomidate and methohexital.
- With the advent of noninvasive imaging techniques, the need to perform the Wada test has been reduced to selected groups of patients for epilepsy surgery.
INTRODUCTION
Epilepsy is a common chronic neurological disorder characterised by recurrent seizures, and 30% of patients remain refractory to medical treatment.1 Among refractory epilepsy, temporal lobe epilepsy (TLE) is an important subtype, as it is often associated with the most favourable surgical outcome. Surgical interventions, such as temporal lobectomy, serve as valuable treatment options for these patients to improve seizurecontrol and quality of life. Comprehensive presurgical evaluation plays an integral role in the selection of surgical candidates and will generally involve a battery of imaging examinations, neurophysiological evaluations and neuropsychological assessments. The main objectives are to identify structural abnormalities and epileptogenic zones and to localise any functional neural tissues near the seizure foci. The feasibility ofsurgery is then determined, balancing the benefits of seizure control against the potential risks of inflicting major neurological deficits.
One of the diagnostic tests performed is the Wada test, during which an anaesthetic drug is selectively injected into each side of the brain toevaluate its individual language and memory functions. The test is offered to patients with technically resectable foci in the temporal lobe and provides information on potential postoperative functional deficit. Anaesthesiologists are involved in administering the anaesthetic drug andmonitoring the patient during the Wada test.
With a particular focus on the Wada test, the aim of this review is to discuss the anaesthetic contributions to the multidisciplinary evaluation of patients for their potential candidacy for epilepsy surgery. Understanding the basic concepts of the Wada test is essential for anaesthesiologists to assist in the investigation and anticipate possible perioperative challenges the patient may encounter.
EPILEPSY SURGERY AND PRESURGICAL EVALUATION
Therapeutic epilepsy surgery encompasses curative resection and palliative intervention. Curative resection aims at the complete removal of epileptogenic zones responsible for triggering the seizures, without disrupting eloquent areas in the vicinity. Lesionectomy is an example in which focal, well-defined lesions are resected. Temporal lobectomy and selective amygdalohippocampectomy are options for TLE with lesions isolated to the temporal lobe, commonly arising from the hippocampus and amygdala.
Both disconnection surgery and neuromodulation are palliative procedures. Disconnection surgery mainly interrupts the nerve pathways that are thought to be involved in the spread of epileptic activity, whereas neuromodulation involves the use of an electrical device to alter nerve activity.
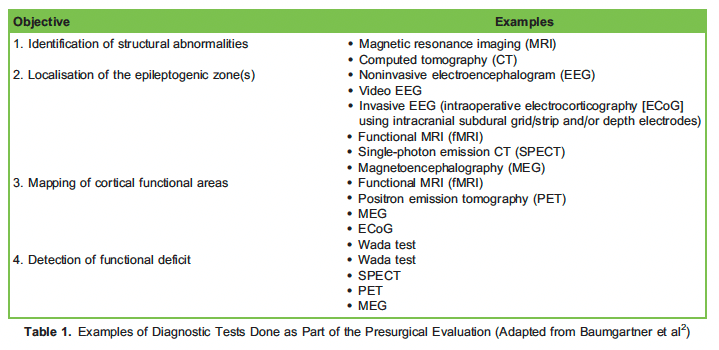
Presurgical evaluation requires multidisciplinary input from neurosurgeons, neurophysiologists, radiologists, anaesthesiolo- gists, psychiatrists and clinical neuropsychologists. A series of diagnostic tests are performed (Table 1), and the information obtained provides insight into the epileptic processes, the cerebral organisation of cognitive skills and the safety of resection. This is relevant in the context of epilepsy surgery,because it is important to preserve the functional regions of the brain during the removal of the epileptogenic cortex, especially those eloquentareas responsible for language and memory. The decision to perform surgery is thus a balance of achieving seizure control, the projected risk of inducing unacceptable cognitive and neurological deficit from surgery.
THE WADA TEST
The Wada test, also known as the intracarotid amobarbital test, is named after Juhn Atsushi Wada, a Japanese neurologist. The test was initiallydeveloped to study the language and memory side effects of electroconvulsive therapy. In the late 1940s, it was incorporated into the preoperative evaluation for cerebral language dominance in epilepsy surgery. The test was later extended to the assessment of hemispheric memory function in the 1960s.3 Nowadays, it is indicated for the assessment of language dominance and memory capacity in patients planned for the resectionof temporal lobe structures.
