Basic Sciences
QUESTIONS
These true/false questions are for you to test your current knowledge. The answers can all be found within the text.
- Regarding the re-absorptive processes in the proximal tubule of the nephron:
- Diffusion requires external energy input to drive it
- Active transport moves substances against their concentration gradient
- Glucose reabsorption is an example of secondary active transport
- Reabsorption all starts with the diffusion of Na+ from the lumen into proximal tubule cells
- In the proximal tubule:
- There are multiple layers of cells to increase reabsorption
- 50% of the glomerular filtrate volume is reabsorbed
- Normally, all of the filtered glucose is reabsorbed
- Bicarbonate is secreted
- Regarding the loop of Henle and vasa recta:
- All the loops of Henle are identical
- The ascending limb is impermeable to water
- The tubular fluid becomes hypotonic
- The vasa recta remove solutes from the medulla
-
Regarding the collecting ducts:
-
All collecting ducts pass through the renal medulla
-
They receive 5 L/day of tubular fluid
-
They are naturally very permeable to water
-
ADH acting on the collecting ducts results in high volume, dilute urine
-
INTRODUCTION
Part 1 of this tutorial explained how the structure of the glomerulus and its blood flow leads to the production of an ultrafiltrate of plasma in Bowman’s capsule. This tutorial will explain how nephrons then modify the 180L/day of glomerular filtrate in order to maintain total body water and electrolyte homeostasis, and produce the usual 1 – 1.5L/day of urine that allows excretion of waste products.
DEFINITIONS
The transformation of glomerular filtrate into urine through reabsorption of electrolytes and water by the nephrons relies on several distinct transport processes. The most important of these are defined and described below.
Diffusion
Molecules of solute in solution are in constant random motion (Brownian motion). Overall, the result of this random motion is that solute molecules move from an area of higher concentration to an area of lower concentration i.e. down a concentration gradient. This is simple diffusion, which can also occur across a membrane, for example a cell membrane, providing the membrane is permeable to the solute in question.
Cell membranes contain numerous proteins. These may take the form of ‘channels’, which can open and allow specific ions to pass through, or through a mechanism that carries specific substances across the cell membrane. This is facilitated diffusion. As with simple diffusion, no external energy input is required as solute movement occurs down a concentration gradient.
Active transport
This process requires energy to move solutes against their concentration gradient. Primary active transport relies on energy from the breakdown of adenosine tri-phosphate (ATP) to power a cell membrane protein which ‘pumps’ solutes across a cell membrane. This process of active transport often pumps solute (e.g. Na+ ions) out of a cell, creating a concentration gradient between the outside and inside of the cell. If the concentration is greater outside the cell, the solute will then tend to diffuse back into the cell down its concentration gradient. This often occurs via specific cell membrane transport proteins, which are able to use the action of one solute moving down its concentration gradient to move a second substance against its concentration gradient. This is secondary active transport. The process of transporting the second solute does not directly require energy from the breakdown of ATP, but energy is needed to establish the first solute’s concentration gradient.
Paracellular and transcellular transport
In the nephron, diffusion of water and solutes occurs across the epithelial cells. Solutes and water can move directly through these epithelial cells (transcellular), crossing the cell membrane on both sides, or through the gaps between the cells (paracellular). Movement by active transport (primary or secondary) must be transcellular as the proteins needed for this are embedded in the cell membrane.
Cotransport and antiport
During secondary active transport the two solutes can be carried across a cell membrane in the same direction (co-transport) or in opposite directions (anti-port).
THE PROXIMAL TUBULE AND PERI-TUBULAR CAPILLARIES
The proximal tubule continues from Bowman’s capsule and receives the glomerular filtrate. The role of the proximal tubule is to reabsorb most of the electrolytes and water it receives.
Structure of the proximal tubule
The first part of the proximal tubule (pars convoluta) is responsible for most of the reabsorption that occurs. A single layer of epithelial cells lines the proximal tubule lumen, which are packed with mitochondria to ensure a continuous supply of energy for active transport of solutes. One of the surfaces of these cells forms the wall of the tubule lumen, and this luminal surface is covered by millions of tiny projections known as the ‘brush border’. This arrangement vastly increases the surface area of the cells in order to improve reabsorption.
Adjacent cells are joined by ‘tight junctions’ at their luminal surface. Located at the opposite side of the cells is the lateral surface where spaces exist between adjacent cells – the lateral intercellular spaces. Adjacent to the lateral surface of the epithelial cells is a basement membrane and a peri-tubular capillary (figure 1). The peritubular capillary is a continuation of the efferent arteriole from the glomerulus of the same nephron.
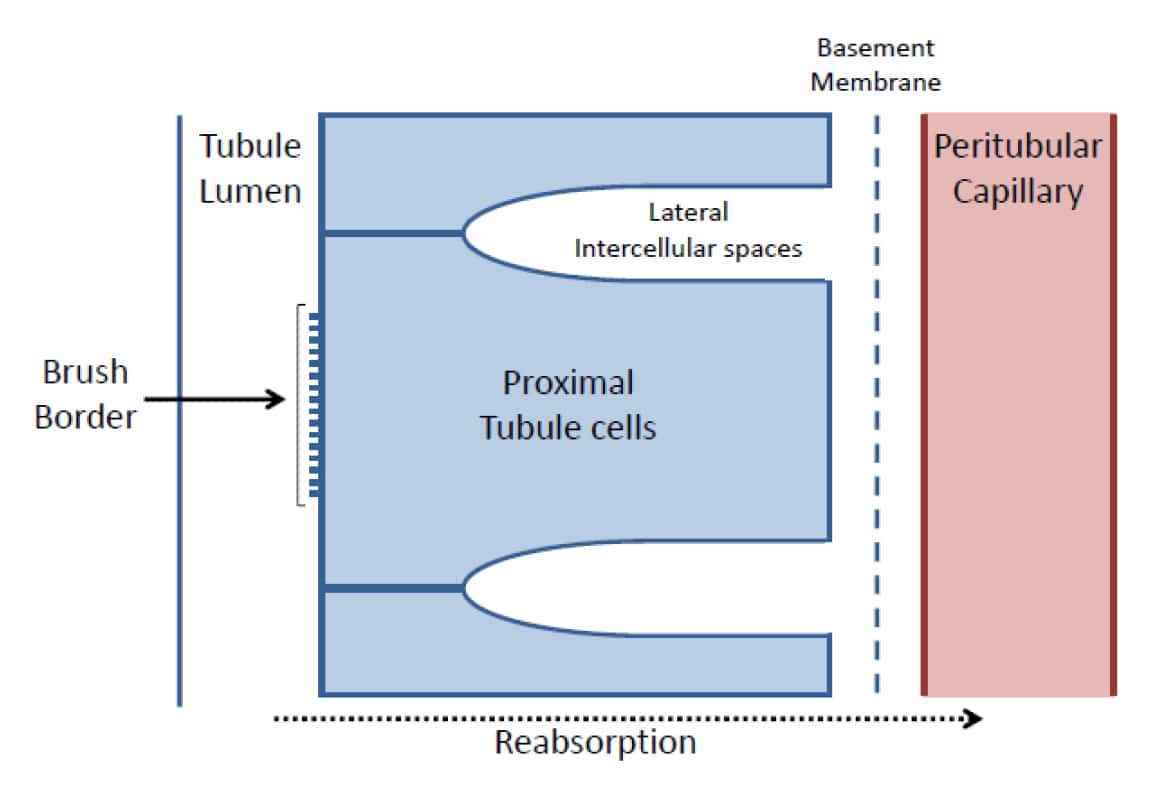
Figure 1. The arrangement of the proximal tubule lumen and epithelial cells, basement membrane and peritubular capillaries.
