Intensive Care Medicine
SELF ASSESSMENT
- Which of the following statements comparing Dialysis with Filtration are true:
- Dialysis depends on diffusion whereas filtration depends on convection
- Filtration is more effective than dialysis at removing small molecules
- Filtration in more effective than dialysis at removing cytokines
- Dialysis is not as effective as Filtration at removing water
- Which of the following statements are true regarding the differences between Continuous RenalReplacement Therapy (CRRT) and Intermittent Haemodialysis (IHD):
- CRRT is more cost effective than IHD
- IHD is preferable to CRRT in patients who are cardiovascularly unstable
- IHD offers an overall survival benefit when compared with CRRT
- CRRT is preferable to IHD in patients with a coexistent acute brain injury
- Are the following statements regarding RRT True or False:
- Poor vascular access often contributes to the clotting of a filter
- RRT has an established role in septic shock with normal renal function
- Protein bound drugs are not easily removed by CRRT/IHD
- The hospital mortality of patients with AKI on RRT is approx 60%
INTRODUCTION
Acute renal failure, also known as acute kidney injury (AKI), is defined as an abrupt (within 48 hours) reduction in kidney function. The AKI network defines the reduction in kidney function as the presence of any one of the following:1
- An absolute increase in serum creatinine of ≥ 0.3 mg.dl-1 (≥ 26.4 mcmol.l-1)
- A percentage increase in serum creatinine of ≥ 50% (1.5-fold from baseline)
- A reduction in urine output (< 0.5 ml.kg-1 per hour for more than six hours).
It is estimated that a third of patients in the critical care setting have an AKI 2 and approximately 5% will require renal replacement therapy (RRT).3 The hospital mortality in patients with an AKI requiring RRT is as high as 60%.4
The initial management of AKI involves treating the underlying cause, stopping nephrotoxic drugs and ensuring that the patient is euvolaemic with an adequate mean arterial blood pressure. However, no specific treatments have been shown to reverse the course of AKI so RRT is the cornerstone of further management.
INDICATIONS FOR RRT
Indications for RRT are:
1. Acute kidney injury (AKI) with:
- Fluid overload (refractory to diuretics)
- Hyperkalemia (K+ > 6.5)
- Severe metabolic acidosis (pH < 7.1)
- Rapidly climbing urea/creatinine (or urea > 30mmol.l-1)
- Symptomatic uraemia: encephalopathy, pericarditis, bleeding, nausea, pruritus
- Oliguria/anuria.
There are no universally accepted levels of urea, creatinine, potassium, or pH at which to start therapy. The figures quoted above are given as a rough guide only. Initiation of RRT should be prompted more by the rate of change of renal parameters and by the patients overall condition than by arbitrary levels.
There is some suggestion that starting RRT early (defined as a urea < 27mmol.l-1 in the PICARD study) may offer a survival benefit, however, guidance as to the exact timing of RRT is still lacking.5
2. Overdose with a dialyzable drug or toxin
Some drugs are removed by RRT but some are not. As a general rule, drugs are cleared by RRT if they are water-soluble and not highly protein-bound. [Please see Table 1]

Table 1. Examples of drugs/toxins that are either removed or not removed by RRT
3. Severe sepsis
There has been recent interest in the potential for haemofiltration to remove inflammatory mediators in patients with severe sepsis/septic shock. A number of small studies (with 25 subjects or less) have suggested that high volume haemofiltration (40-85ml.kg-1.hr-1) may reduce vasopressor requirements and possibly improve survival in patients with septic shock irrespective of whether they have an AKI.6,7,8,9 However, strong recommendations cannot be made about the role of RRT in this area until larger, well designed trials address the issue.
[Please see glossary at end of tutorial for definitions of sepsis, severe sepsis, septic shock]
TYPES OF RRT IN USE IN CRITICAL CARE
RRT encompasses peritoneal dialysis and renal transplantation but for the purpose of this tutorial we will focus on the forms of RRT used in the intensive care setting. These are:
- Intermittent haemodialysis (IHD)
- Continuous renal replacement therapies (CRRT)
- Continuous venovenous haemofiltration (CVVH)
- Continuous venovenous haemodialysis (CVVHD)
- Continuous venovenous haemodiafiltration (CVVHDF)
- Slow continuous ultrafiltration (SCUF)
- Continuous arteriovenous haemofiltration (CAVHD)
- Hybrid therapies e.g. Sustained low-efficiency dialysis (SLED)
The functional differences between the techniques listed above can be classified in terms of:
- The mechanism of solute removal (Filtration versus dialysis)
- The duration of the treatment (Continuous versus intermittent)
MECHANISM OF SOLUTE REMOVAL: Filtration (convection)versus dialysis (diffusion)
Haemofiltration involves blood being pumped through an extracorporeal system that incorporates a semi-permeable membrane. The hydrostatic pressure that is created on the blood-side of the filter drives plasma water across the filter. This process is referred to as ultrafiltration. Molecules that are small enough to pass through the membrane (<50,000 Daltons) are dragged across the membrane with the water by the process of convection. The filtered fluid (ultrafiltrate) is discarded and a replacement fluid is added in an adjustable fashion according to the desired fluid balance.
