General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Rapid sequence induction:
a. is always accompanied by an intravenous opioid
b. requires titration of anaesthetic agent until loss of consciousness is achieved
c. is not required in elective operations
d. is frequently modified to suit clinical circumstances
e. requires at least three personnel - Cricoid pressure:
a. requires a constant 30N of applied pressure
b. should be removed if a poor view is achieved with laryngoscopy
c. is safe in the presence of laryngeal injury
d. reduces aspiration risk by preventing passage of vomited gastric contents
e. can be performed by an untrained assistant - Risks of rapid sequence induction include:
a. elevation of intracranial and intraocular pressure
b. awareness
c. aspiration of gastric contents
d. cardiac arrest
e. bradycardia,in children
Key Points
- Rapid sequence induction (RSI) is intended to reduce the risk of aspiration by minimising the duration of an unprotected airway.
- Preparation and planning – including technique, medications, team member roles, and contingencies – is desirable prior to every RSI.
- Substantial variability in practice exists, therefore institutional factors and clinical circumstances should be considered when determining how to perform an RSI.
INTRODUCTION
Rapid sequence induction (RSI) is a method of achieving rapid control of the airway whilst minimising the risk of regurgitation and aspiration of gastric contents. Intravenous induction of anaesthesia, with the application of cricoid pressure, is swiftly followed by the placement of an endotracheal tube (ETT).
Performance of an RSI is a high priority in many emergency situations when the airway is at risk, and is usually an essential component of anaesthesia for emergency surgical interventions. RSI is only required in patients with preserved airway reflexes. In arrested or completely obtunded patients, an endotracheal tube can usually be placed without the use of medications.
History of RSI
RSI was originally described in 1961 by Sellick1 as:
- Emptying of the stomach via a gastric tube which is then removed
- Pre-oxygenation
- Positioning the patient supine with a head-down tilt
- Induction of anaesthesia with a barbiturate (e.g. thiopentone) or volatile, and a rapid-acting muscle relaxant (e.g. suxamethonium)
- Application of cricoid pressure
- Laryngoscopy and intubation of the trachea with a cuffed tube immediately following fasciculations
The above classic method is now very rarely followed in full. In current clinical practice, a number of modifications have been made to the traditional RSI technique (Figure 1). The term ‘modified RSI’ is sometimes used to describe such variations but this term lacks a commonly accepted definition.

Figure 1: Modifications of RSI technique in current practice
CRICOID PRESSURE
Cricoid pressure is the application of force to the cricoid cartilage of the patient (Figure 2). The rationale is that the upper oesophagus is occluded by being compressed between the trachea and the cervical vertebrae, preventing passive reflux of gastric contents and subsequent development of aspiration pneumonitis. 10 Newtons of force is applied by the thumb and index finger of an assistant increasing to 30N once consciousness is lost. (10N is the equivalent of around 1kg of pressure.) This pressure is maintained until endotracheal tube placement is confirmed.
Cricoid pressure should be reduced or released if laryngoscopy is difficult, or if vomiting occurs (to reduce the chance of oesophageal rupture from active vomiting).
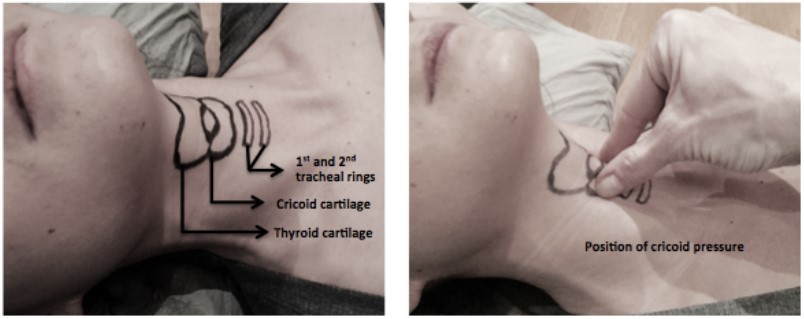
Figure 2: Surface anatomy for cricoid pressure
INDICATIONS
RSI is indicated in patients who require endotracheal intubation and are at increased risk of reflux and aspiration of gastric contents. This means that RSI is almost universally required in situations calling for emergent endotracheal intubation. In fact, non-RSI intubation is almost exclusively the domain of the elective operating environment.