Reabsorption in the proximal tubule
The proximal tubule receives glomerular filtrate from the Bowman’s capsule. Overall the proximal tubules receive 180L/day of glomerular filtrate and reabsorb approximately 70% (127L/day). The most important substances to be reabsorbed in the proximal tubules are sodium (Na+), bicarbonate (HCO3 -), chloride (Cl–), glucose and water. The proximal tubule is also able to secrete certain substances into the lumen for excretion in the urine. Table 1 outlines substances handled by the proximal tubule.

Table 1. Substances that are reabsorbed and secreted in the proximal tubule. () = percentage reabsorbed from the glomerular filtrate.
Sodium
All reabsorption in the proximal tubule can be considered to begin with the active transport of sodium out of the epithelial cells into the lateral spaces by Na+/K+ ATPases. These ATPases pump three Na+ ions out of the cell and two K+ ions into the cell. This lowers the intracellular Na+ concentration and gives the inside of the cell a slight negative charge, thereby setting up concentration and electrochemical gradients for the facilitated diffusion of Na+ ions from the tubular lumen into the cells via specific carrier proteins (figure 2). This movement of sodium ions can be coupled to the reabsorption of other solutes e.g. HCO3 -, glucose and Cl–. Intracellular K+ concentration is much higher than extracellular fluid K+ concentration and cell membranes are permeable to K+. Therefore, the K+ that is pumped into the cell by the Na+/K+ ATPases simply diffuses back out again. The Na+ that accumulates in the lateral spaces then crosses the basement membrane and enters the peritubular capillary.
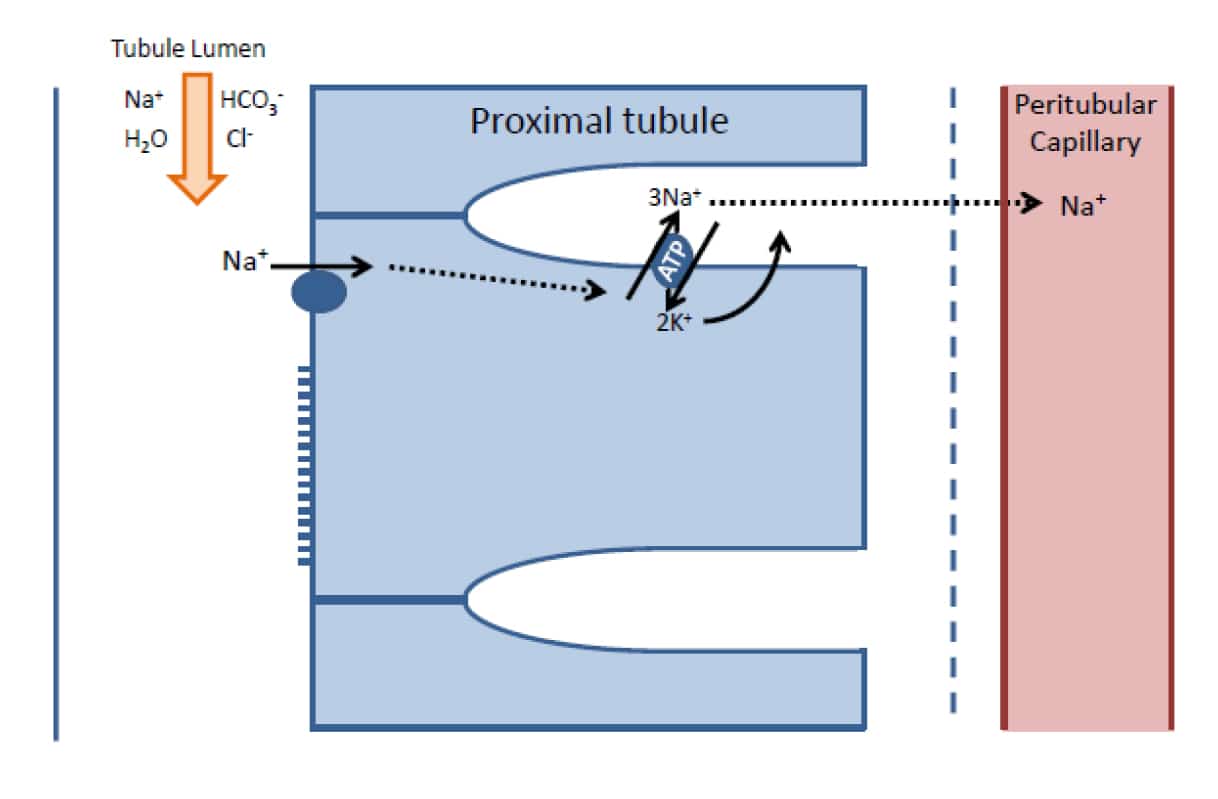
Figure 2. Illustration of the steps involved in sodium reabsorption
Bicarbonate
Some of the carrier proteins that facilitate the diffusion of Na+ into the cells also transport H+ ions out of the cell, into the tubular lumen. In the lumen H+ ions combine with HCO3 – to form carbonic acid (H2CO3), which rapidly dissociates into carbon dioxide (CO2) and water. This series of reactions is catalysed by the enzyme carbonic anhydrase (CA), found in abundance in the brush border of the epithelial cells. The CO2 readily diffuses through the cell membrane into the epithelial cells where the reaction sequence is reversed leading to the formation of H+ (effectively this has been recycled) and HCO3 -. The concentration of HCO3 – within the cell increases, causing it to diffuse into the lateral spaces (figure 3) before entering the peri-tubular capillary. This allows the kidney to conserve the body’s bicarbonate, and is just one of the mechanisms by which the kidney plays a key role in the acid-base homeostasis of the body.