Haemodialysis involves blood being pumped through an extracorporeal system that incorporates a dialyzer. In the dialyzer, blood is separated from a crystalloid solution (dialysate) by a semi-permeable membrane. Solutes move across the membrane along their concentration gradient from one compartment to the other obeying Fick`s laws of diffusion. For example, bicarbonate moves from dialysate to blood whereas urea and potassium move from blood to dialysate. In order to maintain concentration gradients and therefore enhance the efficiency of the system the dialysate flows countercurrent to the flow of blood. When removal of water is required the pressure on the blood-side of the membrane has to be increased forcing water molecules to pass into the dialysate.
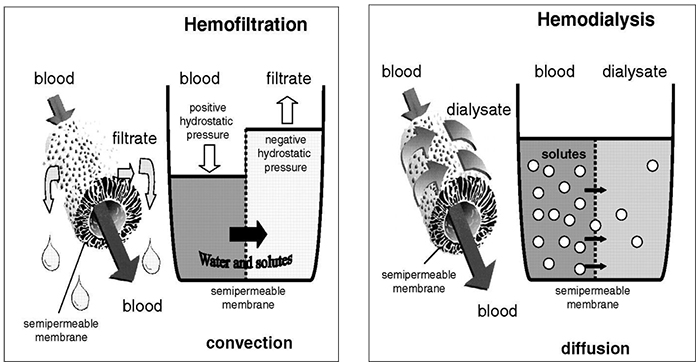
Figure 1. Schematic diagram comparing the different modes of solute removal in haemodialysis and haemofiltration. (Courtesy of emcrit.org)
Haemodiafiltration, as its name suggests, is a combination of filtration and dialysis. It has the benefits of both techniques but to a lesser extent than when the individual techniques are used on their own. There is no evidence to suggest that CVVDF has a survival benefit when compared to CVVH but may be a useful way of increasing clearance of small solutes.
Slow continuous ultrafiltration is used when the only requirement is water removal. Effectively, it is CVVH with a low filtration rate. It can remove up to 6 litres of fluid a day but solute removal is minimal.
DURATION OF TREATMENT: Intermittent (IHD) versus continuous (CRRT) (versus hybrid)
Intermittent haemodialysis involves dialysing with higher flow rates than CRRT for defined periods of time. A typical regime is 3-5 hours of dialysis 3 times a week. The high flow rates and rapid fall in plasma osmolality mean that it is only suitable for patients who are cardiovascularly stable. It forms the basis of long term RRT for chronic renal failure but is not often used in the critical care setting.
CRRT involves filtering and/or dialysing on a continuous basis. It allows better fluid management and creates less haemodynamic disturbance, but it is more expensive than IHD and requires continuous rather than intermittent coagulation. There is some evidence to suggest that CRRT is superior to IHD in patients with sepsis, cardiovascular instability or with a head injury. However, a large RCT comparing IHD with CRRT in patients with an AKI and multiple-organ dysfunction syndrome showed no difference in survival at 60 days.10
Sustained low efficiency dialysis is an example of a hybrid therapy which aims to combine the logistic and cost advantages of IHD with the relative cardiovascular stability of CRRT. Treatments are intermittent but usually daily and with longer session durations than conventional IHD. Solute and fluid removal are slower than IHD, but faster than CRRT. Some are confident that hybrid therapies are the future of RRT in ICU but there is currently a lack of evidence to support its use. At present, it is not a technique used in the UK.
WHICH FORM OF RRT SHOULD WE USE?
No single RRT technique has been shown to offer a survival benefit over the others in the critical care setting, so the decision about which technique to use depends on:
1. What we want to remove from the plasma [Please see Table 2]
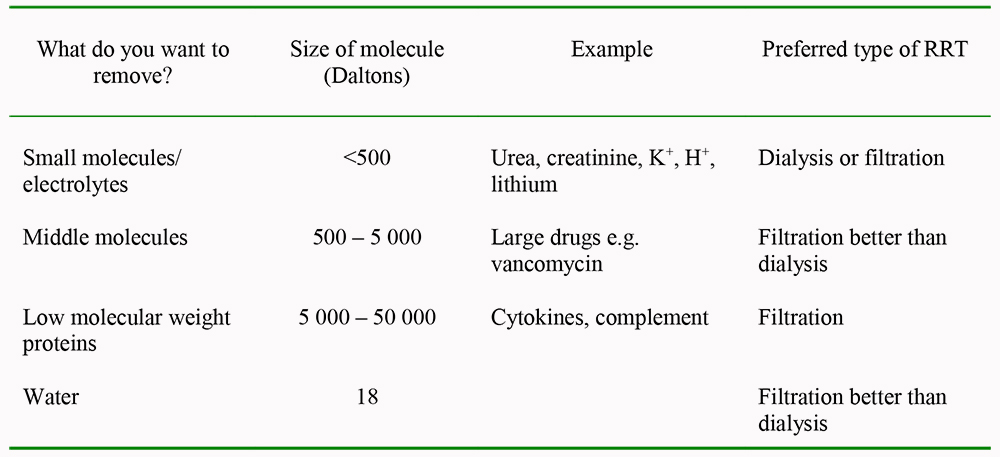
Table 2. How the choice of RRT can be determined by the aim of treatment
2. The patient`s cardiovascular status
- CRRT causes less rapid fluid shifts and is the preferred option if there is any degree of cardiovascular instability.
