General Topics
QUESTION
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Which of the following drugs confers a proven survival benefit when used in cardiac arrest?
a. Adrenaline
b. Amiodarone
c. Atropine
d. Lignocaine
e. None of the above - What is the recommended rate and depth of chest compressions, and recommended ratio of compressions to ventilations in adult Basic Life Support?
- All of the following interventions confer a survival benefit in cardiac arrest EXCEPT:
a. Early defibrillation for VF pulseless VT arrest
b. Early, good quality chest compressions with minimal interruptions
c. Early intubation and positive pressure ventilation
d. Avoidance of both hypoxia and hyperoxia following return of spontaneous circulation (ROSC)
e. Therapeutic hypothermia following ROSC after VF or pulseless VT arrest
INTRODUCTION
Cardiac arrest is the most time-critical medical emergency an anaesthetist may face. Effective basic and advanced life support measures must be applied as early as possible to maximise the chance of survival and minimise hypoxic neurological damage. The International Liaison Committee on Resuscitation (ILCOR) has produced regular consensus guidelines on cardiac arrest management since 2000. The latest update was released in 2010 and has been adapted by various resuscitation advisory bodies worldwide1, 2, 3. This tutorial outlines the consensus algorithms for advanced cardiac life support with emphasis on the most recent recommendations as outlined by the Australian Resuscitation Council and New Zealand Resuscitation Council1
SUMMARY OF BASIC AND ADVANCED LIFE SUPPORT GUIDELINES
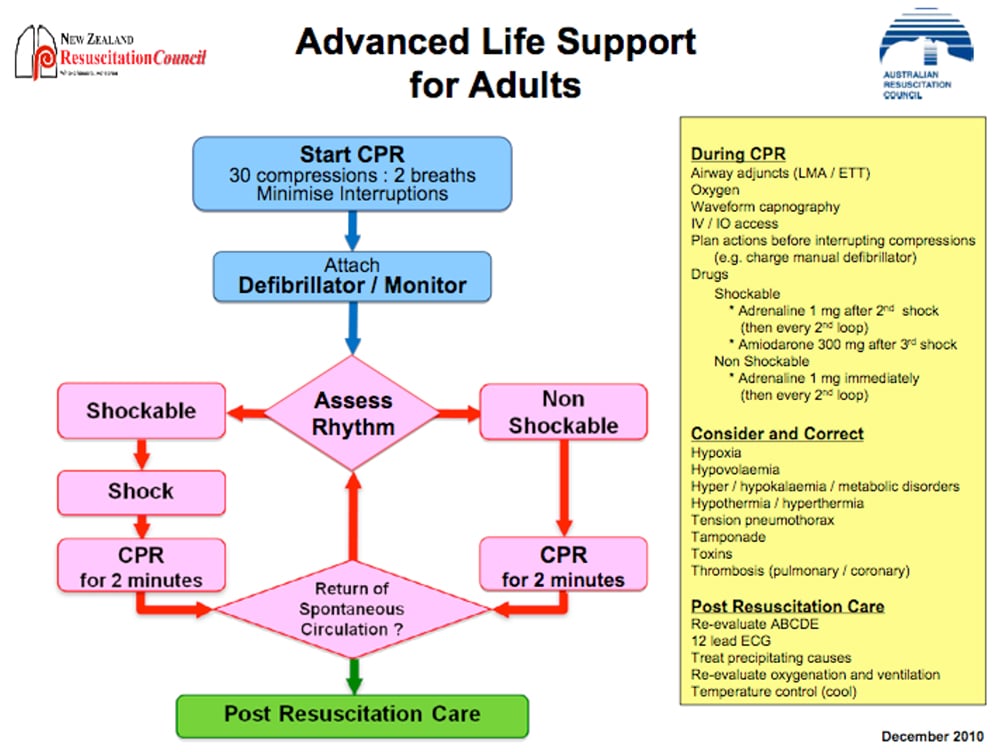
Figure 1: Australian Resuscitation Council (ARC) flowchart for adult ALS
Basic Life Support
Basic life support is commenced when a patient is found to be unresponsive with absent or abnormal breathing (such as agonal gasps). The absence of a palpable pulse is now regarded as an unreliable sign and should not be the only sign used to establish cardiac arrest. If trained to do so, rescuers may attempt to palpate a pulse for up to 10 seconds.
Current guidelines emphasise the quality of chest compressions. The chest should be compressed at least 5 cm and at a rate of approximately 100/min. The chest should be allowed to fully recoil between compressions. Interruptions to chest compressions should be minimised.