Technique
During Wada testing, each cerebral hemisphere is transiently inactivated by injecting a small dose of anaesthetic into the respective supplying internal carotid artery. The capacity of each hemisphere to support language and memory function can then be assessed. This information helps to predict postoperative cognitive deficits and inform risk-benefit discussions when surgical resection is an option.
There is no standardised protocol, and the practice varies among centres. The variability ranges from the choice of drug, the dose used, the rate of infusion, the side tested first, the tools for testing memory and language and the methods of scoring. Commonly, the test is started on thepresumed side of surgery and then repeated on the contralateral side.
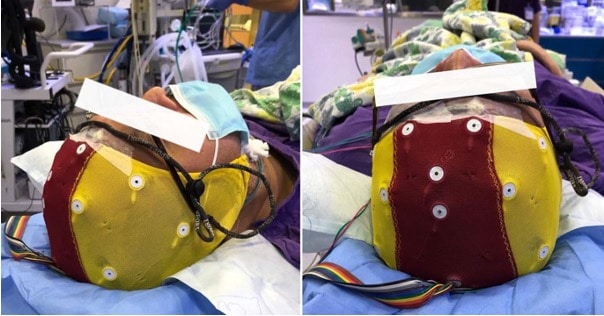
Figure 1: Bilateral electroencephalogram scalp electrodes.
On the day, the patient is brought to the radiology suite with bilateral electroencephalogram (EEG) electrodes attached to the scalp. (Figure 1) Simultaneous EEG recordings is essential to assess the onset and offset of the anaesthetic drug effect and to detect any seizure activity duringthe procedure.
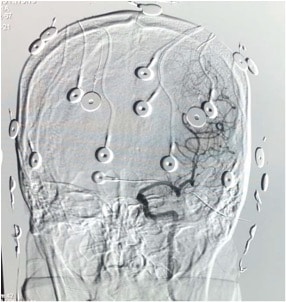
Figure 2. Cerebral angiogram.
Before the testing begins, a neuroradiologist performs a cerebral angiogram. (Figure 2) The femoral artery is punctured after local anaesthesia, and a catheter is advanced into the internal carotid arteries under fluoroscopic guidance. With the catheter positioned just distal to the origin of the internal carotid artery, an angiogram is performed to visualise the cerebral vascular pattern and to exclude any anomalies. A persistenttrigeminal artery should be ruled out due to the concern of inadvertently
anaesthetising the brainstem. Other vascular variations, such as significant interhemispheric cross-flow or alternative supply of the posteriorcerebral artery via the posterior communicating artery, may influence the results.
After the angiogram, the anaesthesiologist administers the anaesthetic drug. Prior to the injection, the patient is asked to raise both arms in the air and spread the fingers while counting out loud. Paralysis of the contralateral arm occurs as the drug is being delivered. A neurologistmonitors the EEG and confirms the efficacy of hemispheric inactivation. Subsequently, a neuropsychologist performs visual, speech andmemory assessments. After completing the first side, the patient should be allowed some time to recover prior to testing the other side. The time taken would depend on the anaesthetic drug used.
Language Testing
To test various components of language, the patient will be presented with tasks, such as naming objects, reading and repeating sentences.Although most healthy, right-handed individuals are left-hemisphere dominant for language, patients with epilepsy have a higher likelihood ofatypical language representation. This is particularly true for those with a left-sided seizure focus, and they may present with bilateral or right-lateralised language dominance. Factors such as the age of seizure onset, the location of seizure focus and handedness may all contribute tocerebral reorganisation and developmental shift in language dominance.4,5
Memory Testing
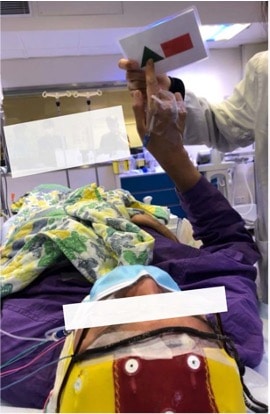
Figure 3. Language testing.
A series of items will be shown to the patient for subsequent recall. (Figure 3) The result of the assessment would reflect the memory capacity of the contralateral, ‘‘lucid’’ hemisphere unaffected by anaesthetics. Once the effect of the drug has dissipated, as reflected by the return of muscle tone and normal EEG, the previously displayed items are presented for recognition. Different centres have different protocolson what is considered a ‘‘pass,’’ and it can be based on the absolute number of correct items or the difference between eachhemisphere’s performance.