3. The availability of resources
- CRRT is more labour intensive and more expensive than IHD
- Availability of equipment may dictate the form of RRT.
4. The clinician`s experience
- It is wise to use a form of RRT that is familiar to all the staff involved.
5. Other specific clinical considerations
- Convective modes of RRT may be beneficial if the patient has septic shock
- CRRT can aid feeding regimes by improving fluid management
- CRRT may be associated with better cerebral perfusion in patients with an acute brain injury or fulminant hepatic failure.11
OPTIMAL FLOW RATES / DOSE OF RRT
When prescribing RRT one is often asked to define a flow rate. The flow rate refers to the ultrafiltrate produced by the filtration process as well as any effluent dialysis flow. The flow rate is a marker of solute clearance so it can simplistically be thought of as the dose of RRT.
Two recent randomised controlled trials have concluded that there is no benefit to increasing the flow rate from 20 to 35ml.kg.-1hr-1:
- The Randomised Evaluation of Normal versus Augmented Level of renal replacement therapy in ICU (RENAL) study randomised 1400 critically ill patients with AKI to intensive (35ml.kg. -1hr-1) or non-intensive (20ml.kg.-1hr-1) CRRT and no difference in mortality was seen in the two groups at 90 days.12
- The Acute Renal Failure Trial Network (ATN) study compared intensive or less-intensive dosing strategies for patients undergoing CRRT (35ml.kg.-1hr-1 versus 20ml.kg.-1hr-1), IHD (daily versus alternate days) and SLED. The recovery of renal function and the mortality at 60 days were the same in both arms of the trial but there were more hypotensive episodes in the intensive group.13
As mentioned earlier in this tutorial, high volume hemofiltration may be of benefit in patients with septic shock so there is currently a trend to increase flow rates in patients with septic shock and an AKI. This area of practice is being looked at by the IVOIRE (Impact of High-volume Venovenous Continuous Hemofiltration in the Early Management of Septic Shock Patients With Acute Renal Failure) study. This is a multicentre randomised controlled trial that is currently recruiting patients. Patients who have septic shock along with an AKI are being randomised to receive CVVH at 70ml.kg. -1hr-1or 35ml.kg.-1hr-1. The primary end point is all-cause mortality at 28 days.
PRACTICAL ISSUES
Vascular access
Venovenous RRT requires a double-lumen vascular catheter placed in a central vein. The tip should be sited in the inferior vena cava for femoral lines or superior vena cava (1-2cm from right atrium) for internal jugular and subclavian lines. The catheters are usually made of polyurethane or silicone and need to be stiff enough to prevent collapse under high negative pressures but soft enough to prevent kinking. The lumens can be arranged in various fashions but assuming each lumen is at least 11 French Gauge there is no evidence that one design is superior to the others.