Rescuer fatigue is associated with worsening rate and depth of chest compression even before the rescuer is aware of fatigue. Where feasible, to avoid fatigue, rescuers should rotate chest compression duties. Rotation at 2 minute intervals, regardless of whether the rescuer feels tired, is optimal and can be integrated with the intervals for rhythm analysis during advanced life support.
Chest compressions and ventilations are delivered at a ratio of 30 chest compressions : 2 ventilations before the airway is secured, regardless of the number of rescuers (previous guidelines recommended a ratio of 15:2 when a second rescuer was available). This maximises the number of compressions given and minimises interruptions, which in turn maximised perfusion.
Advanced Life Support
Once effective cardiopulmonary resuscitation (CPR) is established (30 chest compressions : 2 breaths), a defibrillator is attached and the cardiac rhythm is analysed. Subsequent treatment is determined by whether there is a “shockable” rhythm i.e. VF or VT or a “non-shockable” rhythm i.e. pulseless electrical activity (PEA) or asystole.
Shockable rhythm (VF or VT)
A single DC shock is delivered and CPR is immediately recommenced, without a pause to reassess the rhythm. CPR is continued, interrupted only every 2 minutes for a rhythm check. A further single DC shock is delivered if the rhythm remains VF or VT at each rhythm check. Adrenaline 1 mg IV is administered after the second shock and then every second loop of the ALS algorithm i.e. every second rhythm check or approximately every 3-5 minutes. Amiodarone 300 mg IV is administered after the third DC shock if VF/VT persists.
Non-Shockable rhythm (PEA or asystole)
Adrenaline 1 mg IV is given immediately. Effective CPR is continued, with a pause to reassess the rhythm every 2 minutes. Adrenaline 1 mg IV is given every second loop of the algorithm i.e. every 3-5 minutes.
During CPR
An advanced airway (endotracheal tube or laryngeal mask airway) is placed. Once placed, ventilations are delivered at a rate of 6-10 per minute, with no interruptions to CPR for delivery of breaths. The volume delivered should be sufficient for visible chest rise.
100% oxygen is delivered.
Intravenous access is established for the delivery of medications. If this is impossible, intraosseous access is established.
If available, waveform capnography is used to confirm correct advanced airway placement, guide effectiveness of CPR and detect return of spontaneous circulation (ROSC).
Reversible causes of the cardiac arrest are sought and treated. The mnemonic “Hs and Ts” is helpful. See Figure 2.

Figure 2: Mnemonic for reversible causes of cardiac arrest
Post-Resuscitation Care
Once ROSC is achieved, the phase of post-resuscitation care is commenced. The patient is re- evaluated. Adequate oxygenation and ventilation must be confirmed. Urgent 12-lead ECG and chest X-ray should be considered. The need for coronary reperfusion should be assessed. Reversible causes of the cardiac arrest should be sought and treated.
For patients who remain comatose after ROSC, therapeutic hypothermia should be considered.
MAJOR CHANGES IN THE 2010 RESUSCITATION GUIDELINES
Advanced airway
Reduced emphasis on endotracheal intubation
Endotracheal intubation remains the gold standard for establishment of a secure airway. One advantage of endotracheal intubation is that once achieved, it is possible to continue chest compressions without interruptions for ventilation. A significant disadvantage however, is that endotracheal intubation is usually associated with an undesirable, prolonged pause in chest compressions. Rescuers must weigh the benefits of intubation against the need to provide effective chest compressions.
It is recommended that attempts at intubation not interrupt chest compressions for more than 20 seconds.
Other supraglottic airway devices such as the laryngeal mask airway (LMA) may also be used if rescuers are adequately trained and skilled to do so. There is no evidence that demonstrates a survival advantage with the use of supraglottic airways, but studies suggest that successful insertion rates and insertion times are equal or superior to endotracheal intubation and they can be inserted without interruption to chest compressions. There are however, no data demonstrating the ability to provide effective ventilations without interruptions to chest compressions using a supraglottic airway device – the principle advantage of endotracheal intubation.
There is no evidence to support any one method of securing an airway during cardiac arrest. The choice of airway adjunct should be made on the basis of availability and the experience and skills of the rescuer.
It is reasonable to defer the use or insertion of airway adjuncts until after ROSC in order to avoid long pauses in chest compressions. Providing ventilations via a bag-valve-mask until after ROSC is an appropriate alternative.