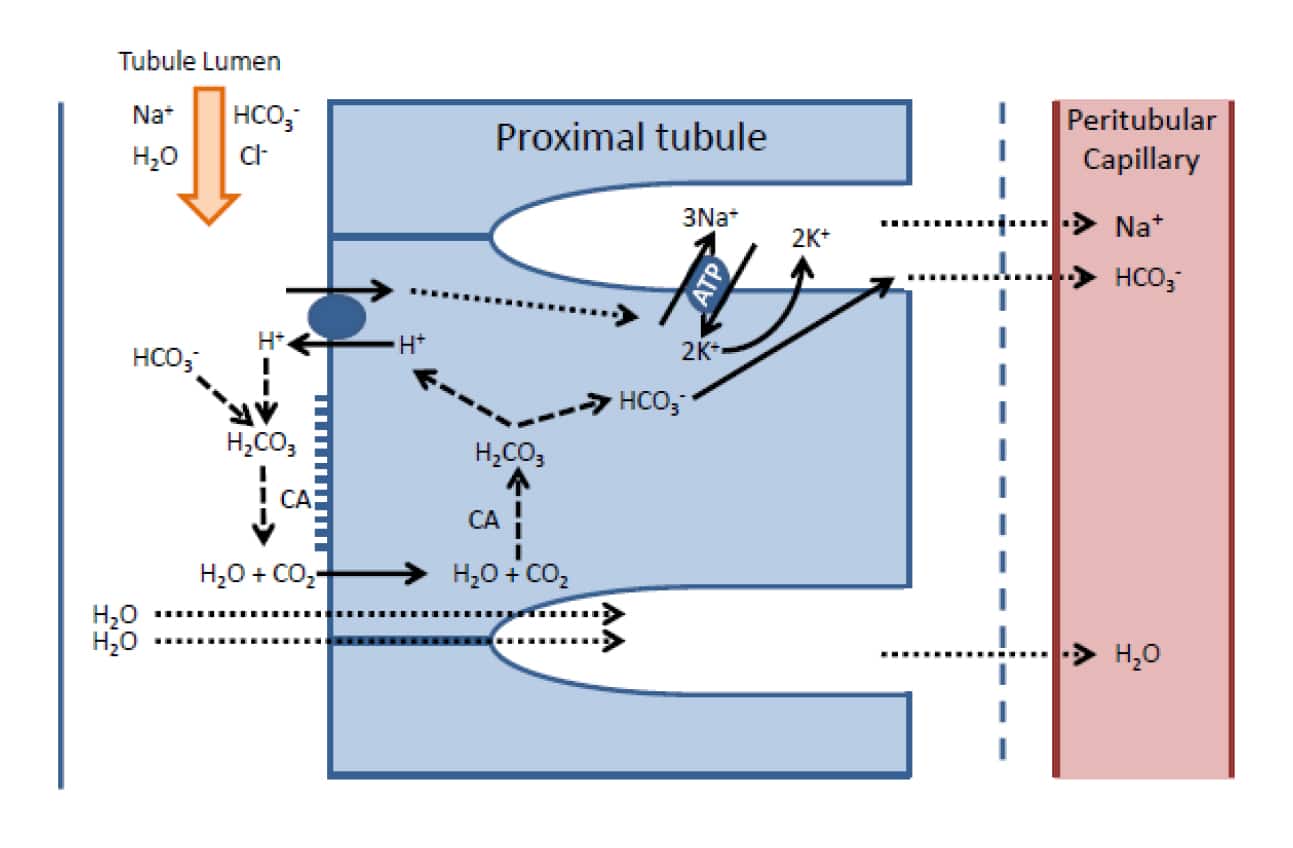
Figure 3. Illustration of the steps involved in Bicarbonate reabsorption, and how water is reabsorbed by osmosis with the electrolytes
Chloride
Cl– is reabsorbed in the proximal tubule in two ways. Firstly, an anti-port mechanism pumps organic anions (HCOOH) into the lumen in exchange for Cl– ions. The organic anions then combine with some of the H+ ions allowing them to be reabsorbed and recycled. Secondly, as Na+ and HCO3 – ions are rapidly reabsorbed early in the proximal tubule the concentration of Cl– ions left in the tubular fluid rises, setting up a concentration gradient for para-cellular diffusion of Cl– ions (figure 4).