Patient factors may dictate the need for certain elements of the RSI to be modified or omitted. Contraindications to suxamethonium such as allergy, susceptibility to malignant hyperthermia or hyperkalaemia should prompt usage of an alternative muscle relaxant such as high-dose rocuronium. A laryngeal injury contra-indicates cricoid pressure.2 Unstable cervical spine fractures will require caution in the application of cricoid pressure due to the possibility of exacerbating damage.3

PREPARATION
Preparation is vital, both of equipment and team members – particularly if the team is unfamiliar with the environment or their colleagues. Anticipation of difficult airway and establishing oxygenation plans prior to conducting RSI are essential. Many organisations advocate the use of a checklist to ensure that all equipment is available and in working order, and that the planned sequence of events is shared with all team members. An example of an emergency intubation checklist is shown in figure 3.
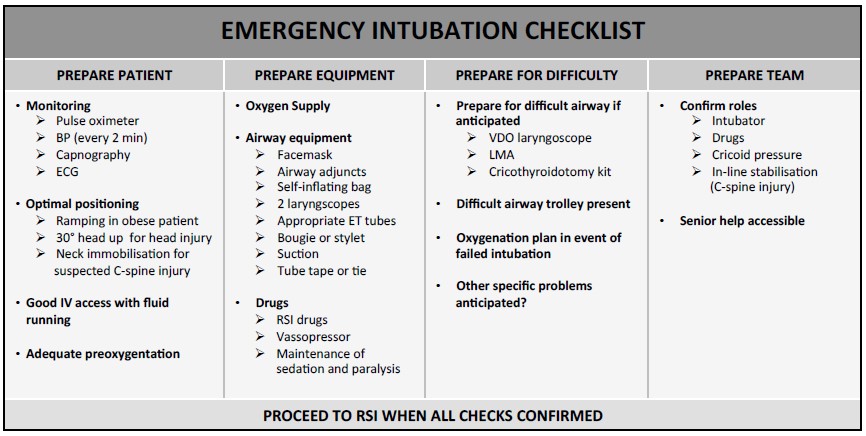
Figure 3: A checklist used in preparation for emergency intubation to ensure that drugs and equipment are available and that all team members are aware of their roles and the planned sequence of events
Preparation of patient
Explanation should be offered to the patient, providing a description of the planned technique including cricoid pressure. Their cooperation for effective preoxygenation (or denitrogenation) will also be required. An alert patient may be offered a non-particulate antacid beforehand, such as 30mL of sodium citrate 0.3 molar: this is most commonly used in the obstetric population.
Preparation of equipment
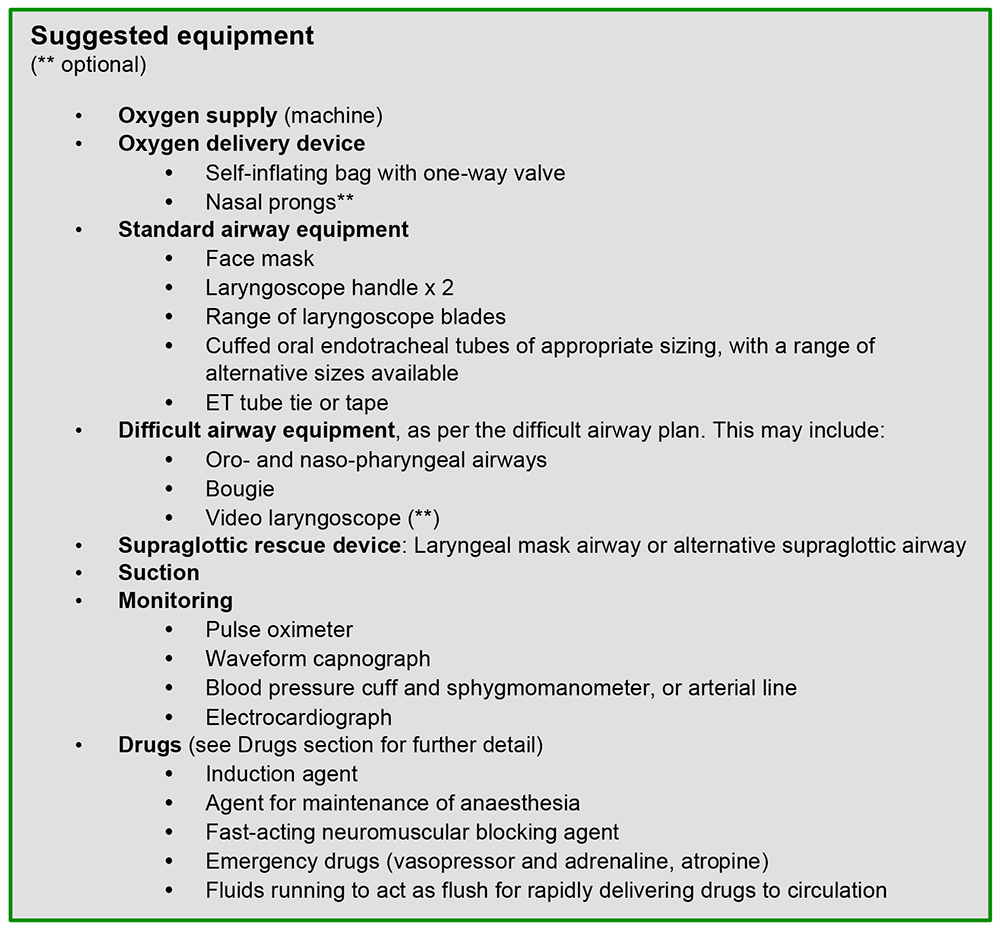
Preparation of drugs
Hypnotics
Five drugs are commonly used to induce anaesthesia: propofol, ketamine, etomidate, thiopentone and midazolam.
Propofol (1-3 mg/kg) is commonly used in the operating theatre for patients who are haemodynamically stable. In elderly or hypovolaemic patients, the dose is drastically reduced: often 0.5-1mg/kg is sufficient, although time to effect is increased due to lower cardiac output.
Ketamine (1-2mg/kg) is increasingly used in pre-hospital settings and in unstable patients. The usual effect is an elevation in heart rate and variable but modest blood pressure changes. Secretions increase, which may necessitate suctioning or premedication with an anti-sialagogue such as atropine or glycopyrrolate.
Etomidate (0.3mg/kg) also has very limited haemodynamic effects. Use has been limited by concerns of adrenal suppression, and there is limited availability in some countries.
Thiopentone (3-5mg/kg) has the most rapid and predictable effect, with less haemodynamic instability than propofol. However, there may be issues with poor availability and the harmful sequelae following extravasation or intra-arterial injection should be considered.