Confirmation of endotracheal tube placement
The performance of clinical assessment in confirming correct tube placement (auscultation of the chest and epigastrium, condensation in the tube and chest rise) is variable: sensitivity 74-100%, specificity 66-100%4.
Secondary confirmation via qualitative end-tidal CO2 or oesophageal detection devices also perform variably and no better than clinical assessment. Colorimetric ETCO2 detectors perform poorly in cardiac arrest patients because pulmonary blood flow and therefore exhaled CO2 may be very low 4.
Waveform capnometry (ETCO2) has the best sensitivity and specificity and is recommended in addition to clinical assessment to confirm and continuously monitor correct tube placement.
Ventilation
Avoid hyperventilation
Hyperventilation is associated with increased intrathoracic pressure, decreased cerebral and coronary perfusion (hypocapnia causes cerebral and coronary vasoconstriction) and may impair ROSC. In order to achieve adequate oxygenation and optimise haemodynamics, once an advanced airway is established, the rate of manual ventilation should be 6-10 breaths per minute, and should not exceed 12 breaths per minute. As a guide, one ventilation can be given after every 15 chest compressions, delivering the ventilation during the relaxation phase of chest compression, without a pause (simultaneous ventilation and compression may impair coronary perfusion and adversely affect survival).
Specific guidelines are not given for optimal tidal volume. Ventilations should be adequate to produce visible chest rise. PaCO2 (but not ETCO2) via arterial blood gas analysis may allow individual titration of ventilation.
Mechanical ventilation
There is insufficient evidence to support or refute the use of automatic ventilators over manual bag-valve ventilation. A mechanical ventilator may enable rescuers to perform more tasks and removes the risk of inadvertent hyperventilation. There is however a risk of hypoventilation due to non-delivery of mechanical breaths.
Monitoring during resuscitation – ETCO2
Continuous waveform end-tidal CO2 (ETCO2) monitoring is a useful adjunct to monitor the effectiveness of chest compressions. Falling ETCO2 may indicate rescuer fatigue or inadequate depth or rate of compressions thus providing real-time feedback to the resuscitation team.
A sudden rise in ETCO2 may also be used for early detection of ROSC, during chest compressions, prior to the next pause for rhythm analysis.
Defibrillation – perform with minimal interruptions to chest compressions
Biphasic (rather than monophasic) defibrillators are currently recommended. The use of adhesive pads is preferred over paddles. The default energy setting should be 200J for all shocks in adults (360J if monophasic).
A single shock strategy is recommended over previously suggested stacked shocks. Single shocks are associated with shorter interruptions to chest compressions.
Immediately after defibrillation, there should not be a pause for rhythm analysis. Chest compressions should immediately be recommenced after shock delivery and continue for two minutes prior to rhythm analysis. So defibrillate first, then perform compressions, then check the rhythm.
The defibrillator should be charged during chest compressions, allowing immediate defibrillation, without pause for charging, once VT or VF is detected at rhythm analysis.
Chest compressions during defibrillation are not currently recommended due to safety concerns, although this technique may be feasible5.
Medications in Cardiac Arrest
Intraosseous administration
In previous guidelines, endotracheal administration of drugs was recommended where intravenous access was unavailable. Currently, the intraosseous route is preferred as it is safe and effective. Administration of drugs via this method should achieve adequate serum concentrations, whereas absorption via the endotracheal route is variable and serum concentrations are generally lower.
Medication and long term outcome
Despite widespread use, there is no evidence that any medication improves the rate of neurologically intact survival from cardiac arrest.
Adrenaline
Adrenaline remains the vasopressor of choice – it may improve likelihood of ROSC and short-term survival. There is no evidence to define the optimal dose or timing of adrenaline, and the role of adrenaline in resuscitation protocols is based on consensus opinion.
Amiodarone
No studies have shown a long-term survival benefit from amiodarone. However amiodarone may improve rates of survival to hospital admission in out-of-hospital cardiac arrest and may improve the response to defibrillation in shock-refractory VT or VF.
Atropine
Atropine is no longer recommended for use in PEA or asystolic cardiac arrest.
Ultrasound during cardiac arrest
If a trained operator and equipment is available, bedside ultrasound may be a useful tool to help diagnose reversible cardiac and non-cardiac causes of cardiac arrest eg. pericardial effusion and tamponade, right ventricular dysfunction due to massive pulmonary embolism, absence of lung artifact in tension pneumothorax.