Good flow through the intravenous catheter is crucial to prevent stasis of blood in the circuit and clotting of the filter. There are a number of things to take into account when choosing the site of the vascular access. [Please see Table 3]
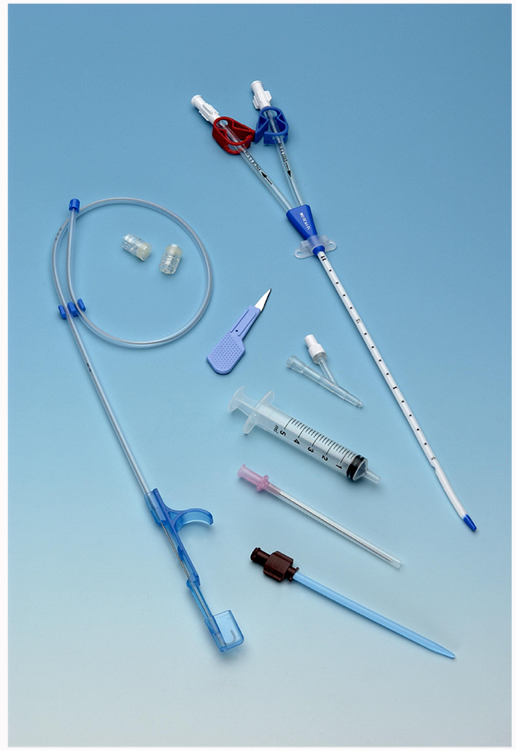
Figure 2. Typical contents of a dual lumen venous catheter insertion pack (Courtesy of Rheomed)
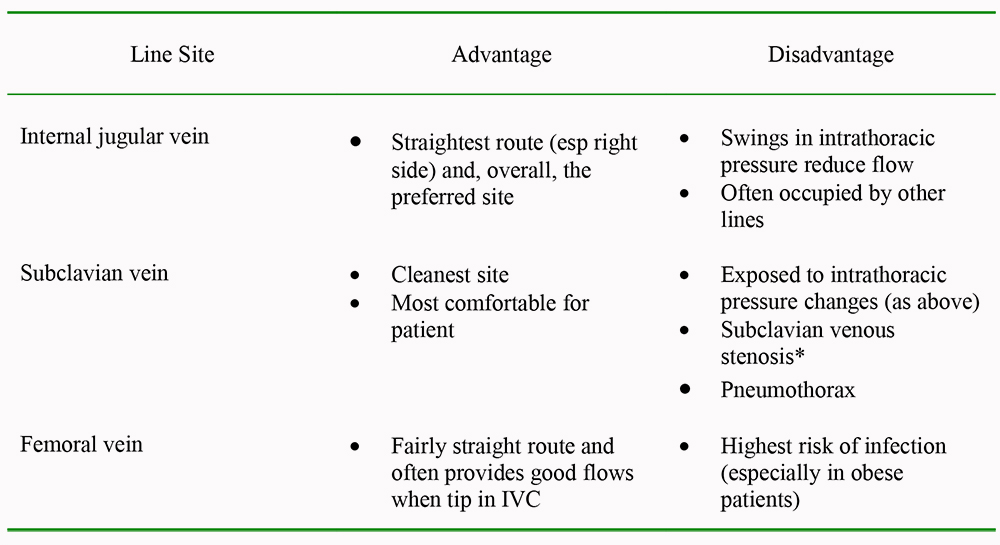
Table 3. Considerations when choosing where to site the central venous catheter
* There is a significant chance of subclavian venous stenosis after large bore venous catheter insertion which is problematic if an arteriovenous fistula is subsequently required for long term dialysis. However, mortality of patients on CRRT is high and those who survive are not usually dialysis dependant.
Extracorporeal circuits
Most CRRT techniques utilise a pump driven venovenous circuit as this provides a high constant flow rate. Arteriovenous techniques are described and were used historically, but are associated with catheter associated complications and are less reliable.
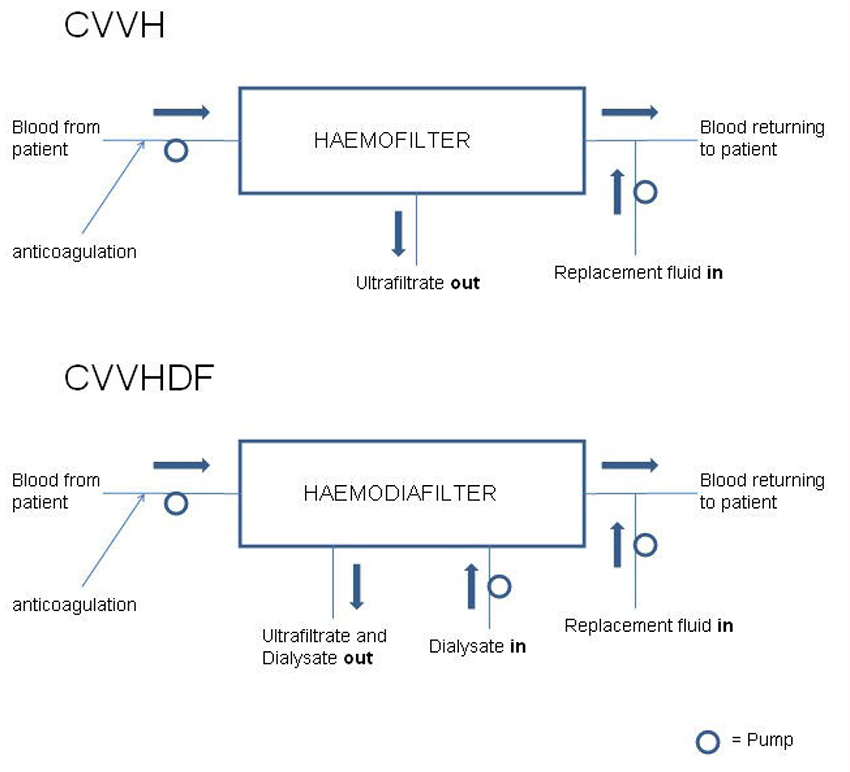
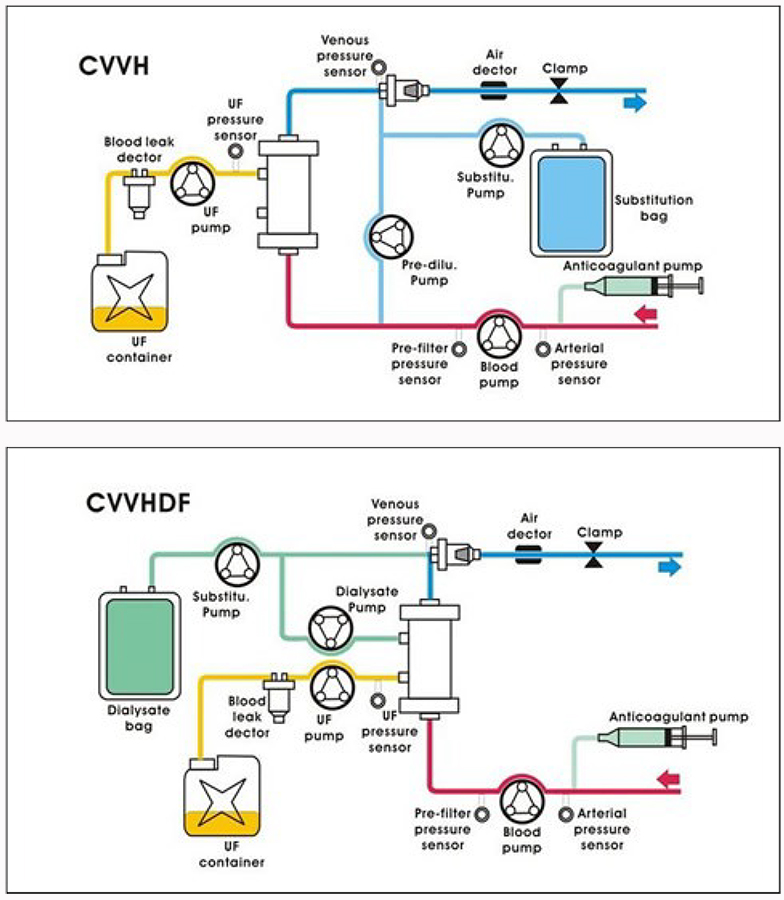
Figure 3. Schematic representation of CVVH and CVVHDF circuits
More detailed diagrams of the circuits used for CVVD and CVVHDF are include in the Appendix at the end of this tutorial
Anticoagulation
All modes of RRT that utilise an extracorporeal circuit will activate coagulation pathways and the premature clotting off of a filter is a common problem. Even a small amount of clot formation will reduce filter performance but if a filter clots off completely the blood contained in the circuit is lost and there an interruption in treatment while a new circuit is prepared.