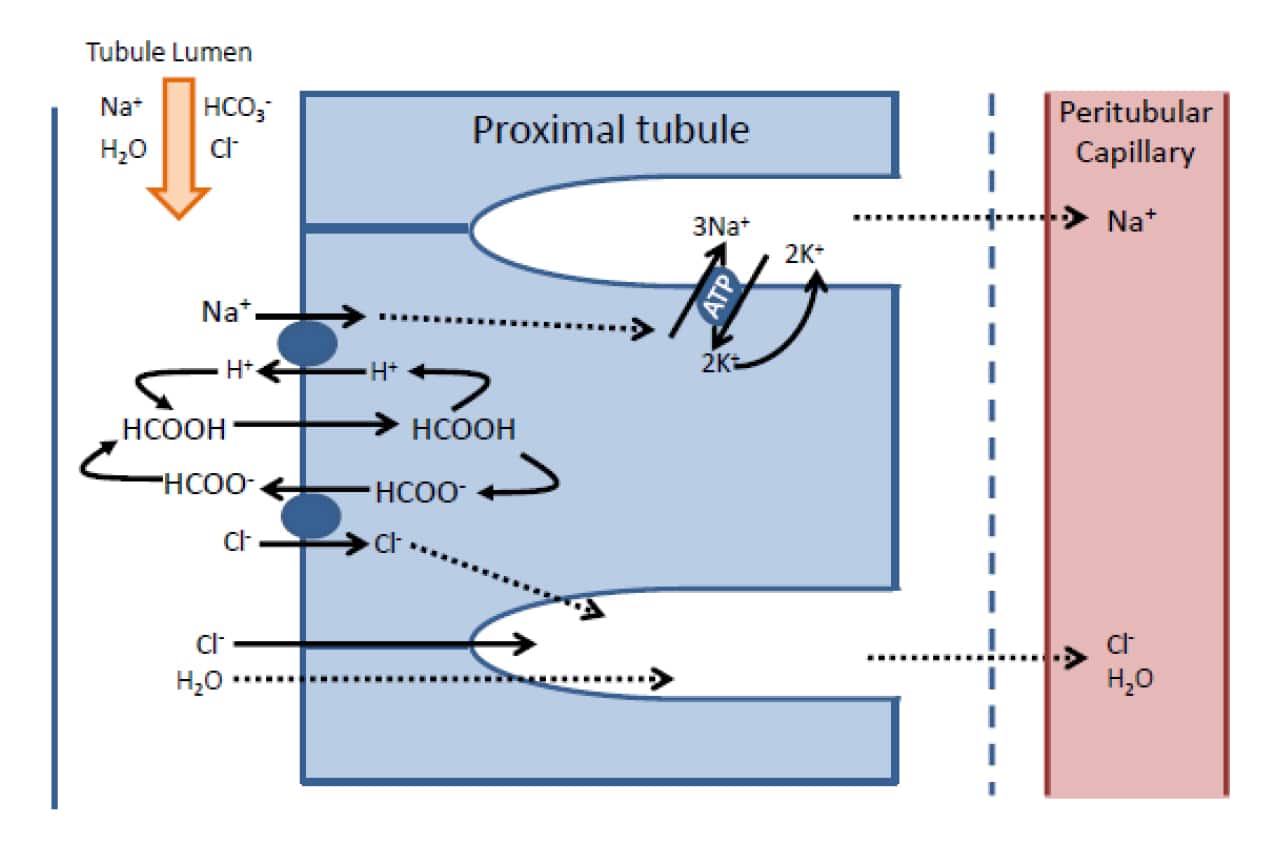
Figure 4. Illustration of the steps involved in chloride and water reabsorption
Water
The epithelial cells lining the proximal tubule are very permeable to water molecules. Every solute ion that is reabsorbed from the tubular fluid into the lateral spaces will reduce the tonicity of the fluid that is left behind in the lumen, and increase the tonicity in the lateral spaces. Water is thus reabsorbed by osmosis via both transcellular and para-cellular routes (figures 3 and 4).
Glucose
Glucose reabsorption is coupled to Na+ reabsorption. As described above, a gradient is established to allow Na+ reabsorption from the tubular fluid. As well as the Na+/H+ ion exchangers involved in HCO3 – reabsorption, there are also glucose co-transporters (sodium glucose transporters, SGLTs). These couple the movement of Na+ ions down their concentration gradient into the cells with the movement of glucose molecules against their concentration gradient into the cells. This is an example of secondary active transport. The glucose concentration within the cell rises. Some of the glucose is used for cellular metabolism and the rest leaves via the lateral surface of the cells (figure 5).
In health, all of the glucose filtered at the glomerulus is reabsorbed in the proximal tubule, with no other mechanisms for reabsorption in more distal parts of the nephron. However, as it is dependent on transport by specific carriers there is a maximum rate at which glucose can be reabsorbed (called the tubular maximum, Tm). This mechanism can be overwhelmed if there is an excess of glucose in the glomerular filtrate due to a high plasma concentration (e.g. diabetes mellitus). If the plasma glucose concentration exceeds approximately 10mmol/L the Tm of the proximal tubule will be surpassed, not all the glucose will be reabsorbed, and some will remain in the tubular fluid and eventually be detectable in the urine.
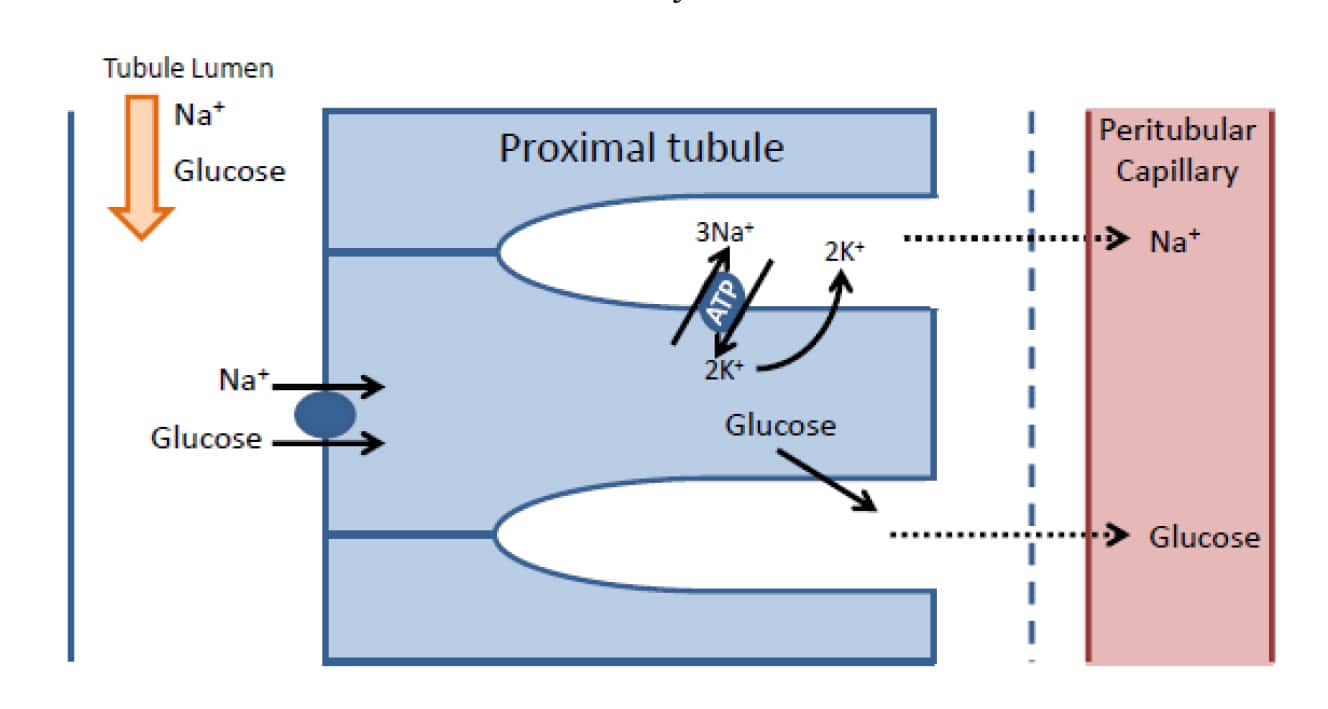
Figure 5. Diagrammatic representation of how Glucose is reabsorbed along with sodium
The Peri-tubular Capillaries
These capillaries are continuations of the efferent arterioles from the same nephrons glomerulus. These capillaries have a very low hydrostatic pressure and a high plasma oncotic pressure as a result of the two sets of arterioles upstream and the loss of water but not proteins as the glomerular filtrate (see part 1 of this tutorial series). The low hydrostatic pressure and high oncotic pressure favours reabsorption of the water and electrolytes that accumulate in the lateral spaces back into the plasma.