Ultrasound may also be used to detect cardiac standstill, the presence of which is highly predictive of death and indicates that further resuscitative efforts are futile.
Post-resuscitation care
Aggressive therapeutic efforts should not cease after ROSC and are likely to significantly influence the patient’s long term outcome. Reversible causes of the arrest must again be evaluated (see Table 2) and treated.
Oxygenation
Adequate oxygenation should be maintained in the post-resuscitation phase, preventing hypoxaemia. However, new evidence suggests that exposure to excessive oxygen (hyperoxaemia) may also be detrimental6. Oxygenation may be monitored either by pulse oximetry or by arterial blood gas andinspired oxygen should be titrated to SaO2 94% – 98%. No PaO2 target is recommended in the guidelines.
Temperature control
Therapeutic hypothermia, in randomised trials7,8 has shown a benefit in terms of intact long-term neurological survival in comatose victims of out-of-hospital VF or VT cardiac arrest. Current guidelines recommend cooling to 32 – 34 degrees C for 12 – 24 hours. Therapeutic hypothermia may also be of benefit after non-shockable rhythms and in-hospital arrests but level I evidence is lacking.
Hypothermia can be induced by rapid infusion of 30mL/kg of cold (4 degrees C) fluid (either 0.9% saline or Hartmann’s solution). Alternatively, ice packs placed against the patient’s neck, axillae and groin are effective. Maintenance of hypothermia can be safely achieved with continued use of ice packs. Shivering, which counteracts lowering of temperature should be suppressed by neuromuscular blocking drugs.
SUMMARY
- The cornerstone of resuscitation is effective chest compressions with minimal interruptions
- compress at 100 per minute
- compress to at least 5 cm depth
- 30 compressions : 2 ventilations
- rotate rescuers to avoid fatigue
- Consider alternatives to endotracheal intubation in order to minimise interruption to chest compression
- Avoid hyperventilation
- Consider End-Tidal CO2 monitoring during resuscitation
- Defibrillation
- single-shock strategy
- charge the defibrillator during chest compressions
- minimise interruptions to chest compressions
- Obtain intraosseous access where intravenous access is impossible
- Adrenaline and amiodarone remain the only routine medications. Atropine is no longer recommended
- Consider bedside ultrasound to assess for reversible causes and to detect cardiac standstill
- Post-resuscitation care
- avoid hypoxaemia and hyperoxaemia institute therapeutic hypothermia where indicated
ANSWERS TO QUESTIONS
- Which of the following drugs confers a proven survival benefit when used in cardiac arrest?
a. adrenaline
b. amiodarone
c. atropine
d. lignocaine
e. none of the above - What is the recommended rate and depth of chest compressions, and recommended ratio of compressions to ventilations in adult Basic Life Support?
100/min, at least 5 cm, 30:2 - All of the following interventions confer a survival benefit in cardiac arrest EXCEPT:
a. early defibrillation for VF or pulseless VT arrest
b. early, good quality chest compressions with minimal interruptions
c. early intubation and positive pressure ventilation
d. avoidance of both hypoxia and hyperoxia following return of spontaneous circulation (ROSC)
e. therapeutic hypothermia following ROSC after VF or pulseless VT arrest
REFERENCES and FURTHER READING
- The Australian Resuscitation Council Online: http://www.resus.org.au/
- American Heart Association: http://guidelines.ecc.org/2010-guidelines-for-cpr.html
- European Resuscitation Council: http://www.cprguidelines.eu/2010/
- Jerry P. Nolan, Jasmeet Soar, David A. Zideman, Dominique Biarent, Leo L. Bossaert, Charles Deakin, et al. European Resuscitation Council Guidelines for Resuscitation 2010 Section 1. Executive summary. Resuscitation 81 (2010) 1219–1276
- Lloyd MS, Heeke B, Walter PF, Langberg JJ. Hands-on defibrillation: an analysis of electrical current flow through rescuers in direct contact with patients during biphasic external defibrillation. Circulation. 2008 May 13; 117(19): 2510-4.
- J. Hope Kilgannon, Alan E. Jones, Nathan I. Shapiro, Mark G Angelos, Barry Milcarek, Krystal Hunter, et al. Association Between Arterial Hyperoxia Following Resuscitation From Cardiac Arrest and In-Hospital Mortality, JAMA 2010; 303(21): 2165-2171
- Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med 2002;346(8):557-63
- Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med 2002;346(8):549-5