Clot formation in the filter will trigger the transmembrane pressure alarm whereas clot in the venous catheter will trigger the access pressure alarm. Kinking of the catheter or a collapsing vein can also be responsible for triggering the access pressure alarm.
Non-pharmacological measures that can be taken to reduce clot formation include ensuring the patient has an adequate central venous pressure, optimising vascular access and adding a proportion of the replacement fluid to the patients blood before it passes through the haemofilter (this is predilution). Guidelines published in 2009 by Intensive Care Society suggest that anticoagulation is NOT required when:14
- There is a already a degree of coagulopathy
- INR > 2-2.5
- APTT > 60 seconds
- platelet count < 60 x 103 .mm-3
- There is a high risk of bleeding
- The patient is receiving activated protein C.
Anticoagulation should be considered in all other situations and the aim is to anticoagulate the filter and not the patient. In practice, this can be more difficult than it sounds. The forms of anticoagulation available are:
Unfractionated or low molecular weight heparins
Unfractionated heparin (UFH) [5-30kDa] is the most commonly used anticoagulant in the UK and a typical regime involves a 40-70 IU.kg-1 bolus followed by a pre-filter infusion at 5-10 IU.kg.-1hr-1. It is the most cost effective anticoagulant and is fully reversible with protamine. The APTT should be monitored to avoid excessive anti-coagulation but there is no evidence that elevating the APTT prolongs filter life.