THE LOOP OF HENLE AND VASA RECTA
The function of the loop of Henle
The main role of the loop of Henle is to produce a hypertonic environment within the renal medulla to allow the reabsorption of water by osmosis from the collecting tubules in order to retain water within the body and produce concentrated urine. Paradoxically, as part of this process the loop of Henle also alters the tubular fluid osmolality from being isotonic to plasma (290mosm/Kg H2O) to being hypotonic to plasma (i.e. it effectively dilutes it, see below). The descending limb of the loop of Henle is permeable to water, and a further 30L/day of the glomerular filtrate is reabsorbed here. The ascending limb of the loop of Henle is impermeable to water.
As explained in part 1 of this tutorial, there are both short (85%) and long (15%) loops of Henle. Only the long loops of Henle extend down into the renal medulla, and it is only these that play a role in producing the medullary hypertonicity. The mechanism that allows the loop of Henle to establish this large osmotic gradient between the tubular lumen and the medullary interstitium is known as ‘counter current multiplication’.
Counter current multiplication
The tubular cells of the thick part of the ascending limb of the loop of Henle contain Na+/K+ ATPases which actively pump Na+ ions out of the cell and into the medullary interstitium. This sets up a concentration gradient from the tubular lumen into the cell for the reabsorption of other ions (like in the proximal tubule). In this case it leads to the reabsorption of one Na+, one K+ and two Cl– ions via a specific co-transporter. However, as is seen elsewhere in the nephron the K+ readily diffuses back out of the cells meaning that it is only really Na+ and Clthat are reabsorbed (figure 6). Also, the ascending limb of the loop of Henle is impermeable to water. The result is to deplete the tubular fluid of its osmotically active particles rendering it hypotonic, while increasing the concentration of osmotically active particles in the medullary interstitium making it hypertonic.
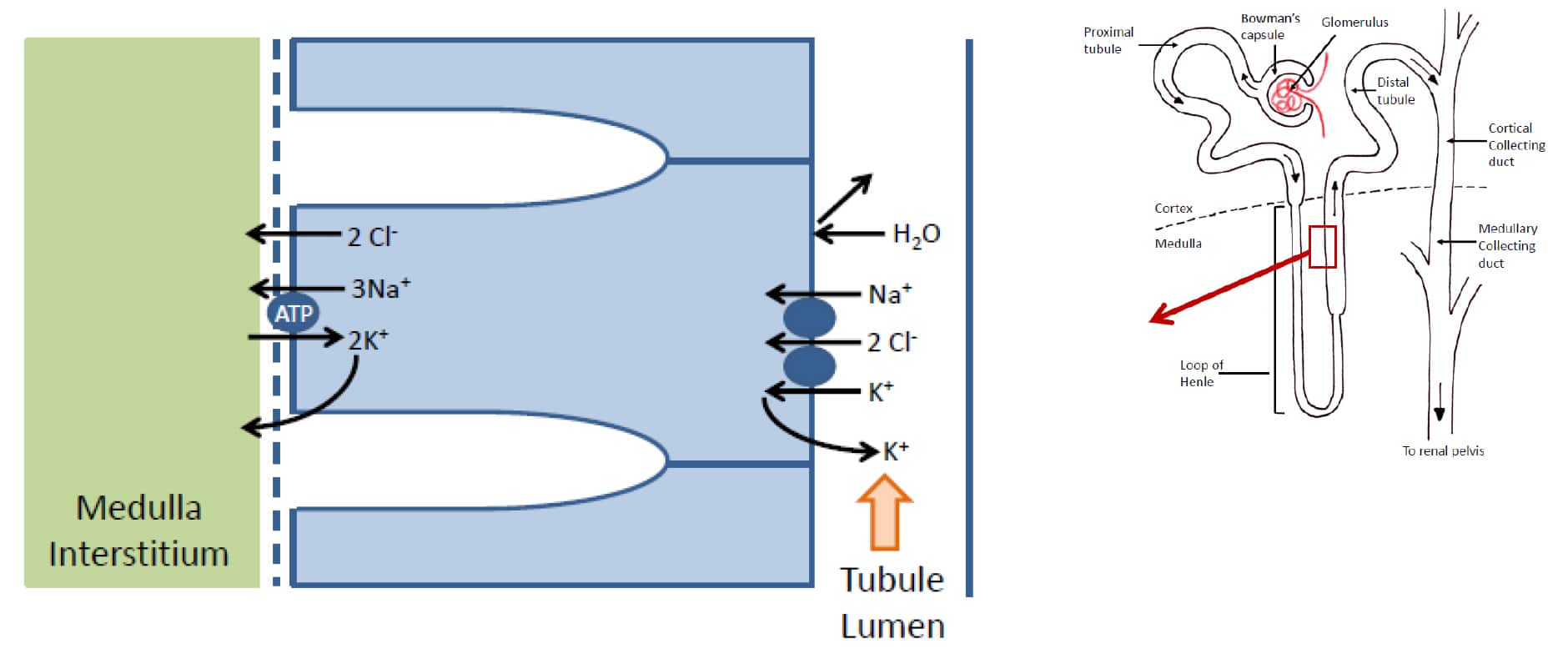
Figure 6. Diagrammatic representation of the reabsorption of electrolytes, but not water, into the medullary interstitium in the ascending loop of Henle
The descending limb of the loop of Henle is permeable to water and ions and so readily equilibrates with the interstitium. The Na+ and Cl– reabsorbed from the ascending limb diffuse into the descending limb. Therefore, in effect, an osmotic gradient is established between the descending and ascending limbs of the loop of Henle. A difference in osmolality between the ascending and descending limbs at any given level within the medulla of around 200mosm/Kg H2O can be achieved (figure 7).