Midazolam (0.1-0.2mg/kg) may be used, although the time to effect may be very prolonged. It is most suitable in patients who are already obtunded and primarily require amnesia rather than true anaesthesia.
Careful dosing of any drug used is more important than the choice of drug. In shocked patients a very modest dose of hypnotic may be sufficient as these drugs can easily lead to circulatory collapse and cardiac arrest. Resuscitation drugs should be readily available.
Neuromuscular blocking agents and reversal agents
For many decades, suxamethonium (succinylcholine) has been the standard of care for RSI. It is usually easily available and reliable. In full dose (1-2mg/kg) it produces fasciculations, paralysis, and excellent intubating conditions within one circulation time (15-45 seconds).
Adverse effects are well-recognised, although uncommon. Myalgia is the most common, but bradycardia, hyperkalaemia-induced cardiac arrest, anaphylaxis, and triggering of malignant hyperthermia can all occur. Many clinicians continue to use suxamethonium unless there is evidence of susceptibility to one or more of these severe adverse events – for example: recent burns, spinal cord injury or a history of muscular dystrophy.
Rocuronium is an alternative agent. In high dose (a range of 0.9-1.6mg/kg has been described) profound relaxation is obtained within 45-60 seconds.4 One disadvantage is a lack of fasciculations, so other methods of ensuring adequate laryngeal paralysis must be used. The prolonged duration of action must be taken into account if the airway is likely to be difficult and if the specific reversal agent is not available. Anaphylaxis is still a possibility, but the risks of myalgia, hyperkalaemia, and malignant hyperthermia are avoided.
A further benefit of rocuronium is the existence of a specific reversal agent. Sugammadex binds avidly to rocuronium, making it unavailable to bind at the neuromuscular junction and reversing the effect. This is useful for elective reversal preceding extubation, and also for restoring neuromuscular function rapidly if required by the difficult airway plan. Sugammadex 16mg/kg can rapidly reverse the effect of rocuronium but usage may be inhibited by the high cost and resulting limited stock. It may also be completely unavailable in some settings, and there can be issues with unfamiliarity with its use. Side effects include interaction with hormonal contraceptive drugs. There is only limited availability in some countries (sugammadex only achieved US FDA approval in December 2015). Other common neuromuscular blocking agents are too slow in onset to provide sufficiently rapid intubating conditions and so would require prolonged mask ventilation of the patient.