Two theories have been described to explain hippocampal function in processing memory: functional reserve versus functional adequacy.6Anaesthetising the epileptogenic hemisphere tests the functional reserve of the contralateral temporal lobe, that is, its capacity to support memory after surgery. Poor performance would predict memory impairment after resection. On the contrary,anaesthetising the contralateral side tests the functional adequacy of the epileptogenic hemisphere, and this usually elicits defective memory performance owing to the functional deficit of the diseased temporal lobe. If the epileptogenic hemisphere is found to support considerable memory function, a more significant memory decline would be expected. In the case of left TLE, reduced functional reserve of the nonepileptogenic hemisphere and high functional adequacy of the epileptogenic hemisphere would suggest a major risk for postoperativememory decline.
Drug Choice
The intracarotid administration of anaesthetic drugs presents unique pharmacokinetic challenges as the intra-arterial dose required is much lessthan the intravenous dose, and this generally does not vary with the patient’s weight.
The standard drug used for the Wada test is sodium amobarbital, which is a barbiturate with low toxicity and short duration of action. However, its worldwide shortage has led to the exploration of various anaesthetics. Ideally, such drug should possess rapid onset and offset of action, lowtoxicity and low epileptogenic potential. Furthermore, it should produce consistent clinical and electrophysiological effects while causing minimal residual disturbance in consciousness to allow multiple testing in a single session.
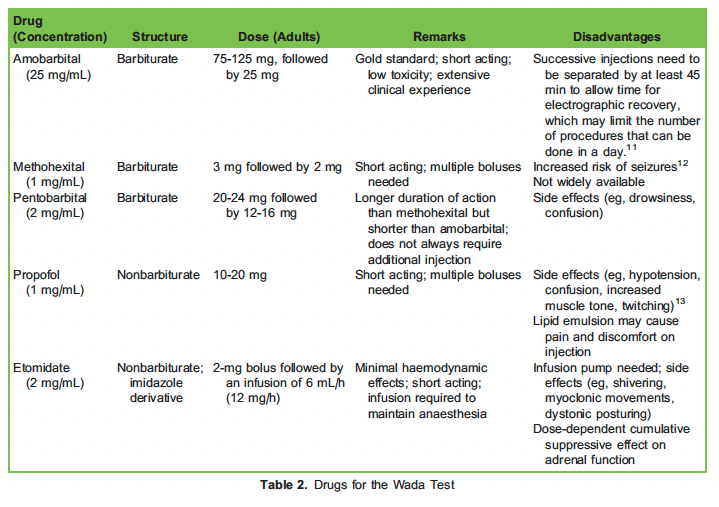
Methohexital, pentobarbital, propofol and etomidate have all been investigated as substitutes (Table 2).7–10
Potential Complications
One case series documented an overall complication rate of 10.9%.14 Neurologic complications include seizure, intracranial vessel injury, carotidartery dissection, arterial vasospasm with or without transient deficits, stroke and transient encephalopathy.14,15 Examples of nonneurologic complications are contrast reactions, contrast-induced nephropathy and haematoma at the puncture sites. Transient symptoms related toanaesthetic injection are also common, ranging from myriad physical symptoms to a change in mental state. Inadvertent reflux of the anaestheticdrug into the basilar artery during posterior cerebral artery injection can cause respiratory arrest and loss of consciousness.