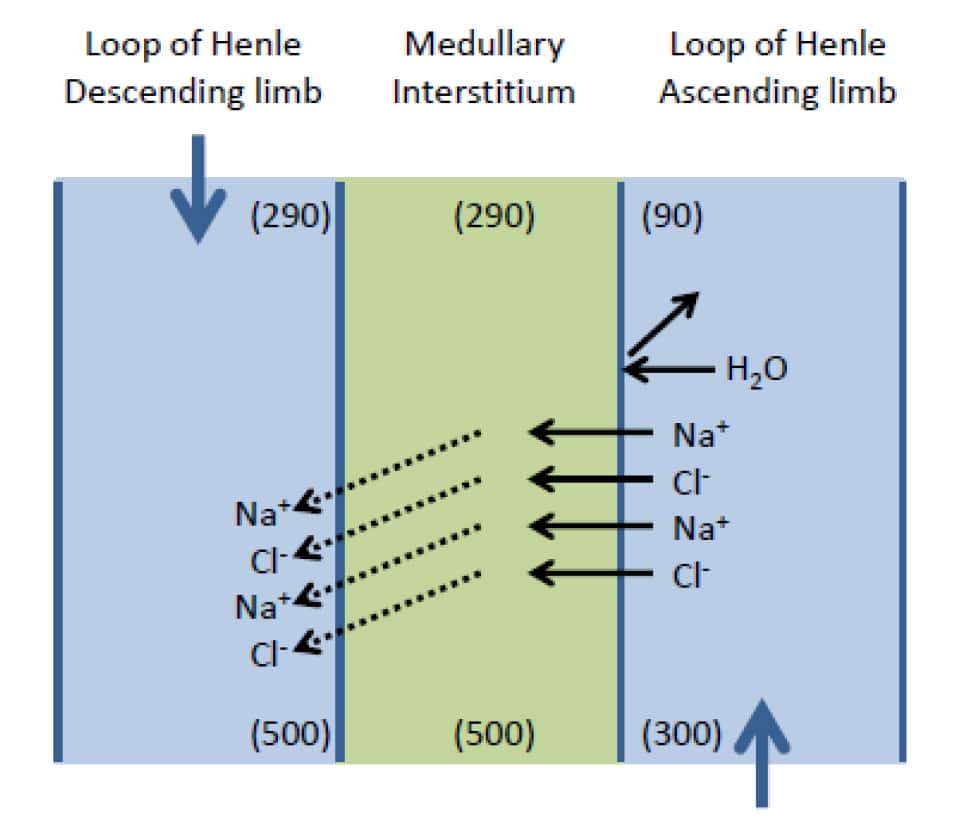
Figure 7. Illustration of the establishment of osmotic difference between the two limbs of the loop of Henle by the transfer of Na+ and Cl–
The absolute values of osmolality in the lumen and interstitium vary hugely up and down the medulla. The constant addition of Na+ and Cl– to the fluid of the descending limb of the loop of Henle leads to a progressive increase in its tonicity until it reaches the tip of the loop of Henle (figure 8). In the ascending limb the tonicity of the fluid decreases as the Na+ and Cl– ions are reabsorbed as described above, ‘trapping’ a very high solute concentration within the medulla. By the time the fluid leaves the loop of Henle its tonicity has fallen to around 90mosm/Kg H2O.
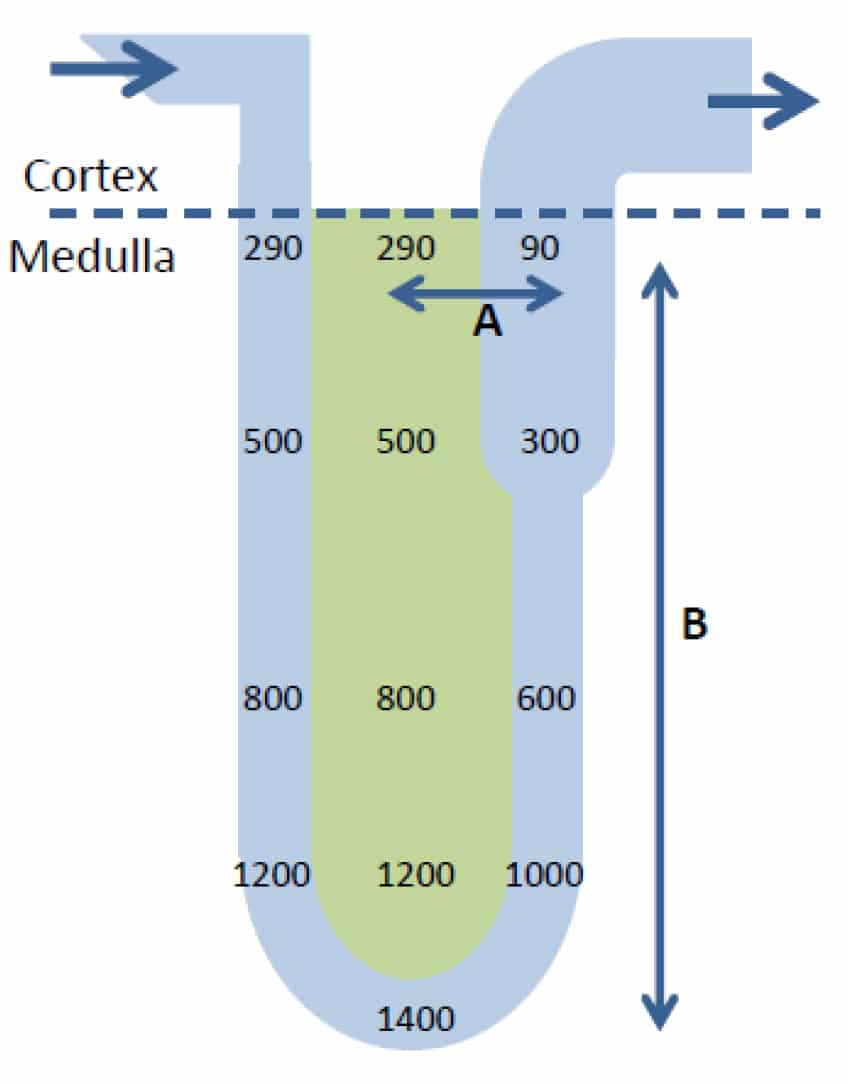
Figure 8. Illustration of how the small osmotic gradient at any given level (A) is multiplied into a much larger osmotic gradient (B) up and down the medulla
As well as the electrolytes described above, a significant contribution (up to 50%) to the final osmolality of the medullary interstitium comes from a high urea concentration in this tissue. Urea from the interstitium diffuses down its concentration gradient into the descending loop of Henle, resulting in a high urea concentration in the tubular fluid. The tubular fluid retains the high urea concentration all the way to the medullary part of the collecting ducts, where it is reabsorbed under the control of anti-diuretic hormone (see below for more details). Through this mechanism the urea is recycled and can help make the medullary interstitium even more hypertonic than the counter-current multiplication mechanism could alone.