Pharmacological adjuncts
In shocked patients, no adjuncts may be required. However, in systemically well patients, or patients at risk of severe hypertension during induction (for example pre-eclampsia, head injury or an unprotected intracranial aneurysm), ablating the pressor response to laryngoscopy is often desirable.
Opioids are commonly used: fentanyl (1-2mcg/kg), alfentanil (10-15mcg/kg), or remifentanil (0.5-1 mcg/kg) are all sufficiently rapid-acting for use in RSI.
Lidocaine (lignocaine) (1-1.5 mg/kg) is also effective at reducing cough and bronchospasm, solely or in combination with an opioid.5
Preparation of team members
Tasks that need to be allocated and performed include:
- Pre-oxygenation
- Intubation
- Assisting the intubator (passing equipment, etc.)
- Drug administration
- Cricoid pressure application (if used)
- Manual in-line stabilisation (if indicated)
A minimum of two people are required to fill these roles. Commonly the lead intubator will also pre-oxygenate and administer drugs, while the assistant applies cricoid pressure, and passes equipment to the intubator. A third person may be required for manual in-line stabilisation of the neck if cervical spine injury is suspected.
RSI technique
The patient should be positioned appropriately for pre-oxygenation and intubation; this may involve ramping, manual inline stabilisation, or a semi-recumbent position for pre-oxygenation if respiratory function is impaired by lying supine.
A reliable intravenous cannula should be placed, and a carrier fluid should run freely to maximise drug delivery to the central circulation. Alternatively, a large syringe of saline may be used to flush medication following administration.
Preoxygenation, or denitrogenation, should be performed as completely as possible having regard to the clinical urgency. At a minimum, three minutes of tidal breathing, or eight vital capacity breaths in one minute should be performed with an inspired oxygen concentration of 100%. Alternatively, if a gas analyser is available, the end-tidal oxygen fraction should reach at least 0.8. If the available device offers an option for positive end-expiratory pressure (PEEP) or positive pressure during patient initiated breaths (pressure support ventilation, BiPAP or similar), then this may be useful to aid pre-oxygenation in some patients.
When satisfactory pre-oxygenation has been obtained, and all team members are ready to proceed, the chosen medications should be administered and the patient should be observed for evidence of effect. If cricoid pressure is to be used, it should be in situ and increased from 10N to 30N at the moment consciousness is lost.
When intubating conditions are obtained, either by observing fasciculations (if suxamethonium is used), waiting for an appropriate time period, or using a neuromuscular monitor, intubation should be performed. Given the need to rapidly secure the airway, first pass success is highly desirable. Accordingly, many practitioners use a bougie or stylet as routine, and if available a video laryngoscope may maximise chance of success.
If regurgitation is observed, suction should be rapidly applied, and the bed should be placed in a head-down (Trendelenburg) position to minimise the chance of aspiration into the trachea.
Once the endotracheal tube is placed, the cuff is immediately inflated and correct position should be confirmed by multiple means. Observing chest rise and fall, tube misting, and a normal feeling of airflow in and out of the endotracheal tube are useful, but neither sufficiently sensitive nor specific. The gold standard is the appearance of a 4 phase capnography waveform for 5 breaths, although this is reliant on cardiac output. Auscultation, in conjunction with the clinical assessment methods described above, must be used if capnography is unavailable. Cricoid pressure, if used, should be released only when endotracheal intubation is confirmed. Auscultation of the chest also helps to exclude endobronchial intubation (although it is poorly sensitive for this diagnosis), and in high resource environments a chest radiograph is often obtained when prolonged intubation is anticipated (for example, in the intensive care unit following an intubation for head injury). Ongoing sedation should be provided, and if required, a long-acting muscle relaxant may be administered.
FAILED INTUBATION
Inability to intubate the patient during an RSI should prompt the usual approach to a difficult intubation, which should be communicated to the team prior to induction. However, if the indication was, for example, respiratory failure, allowing the patient to wake and breathe spontaneously may not be feasible.
If the initial intubation attempt is unsuccessful, a best attempt at face-mask ventilation should be performed while preparing either a supraglottic airway, a different laryngoscopy technique or a new operator. Attempts at intubation should be limited, and the persistent risk of regurgitation and aspiration is remembered. Rarely, a surgical airway may be required and equipment for this should be available at every RSI.