Low molecular weight heparins (LMWH) [4.5-6kDa] are only used for RRT in 4% of intensive care units in the UK.15 They are dependant on renal elimination so in this setting their dosing needs to be guided by anti-factor Xa levels (aiming for 0.25-0.35 IU.ml-1). The half life of LMWHs is longer than for UFH (2-6 hrs versus 1.5-3hrs) and their effect can only be partially reversed with protamine. There is not a huge amount of data on the use of LMWH in CRRT and there is no evidence to suggest that they are superior to UFH.16
Prostaglandins
Prostaglandins (prostacyclin or prostaglandin E2) inhibit platelet function and can either be used on their own or in combination with heparin whereby they have a synergistic effect. Prostaglandins have a short half life (several minutes) so are administered as an infusion (2.5 – 10 ng.kg1 .min-1). The anticoagulant effect stops within 2 hours of discontinuing the infusion making them a useful alternative to heparin in patients at high risk of bleeding. The main side effect is vasodilation which may include a reduction in hypoxic pulmonary vasoconstriction leading to hypoxemia. The other downside is that they are expensive and so are only used as second line therapy
Regional citrate anticoagulation
Regional citrate anticoagulation is an effective therapy especially when there is an increased risk of bleeding. It is often used as an alternative to heparin in the USA but it is rarely used in the UK.
Sodium citrate is infused into the circuit pre-filter which chelates calcium and inhibits clot formation. The calcium citrate complex is freely filtered so a calcium infusion is required post-filter. This form of anticoagulation is limited by the metabolic derangements that it can cause: Hypocalcaemia, hypomagnesaemia (Mg2+ is also chelated), hypernatraemia (sodium load in sodium citrate), metabolic alkalosis (citrate is metabolised to bicarbonate), metabolic acidosis (caused by the citrate especially if the body`s citrate handling is impaired e.g. liver failure).
Others
There is no evidence to suggest newer heparin alternatives such as danaparoid, hirudin, fondaparinux or argatroban are better than UFH/LMWHs.
Filters
The properties of a filter that have an impact on its function are:
- Biocompatibility – The degree to which the membrane will activate the patient`s inflammatory and coagulation pathways. The greater the biocompatibility of a membrane the less activation it will cause.
- Flux – The permeability of the filter. High flux membranes are hydrophobic and may have more or larger pores allowing more water and solute to move across the membrane.
- Adsorption – The ability of larger solutes to adhere to the surface of the membrane. A highly adsorptive membrane offers the potential benefit of adsorbing midsized molecules including inflammatory mediators but only until it is saturated with them (usually after the first few hours).
- Thickness – Thinner membranes allow greater movement of solute by diffusion and also favour convective movement
- Surface area – The surface area of the membrane determines the available area for diffusion and ultrafiltration
Filters are either cellulose-based or synthetic. Synthetic filters such as polysulphone and polyamide are more biocompatible and are higher-flux membranes so seem more suitable for CRRT, however, there is no conclusive evidence that they improve outcome.
In practice, most filters used for CRRT are synthetic, high-flux membranes with a surface area of 0.6–1.2m2 and a pore size allowing the passage of molecules up to 50,000 Daltons.
Replacement fluid
Replacement fluids vary slightly in their composition but are all are balanced salt solutions with either a lactate or bicarbonate buffer.
Lactate-based solutions are stable and hence the cheaper and more practical option, however, their buffering capacity depends on the conversion of lactate into bicarbonate. Under normal physiological conditions the body converts lactate into bicarbonate on an equimolar basis. This is not always the case in critically ill patients, particularly if they have impaired liver function or already have a lactic acidosis. In these situations, RRT using a lactate-based replacement fluid can worsen the patient`s acidosis so a bicarbonate-based replacement solution should be used. If, however, this is not possible and serum lactate levels are not excessive then an alternative option is to continue with the lactate based replacement solution and commence an intravenous infusion of bicarbonate.
Bicarbonate-based replacement solutions have a more reliable buffering capacity but need to be prepared just prior to use. At present, there is no evidence to suggest that the choice of replacement fluid has an impact on survival or renal recovery.
Replacement fluid can be added pre- or post-filter in varying ratios. The benefit of adding some of the replacement fluid pre-filter is that it lowers the haematocrit of the blood which reduces the likelihood of the filter clotting. The downside is that pre-dilution reduces solute clearance and a compensatory increase in flow rates should be considered. (15% for ultrafiltration rates of 2l.h-1 and up to 40% for rates of 4.5l.-1)
Pharmacokinetics while on RRT
Some say that while a patient is receiving RRT drugs should be dosed as if the GFR is 10-50 ml.min -1 but, unfortunately, it is probably not as simple as that since there are numerous variables. The most reliable guide to dosing is by measuring drug levels but this is not usually a feasible option so referring to the drug manufacturer’s recommendations is a reasonable place to start.
The factors that affect the pharmacokinetics while on RRT are:
- Protein binding – Drugs that are highly protein bound (e.g. warfarin, diazepam, propranolol and phenytoin) are only cleared by RRT in small amounts. However, as the patients protein levels fall the free fraction of the drug increases and so does its clearance.
- Size of drug molecule and mode of RRT – Small molecules (<500 Daltons) are cleared by all (convective versus diffusive) types of RRT but as molecule size increases diffusion becomes less effective.
- Timing of RRT – Drugs given between sessions of IHD or SLED (intermittent versus continuous) will not be cleared until the subsequent session.
- Dose of RRT – Reduced flow rates/ shorter sessions will reduce clearance of drugs.