Counter current exchange
There is also a specialised network of blood vessels that mirrors the arrangement of the loops of Henle (figure 9). These looped blood vessels are called the ‘vasa recta’ and are necessary in order to supply blood to the renal medulla and still maintain the osmotic gradients established in the medulla by the loop of Henle. If blood was supplied to the medulla and loop of Henle by a simple straight capillary, the hypertonic interstitium deep in the medulla would draw water out of the capillary by osmosis and ruin the work of the loop of Henle by reducing the osmotic gradient. In the descending limb of the vasa recta water is drawn out of the capillaries and solutes enter to maintain osmotic equilibrium as the medullary interstitium becomes increasingly hypertonic. This process is then reversed in the ascending limb of the vasa recta with the water re-entering the capillary and solutes leaving (figure 9) – this is the ‘counter current exchange’. At the same time the blood flowing in the vasa recta delivers nutrients (oxygen, glucose) to, and removes waste products (carbon dioxide) from the renal medulla. The net result of the process is to allow blood to flow through the medulla without altering the osmotic gradients that are present.
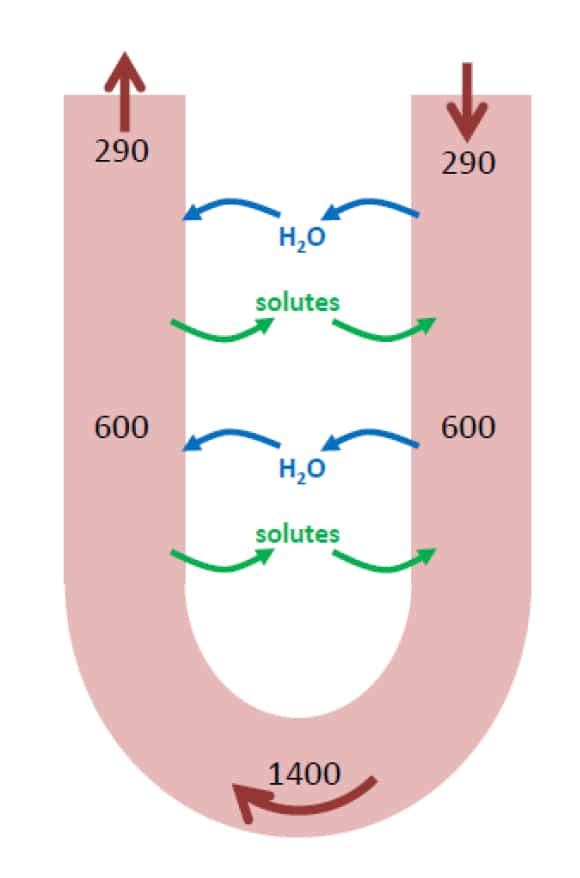
Figure 9. Diagrammatic representation of the loop arrangement of the vasa recta, indicating how the blood flows in the opposite direction to the fluid flow in the loop of Henle. There is solute and water movement in and out of both limbs of the vasa recta, but overall the osmolality of the medulla s not altered.
THE DISTAL TUBULE AND COLLECTING DUCTS
Of the original 180L/day of glomerular filtrate, 127L is reabsorbed in the proximal tubules and a further 30L is reabsorbed in the descending limb of the loop of Henle leaving 23L/day to enter the distal tubules and collecting ducts. Reabsorption of this part of the glomerular filtrate can be varied depending upon extra-cellular fluid volume and osmolality. Clearly this is still a very large volume relative to total body water (more than half of the total body water volume of an average adult), and the vast majority of this must be reabsorbed every day to prevent rapid dehydration. In healthy adults the concentration and volume of urine produced can vary enormously. The range is from about 90mosmol/Kg H2O and 23L/day at its most dilute (if no water is reabsorbed in the collecting ducts) to 1400mosmol/Kg H2O and 400ml/day (if the maximum amount of water is reabsorbed in the collecting ducts). The maximum achievable osmolality of the medulla is reduced at the extremes of age. Babies, young children and the elderly are not able to produce urine with as high concentration and low volume, rendering them more susceptible to dehydration.
The distal tubule is impermeable to water, but some Na+ and Cl– is reabsorbed here. The collecting duct is responsible for controlling what happens to this final 23L/day of fluid. The collecting ducts can be divided into two regions; that part in the cortex of the kidney and the part in the medulla (figure 10). There are several hormones that act on the collecting ducts, but the two most important are anti-diuretic hormone (ADH) and aldosterone.