Several algorithms for managing failed intubation have been produced, including the Difficult Airway Society guidelines6 and the Vortex approach.7
CONTROVERSY AROUND CRICOID PRESSURE
Although routinely performed in many parts of the world – particularly the United Kingdom, North America, and Australia – cricoid pressure is not established practice elsewhere and is contentious. It is not common practice in Europe, and some pre-hospital organisations do not endorse its use. It may also not be used in areas of the world where there is a lack of dedicated personnel to assist the anaesthetist.
Concerns include:
- reduced quality of laryngoscopy,
- lack of evidence of effectiveness in preventing reflux and aspiration,
- reduced lower oesophageal sphincter tone and therefore increasing reflux risk,
- worsening of undetected laryngeal or cervical spine injury,
- unmeasurable and likely highly variable location, direction, and degree of force being applied by the operator,8
- patient discomfort, gagging or coughing, and
- increasing physical and cognitive workload for the operators.
A recent systematic review has found no data from randomised trials providing any clinically relevant outcome measures.9 Despite the ongoing controversy, use is considered standard of care in many settings. It is advisable to seek guidance from individual institutions about their expectations and guidelines.
RECENT DEVELOPMENTS
Classically ventilation is not usually provided during the apnoeic period, (to avoid inflation of the stomach and associated increased risk of regurgitation) but some anaesthetists may give a single breath, or several gentle breaths, to both confirm that mask ventilation is possible and reduce the development of hypercapnia, acidaemia, and hypoxia. Some recent guidelines now advocate use of mask ventilation for this reason in patients at elevated risk of hypoxia, for example, the pregnant patient.10,11
Recently apnoeic oxygenation is increasingly being used, especially in critically unwell patients, to provide an oxygenrich environment in the oropharynx to minimise hypoxia during the apnoeic period of RSI.12 This is provided by an alternative oxygen source; commonly via nasal prongs with oxygen flow at 10 litres per minute or more, or via insertion of tubing carrying oxygen into the oropharynx. Limitations can include difficulty with the face-mask seal, pressure-induced damage from misplaced tubing (e.g. gastric rupture), and lack of efficacy in at least one trial.13
SPECIFIC CLINICAL SITUATIONS
Obstetrics
Patients in the second or third trimester of pregnancy are at higher risk of aspiration due to anatomical and physiological changes. These patients are also more likely to be difficult to intubate, and desaturate faster than non-pregnant women.
For these reasons, RSI is employed with meticulous attention to positioning, pre-oxygenation, and availability of difficult airway equipment and expertise. Routine, gentle face-mask ventilation may be utilised.10 Use of opioids as an adjunct to induction may be required if the pregnancy is complicated by hypertension or pre-eclampsia.
As with all advanced pregnancies, the woman should be positioned with a left tilt using a wedge or a tilted table.
Paediatrics
Neonates, infants and children desaturate rapidly and can have pronounced vagal responses to laryngoscopy. A standard approach to RSI is generally performed, with a range of appropriately-sized equipment, and carefully calculated drug dosages. Required doses may be higher than for adults on a per kilogram basis – for example, a 3 year old child will often require 5mg/kg of propofol, several times higher than the proportional adult dose due to the larger volume of distribution. Atropine as an additional adjunct is often used in a dose of 20mcg/kg to reduce bradycardia.
Suspected or known cervical spine injury
In patients who have an unstable cervical spine injury, laryngoscopy with attendant manipulation of the head and neck presents a risk of worsening any injury. There is no consensus as to the safest way to intubate these patients, but two alternative approaches are commonly practiced. One is to perform a laryngoscopy with minimal movement of the cervical spine, often with a hyper-angulated video laryngoscope if it is available, whilst the neck is immobilised by an assistant performing manual in-line stabilisation. The second option is an awake fibre-optic technique, using local anaesthesia only.
EMERGENCE
In patients for whom an RSI was indicated due to aspiration risk, emergence remains a high-risk time for further aspiration events. Strong consideration should be given to extubating the patient awake with full reversal of neuromuscular blockade. Left lateral head-down positioning may further reduce the chance of aspiration, at the expense of reduced access to the airway.
SUMMARY
- Rapid sequence induction is performed to secure the airway in patients at elevated risk of aspiration.