- Membrane permeability – The high-flux haemofilter membranes used in CRRT are permeable to most non-protein-bound drugs.
- The patient’s residual GFR – Also needs to be taken into consideration.
Prescription of RRT
A typical prescription for a 75kg patient requiring CRRT for an AKI would be as follows:
Anticoagulation:
- Unfractionated Heparin: 5,000 IU bolus followed by a pre-filter infusion at 500 IU.hr.-1
- Aim to anticoagulate filter but ensure APTTR<2
Fluid balance over 24 hours:
- Aim for an even balance if the patient is euvolaemic
- Aim for the appropriate negative balance if the patient is fluid overloaded (<1500ml/24hrs)
Type of Replacement fluid/Dialysate:
- Use solutions without potassium if serum potassium is high but switch to potassium containing solutions as serum potassium normalises.
- Use a bicarbonate-based buffer rather than a lactate-based buffer if there are concerns about lactate metabolism or if serum lactate>8mmol.l.-1 [Note- An intravenous bicarbonate infusion may be required if a lactate-based buffer is used]
Exchange rate/treatment dose:
- 1500ml.hr.-1 (75kg x 20ml.kg.-1hr-1)
The treatment dose is usually prescribed as an hourly “exchange rate” which is the desired hourly flow rate adjusted for the patient`s weight. In the case of CVVH, the exchange rate simply represents the ultrafiltration rate whereas in CVVHDF it represents a combination of the ultrafiltration rate and the dialysate flow rate. In CVVHDF, the ratio of ultrafiltration to dialysate flow is often set at 1:1 but it can be altered to put the emphasis on either the dialysis or filtration component. [Please see Table 4].
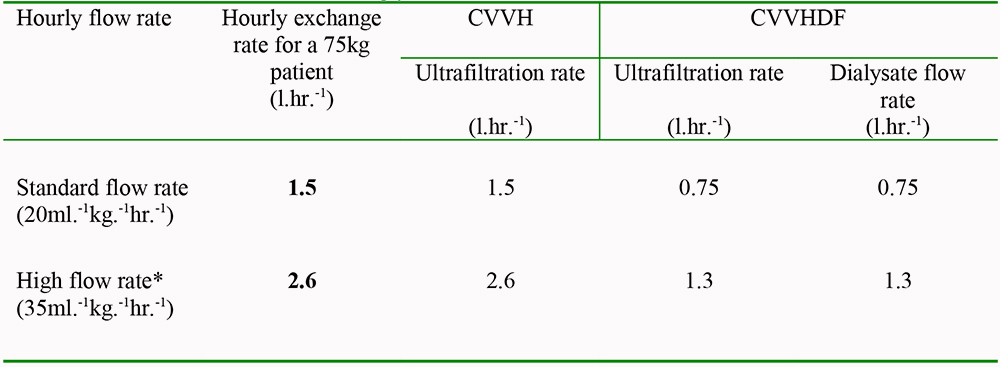
Table 4. CRRT flow rates for a 75kg patient with an AKI.
*Consider high flow rates in patients with septic shock and AKI
COMPLICATIONS
Complications common to IHD, CRRT, and hybrid therapies include:
- Complications related to the vascath (including line-related sepsis)
- Haemodynamic instability
- Air emboli
- Platelet consumption
- Blood loss
- Electrolyte imbalances
- Hypothermia
- Effects of anticoagulation (bleeding or specific side-effects of the anticoagulant used e.g. heparin induced thrombocytopenia).
PROGNOSIS OF PATIENTS WITH AKI ON RRT
Bagshaw et al looked at the outcomes in 240 patients with ARF requiring RRT and showed that although the mortality rate was high (around 60%) the majority of survivors (78%) were free from RRT at one year. Of those requiring chronic RRT, 63% had pre-existing chronic renal impairment with a median pre-admission creatinine of 232mcmol.l-1.4
OVERVIEW/SUMMARY
- AKI is common and 5% of the critical care population receive RRT.
- There are various forms of RRT but they all remove unwanted solutes using the processes of diffusion (dialysis) and/or convection (filtration). RRT can be administered continuously or intermittently.
- No single form of RRT has been shown to offer a survival benefit over the others but there are often other reasons why a particular technique may be preferable in a given situation.
- There is some evidence that high volume haemofiltration may improve survival in patients with septic shock but there have been no large randomised controlled trials in this area.
- Lifespan of the circuit is dependant on good quality vascular access and appropriate anticoagulation.
- Pharmacokinetics while on RRT needs to be looked at on an individual basis but there are some general rules such as drugs which are protein bound are not easily removed.
- 60% of people receiving RRT for AKI will die during that admission but 80% of the survivors will be free from RRT one year later.