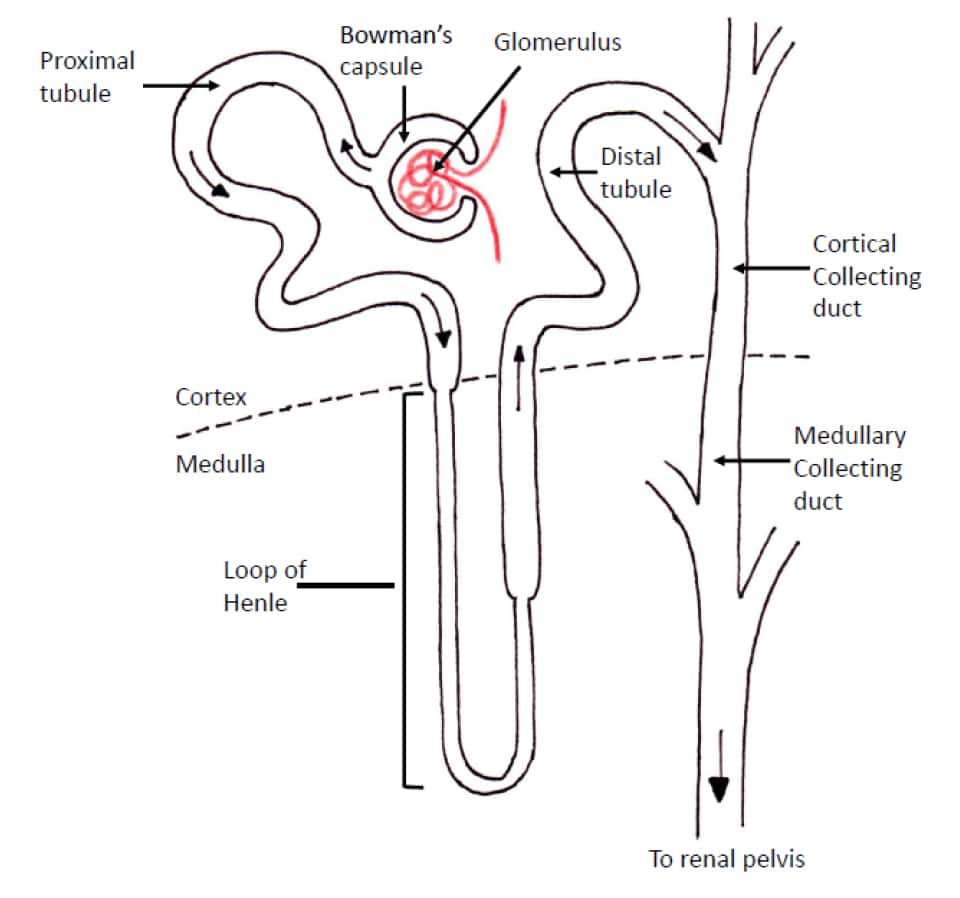
Figure 10. Anatomical arrangement of the distal tubule, with the collecting ducts divided into cortical and medullary parts
Anti-diuretic hormone and the control of extracellular fluid osmolality
In their natural state the collecting ducts are not permeable to water even though there is a huge osmotic gradient between the lumen and the surrounding medullary interstitium. Collecting ducts only become permeable to water under the influence of ADH. ADH is produced in the hypothalamus and secreted into the bloodstream from the posterior pituitary, mainly in response to extra-cellular fluid (ECF) osmolality. ADH controls the retention or loss of water without any electrolytes, so called ‘free’ water. At all other points in the nephron water reabsorption occurs with solute reabsorption so osmolality doesn’t vary. By being able to retain or lose free water in this way ADH controls the ECF osmolality.
If ECF osmolality increases e.g. due to dehydration, an increase in ADH secretion will occur. ADH binds to receptors on the lateral surfaces of the collecting duct cells and leads to an increased production of aquaporins within these cells. Aquaporins are specialised protein channels which insert into the luminal membrane of the collecting duct cells to make them permeable to water. This allows water to be reabsorbed from the collecting ducts by osmosis. ADH also makes the cells of the medullary part of the collecting duct permeable to urea, allowing urea to enter the medullary interstitium where it is osmotically active. This helps to make the medullary interstitium hypertonic so that the maximum amount of water can be reabsorbed from the collecting ducts to try and return ECF osmolality back to normal. The resulting urine will be maximally concentrated and low in volume.
If ECF osmolality decreases e.g. after ingestion of water, there will be a decrease in ADH secretion. As a result less urea and less water will be reabsorbed from the collecting ducts, which will reduce the urine concentration and increase water loss, to return ECF osmolality back to normal.
Aldosterone and the control of extracellular fluid volume
Aldosterone is synthesised in the zona glomerulosa of the adrenal glands, and acts on the cortical part of the collecting duct. Its release is stimulated by a rise in plasma K+ concentration and also by angiotensin II if there is a reduction in the extracellular fluid (ECF) volume. A reduction in ECF volume e.g. haemorrhage, leads to sympathetic nervous system (SNS) activation, a decreased blood flow through renal afferent arterioles and a reduced GFR and nephron tubular fluid flow. These three effects stimulate renin release and hence increase angiotensin II concentration (see part 1 of this tutorial).
Aldosterone increases Na+ reabsorption from the tubular fluid in exchange for K+ and H+ ions. The Na+ that is reabsorbed in this way returns to the ECF and does not lead to water reabsorption as the cortical collecting ducts are impermeable to water. Therefore Aldosterone acts to increase ECF Na+ content, which then via ADH dependent osmoregulatory mechanisms (see above) will lead to retention of water and restoration of ECF volume.
SUMMARY
- The proximal tubule reabsorbs 70% of the volume of glomerular filtrate (along with 70% of most of the electrolytes)
- In health the proximal tubule reabsorbs 100% of the glucose from the glomerular filtrate
- The long loops of Henle establish a hypertonic medullary interstitium which then allows water reabsorption from the collecting ducts of all nephrons
- The variability in urine volume and concentration is dependent on what happens in the collecting ducts
- For simplicity it can be considered that ADH regulates ECF osmolality and aldosterone regulates ECF volume
ANSWERS
- FTTF – Diffusion does not require any external energy. Active transport does require energy to move solute against their concentration gradient. Glucose reabsorption is an example of secondary active transport. All reabsorption starts with the action of the Na+/K+ ATPases acting to lower intracellular Na+ concentration.
- FFTF – There is a single layer of epeithelial cells lining the nephron lumen. 70% of the glomerular filtrate volume, but 100% of the glucose is reabsorbed in the proximal tubule. Bicarbonate is reabsorbed in the proximal tubule.
- FTTF – There are two types of loop of Henle; long and short. The ascending limb of all the loops of Henle is impermeable to water. By the end of the loop of Henle the tubular fluid is hypotonic. The purpose of the vasa recta is to deliver nutrients to the medulla without removing the solutes that have accumulated there.
- All collecting ducts pass through the renal medulla to drain into the renal pelvis. Collectively they receive 23L of tubular fluid per day. In their natural state they are impermeable to water. They become permeable to water due to the action of ADH. This allows most of the water to be reabsorbed resulting in a low volume, concentrated urine.