Limitations
There are several limitations of the Wada test. First, it solely examines lateralisation without functional localisation within each hemisphere. Second, the hippocampus is perfused by branches from both the middle and posterior cerebral arteries, and variable vascularisation may affectthe distribution of anaesthetic agents. Third, drug administered via the internal carotid artery causing widespread inactivation of the cortex may lead to aphasia, which may interfere with memory testing. Therefore, a superselective Wada test has been developed, targeting an arterial branch that supplies a specific functional and anatomical region in the brain. For instance, posterior cerebral artery injection inactivates the posterior portion of the hippocampus and adjacent mesial temporal lobe structures, with minimal effects on language.16
ANAESTHETIC MANAGEMENT OF WADA TESTING AND CURRENT ROLE OF WADA TESTING
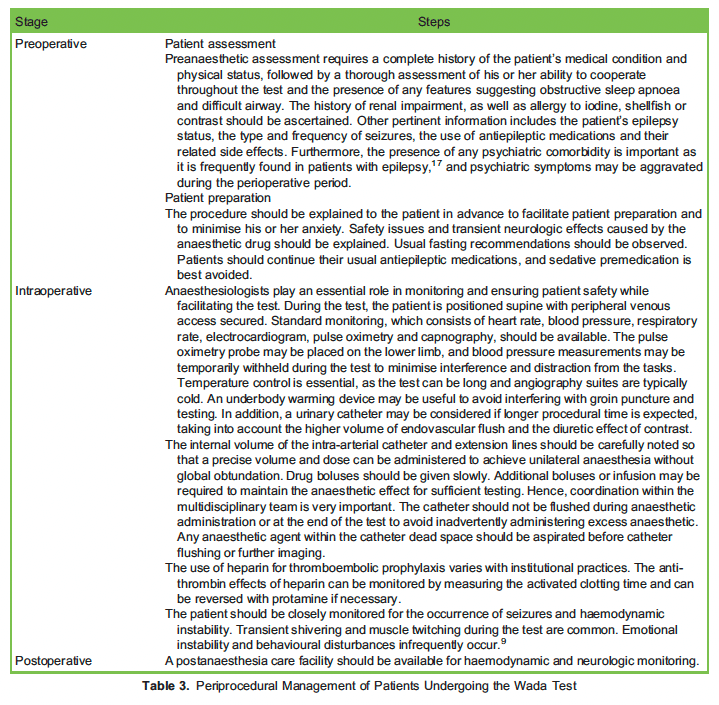
The anaesthetic management of Wada testing is summarised in Table 3. The Wada test’s invasive nature precludes its routine use in theevaluation of epilepsy surgical candidates, especially when the results of other noninvasive tests are considered
adequate for treatment decisions. Functional magnetic resonance imaging (fMRI), being a noninvasive alternative, is increasingly used to assesscerebral language and memory dominance. It has high concordance with the Wada test in subjects with clear left lateralisation and is capable of replacing the Wada test in determining language dominance. However, this method is less sensitive for bilateral or atypical language representation.18–20 On the other hand, there have been attempts to measure the lateralisation of material-specific memory using various fMRIparadigms. Yet, the concordance between fMRI and the Wada test for memory lateralisation has been less consistent.21,22
To date, a highly specific and clinically applicable fMRI protocol that can reliably predict postsurgical outcomes has not yet been established. Therefore, some centres still choose to perform the Wada test in selected groups of patients who are at risk of postoperative global amnesia orwhen their imaging results on language and memory lateralisation are inconclusive or atypical. One must note that fMRI and Wada testing arefundamentally different: the former is an activation study, whereas the latter is a suppressive method that tests the ability of one hemisphere tosustain a function without influence from another.
WADA TESTING IN PAEDIATRIC PATIENTS
Performing the Wada test with children presents a special challenge, as it requires a long period of concentration and cooperation. Moreover,many of these patients are cognitively impaired. The setting and procedure may be particularly stressful for them, and they may become agitated, especially during femoral arterial cannulation, increasing the risk of femoral artery injury. Sedation may be required, and the use of propofol23 and dexmedetomidine24 for angiography has been reported. The child’s ability to cooperate for testing must be carefully assessed, asan uncooperative child not being able to complete the test will not only fail to provide memory and language information but also subject the child tothe potential harms of angiography as outlined above.
CONCLUSION
Presurgical planning for patients with temporal lobe epilepsy requires the close collaboration of multiple specialties. Despite the recent drive to replace it with noninvasive techniques, the Wada test remains a useful instrument in selected candidates for assessing cerebral dominance andpredicting postoperative cognitive decline. Understanding the principles of the Wada test is essential for anaesthesiologists to facilitate the procedure and prepare for any perioperative challenges and neurocognitive risks that the patient may encounter.
REFERENCES
- Kwan P, Sander The natural history of epilepsy: an epidemiological view. J Neurol Neurosurg Psychiatry. 2004;75(10):1376-1381.
- Baumgartner C, Koren JP, Britto-Arias M, Zoche L, Pirker Presurgical epilepsy evaluation and epilepsy surgery. F1000Res. 2019;8:F1000 Faculty Rev-818.