ANSWERS TO QUESTIONS
- TFTT
- FFFT
- TFTT
GLOSSARY OF USEFUL PHYSIOLOGICAL TERMS
Convection – Solute transport across a membrane together with a solvent (usually water) in response to a pressure gradient across the membrane.
Dalton – A unit of mass used to express atomic and molecular masses. It is the approximate mass of a hydrogen atom, a proton, or a neutron.
Diffusion – Solute transport from a compartment with high concentration to a compartment with low concentration.
Fick`s Law of diffusion – States that the rate of diffusion across a membrane is proportional to the concentration gradient across that membrane
Hydrostatic pressure – The pressure exerted by a fluid at equilibrium at a given point within the fluid, due to the force of gravity. In the context of haemofiltration, this pressure is created by the rollerball pump system of the extracorporeal circuit.
Semipermeable membrane – A barrier, either cellulose or synthetic, that allows water, electrolytes and other molecules to pass through while cellular components and larger molecules are held on one side.
Sepsis – The diagnosis of “Sepsis” requires the presence of proven infection with two or more features of the Systemic Inflammatory Response Syndrome (SIRS):
- Temperature > 38 or < 36°C
- Respiratory rate > 20
- Heart Rate > 90
- White Blood Cell count < 4 or > 12.
Severe Sepsis – Sepsis with evidence of organ dysfunction
Septic Shock – Severe Sepsis with hypotension that is refractory to fluid resuscitation.
Ultrafiltrate – Plasma water and solutes that pass through the semipermeable membrane.
Ultrafiltration – Transport of water across a membrane by a pressure gradient. In haemofiltration, it is the process by which plasma water and filterable solutes are separated from whole blood.
REFERENCES
- Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, Levin A. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 2007; 11: R31.
- Ostermann M, Chang RW: Acute kidney injury in the intensive care unit according to RIFLE. Crit Care Med 2007; 35: 1837–43.
- Uchino S, Kellum JA, Bellomo R, et al. Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA 2005; 294: 813–8.
- Bagshaw SM, Laupland KB, Doig CJ, Mortis G, Fick GH, Mucenski M, Godinez-Luna T, Svenson LW, Rosenal T. Prognosis for long-term survival and renal recovery in critically ill patients with severe acute renal failure: a population-based study. Crit Care 2005; 9: 700-9.
- Liu, KD, Himmelfarb, J, Paganini, E, et al. Timing of initiation of dialysis in critically ill patients with acute kidney injury. Clin J Am Soc Nephrol 2006; 1: 915-9.
- Honore PM, Jamez J, Wauthier M et al. Prospective evaluation of short-term, high volume isovolemic haemofiltration on the haemodynamic course and outcome in patients with intractable circulatory failure resulting from septic shock. Crit Care Med 2000; 28: 3581-87.
- Joannes-Boyau O, Rapaport S, Bazin R et al. Impact of high volume haemofiltration on haemodynamic disturbance and outcome during septic shock. ASAIO J 2004; 50: 102-9.
- Ratanarat R, Brendolan A, Piccinni P et al. Pulse high-volume haemofiltration in critically ill patients: Effects on hemodynamics and survival. Crit Care 2005; 9: 294-302.
- Cornejo R, Downey P, Castro R et al. High-volume haemofiltration as salvage therapy in severe hyperdynamic septic shock. Intensive Care Med 2006; 32: 713-22.
- Vinsonneau, C, Camus, C, Combes, A, et al. Continuous venovenous hemodiafiltration versus intermittent hemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre, randomized trial. Lancet 2006; 368: 379-85.
- Davenport A, Will EJ, Davison AM. Continuous Vs intermittent forms of haemofiltration and/or dialysis in the management of acute renal failure in patients with defective autoregulation at risk of cerebral oedema. Contrib Nephrol 1991; 93: 225-33.
- Bellomo R, Cass A, Cole L, Finfer S, Gallagher M, Lo S, McArthur C, McGuinness S, Myburgh J, Norton R, Scheinkestel C, Su S. Intensity of continuous renal-replacement therapy in critically ill patients (RENAL study). N Engl J Med 2009; 361: 1627-38.
- The VA/NIH Acute Renal Failure Trial Network. Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med 2008; 359: 7-20.
- J.J. Gatward, G.J. Gibbon, G. Wrathall and A. Padkin. Standards and Recommendations for the Provision of Renal Replacement Therapy on Intensive Care Units in the United Kingdom. Intensive Care Society (2009). Website: ics.ac.uk
- Renal replacement therapy for acute renal failure: a survey of practice in adult intensive care units in the United Kingdom. Anaesthesia 2008; 63: 959-66.
- Oudemans-van Straaten HM, Wester JPJ, de Pont AC et al. Anticoagulation strategies in continuous renal replacement therapy: can the choice be evidence based? Intensive Care Med 2006; 32: 188-202.
Appendix
The arrangement of a haemofiltration and a haemodiafiltration circuit (available at: www.smilegroup.com.tw/t2-2.asp)