- Preparation of equipment, drugs, the team and the patient is essential; excellent communication should be routine.
- The technique may be tailored to the specifics of the clinical scenario.
ANSWERS TO QUESTIONS
- Rapid sequence induction:
a) False. RSI often is accompanied by an opioid, but may be performed without adjuncts.
b) False. RSI is classically described as administration of a pre-determined dose of anaesthetic agent, and some practitioners perform RSI in this manner when speed is a priority (for example, emergency general anaesthesia for caesarean sections).
c) False. May be required in an elective, fasted, patient if they are at higher risk of aspiration due to pregnancy, gastro-oesophageal reflux, gastric banding, etc.
d) True. RSI is usually modified from the classical description.
e) False. Two people is the minimum for safely when performing an RSI, although further assistants may be useful. - Cricoid pressure
a) False. Cricoid pressure is initially 10N, then increases to 30N with loss of consciousness.
b) True. If laryngoscopy is difficult, cricoid pressure should be removed.
c) False. Laryngeal injury contra-indicates cricoid pressure.
d) False. Vomiting is a contra-indication to cricoid pressure as this risks oesophageal rupture. Only passive regurgitation is reduced by cricoid pressure.
e) False. Some instruction is required to enable cricoid pressure to be safely and effectively applied. - Risks of rapid sequence induction include
a) True. Elevation of intracranial and intraocular pressure can occur from the stimulation of laryngoscopy, or from use of suxamethonium.
b) True. RSI is a risk factor for awareness, due to either a prolonged intubation attempt, relative underdose of induction drugs, or neglecting to provide ongoing sedation.
c) True. Aspiration is a risk whenever intubation is attempted.
d) True. Cardiac arrest can result from relative overdose of induction drugs, hypoxaemia, acidaemia, or anaphylaxis.
e) True. Well-developed vagal responses in children can result in profound bradycardia in response to laryngoscopy.
REFERENCES AND FURTHER READING
- Sellick BA. Cricoid pressure to control regurgitation of stomach contents during induction of anaesthesia. Lancet 1961;2:404-406.
- Cicala RS, Kudsk KA, Butts A, et al. Initial evaluation and management of upper airway injuries in trauma patients. J Clin Anesth 1991;3:91-98.
- Prasarn ML, Horodyski M, Schneider P, et al. The effect of cricoid pressure on the unstable cervical spine. J Emerg Med 2015 Oct 13. pii: S0736-4679(15)00939-7. doi: 10.1016/j.jemermed.2015.09.009.
- Han T-H, Martyn JAJ. Onset and effectiveness of rocuronium for rapid onset of paralysis in patients with major burns: priming or large bolus. Br J Anaesth 2009;102:55-60.
- Davidson JA, Gillespie JA. Tracheal intubation after induction of anaesthesia with propofol, alfentanil and i.v. lignocaine. Br J Anaesth 1993;70:163.
- Difficult Airway Society Guidelines available online at: https://www.das.uk.com/guidelines/das_intubation_guidelines (accessed on21/11/15).
- Chrimes N, Fritz P. The vortex approach: management of the unanticipated difficult airway. Available online at: http://www.vortexapproach.com (accessed on 21/11/15).
- Walton S, Pearce A. Auditing the application of cricoid pressure. Anaesthesia 2000;55:1028.
- Algie CM, Mahar RK, Tan HB, et al. Effectiveness and risks of cricoid pressure during rapid sequence induction for endotracheal intubation. Cochrane Database Syst Rev 2015, Issue 11. Art. No.: CD011656. DOI: 10.1002/14651858.CD011656.pub2.
- Mushambi MC, Kinsella SM, Popat M, et al. Obstetric Anaesthetists’ Association and Difficult Airway Society guidelines for the management of difficult and failed tracheal intubation in obstetrics. Anaesthesia 2015;70:1286-1306.
- Law JA, Broemling N, Cooper RM, et al. The difficult airway with recommendations for management. Can J Anesth 2013;60:1089-1118.
- Moran C, Karalapillai D, Darvall J, et al. Is it time for apnoeic oxygenation during endotracheal intubation in critically ill patients? Crit Care Resusc 2014;16:233-235.
- Semmler MW, Janz DR, Lentz JR, et al. Randomized trial of apneic oxygenation during endotracheal intubation of the critically ill. Am J Respir Crit Care Med 2015 Oct 1. doi: 10.1164/rccm.201507-1294OC.