- Conradi N, Rosenberg F, Knake S, et Wada test results contribute to the prediction of change in verbal learning and verbal memoryfunction after temporal lobe epilepsy surgery. Sci Rep. 2021;11(1):10979.
- Hamberger MJ, Cole Language organization and reorganization in epilepsy. Neuropsychol Rev. 2011;21(3):240-251.
- Stewart CC, Swanson SJ, Sabsevitz DS, Rozman ME, Janecek JK, Binder Predictors of language lateralization in temporal lobeepilepsy. Neuropsychologia. 2014;60:93-102.
- Chelune Hippocampal adequacy versus functional reserve: predicting memory functions following temporal lobectomy. Arch Clin Neuropsychol. 1995;10(5):413-432
- Patel A, Wordell C, Szarlej Alternatives to sodium amobarbital in the Wada test. Ann Pharmacother. 2011;45(3):395- 401.
- Joshi S, Wang M, Etu J, Nishanian Comparison of intracarotid anesthetics for EEG silence. J Neurosurg Anesthesiol. 2006;18(2):112-118.
- Mariappan R, Manninen P, McAndrews MP, et Intracarotid etomidate is a safe alternative to sodium amobarbital for the Wada test. JNeurosurg Anesthesiol. 2013;25(4):408-413.
- Chui J, Venkatraghavan L, Manninen Presurgical evaluation of patients with epilepsy: the role of the anesthesiologist. Anesth Analg. 2013;116(4):881-888.
- Selwa LM, Buchtel HA, Henry Electrocerebral recovery during the intracarotid amobarbital procedure: influence of interval betweeninjections. Epilepsia. 1997;38(12):1294-1299.
- Loddenkemper T, Mo¨ddel G, Schuele SU, Wyllie E, Morris HH Seizures during intracarotid methohexital and amobarbitaltesting. Epilepsy Behav. 2007;10(1):49-54.
- Mikuni N, Takayama M, Satow T, et Evaluation of adverse effects in intracarotid propofol injection for Wada test. Neurology. 2005;65(11):1813-1816.
- Loddenkemper T, Morris HH, Mo¨ddel Complications during the Wada test. Epilepsy Behav. 2008;13(3):551-553.
- Beimer NJ, Buchtel HA, Glynn One center’s experience with complications during the Wada test. Epilepsia. 2015;56(8):e110-e113.
- Catapano JS, Whiting AC, Wang DJ, et al. Selective posterior cerebral artery amobarbital test: a predictor of memory followingsubtemporal selective J Neurointervent Surg. 2020;12(2):165.
- Tellez-Zenteno JF, Patten SB, Jette´N, Williams J, Wiebe Psychiatric comorbidity in epilepsy: a population-based analysis.Epilepsia. 2007;48(12):2336-2344.
- McDonald BC, Saykin AJ, Williams JM, Assaf BA. fMRI Wada test: prospects for presurgical mapping of language and In: Faro SH, Mohamed FB, eds. Functional MRI: Basic Principles and Clinical Applications. New York, NY: Springer New York; 2006;278-314.
- Bauer PR, Reitsma JB, Houweling BM, Ferrier CH, Ramsey NF. Can fMRI safely replace the Wada test for preoperative assessment oflanguage lateralisation? A meta-analysis and systematic J Neurol Neurosurg Psychiatry. 2014;85(5):581.
- Massot-Tarru´s A, Mousavi SR, Mirsattari Comparing the intracarotid amobarbital test and functional MRI for the presurgicalevaluation of language in epilepsy. Curr Neurol Neurosci Rep. 2017;17(7):54.
- Szaflarski JP, Gloss D, Binder JR, et Practice guideline summary: Use of fMRI in the presurgical evaluation of patients with epilepsy.Neurology. 2017;88(4):395.
- Massot-Tarru´s A, White K, Mirsattari Comparing the Wada test and functional MRI for the presurgical evaluation of memory intemporal lobe epilepsy. Curr Neurol Neurosci Rep. 2019;19(6):31.
- Masters LT, Perrine K, Devinsky O, Nelson PK. Wada testing in pediatric patients by use of propofol anesthesia. Am J Neuroradiol.2000;21(7):1302.
- Bharadwaj S, Venkatraghavan Dexmedetomidine sedation for WADA test with intracarotid propofol in pediatric patients. J Neurosurg Anesthesiol. 2015;27(4):352-353.



