Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article.
- Where does the Coronary Sinus empty?
a. In the inter-atrial septum
b. In the right atrium
c. In the right ventricle
d. In the left atrium - What is the right ventricular systolic pressure in a normal subject?
a. 90-140 mmHg
b. 60-100 mmHg
c. 15-30 mmHg
d. 0-8 mmHg - What is the hangout interval?
a. presence of forward flow despite a negative ventricular-arterial pressure gradient
b. the pause in ventricular contraction
c. the time between end systole and closure of the pulmonary valve
d. backward flow occurring before pulmonary valve closure
INTRODUCTION
In most physiology textbooks and tutorials the right heart is seen as a recipient of the venous return and a “passive” conduit to the lung circulation. Despite its low pressures it plays a key role in cardiovascular system (CVS) homeostasis and the management of the right heart failure is a great challenge to current medical practice.
ANATOMY
Right Atrium (RA)
The right atrium is an irregular thin walled chamber. The anterior wall hosts the right atrial appendage, the left wall is shared with the left atrium and called the interatrial septum, and this traverses an indentation called the fossa ovalis, a sign of the of the pre-natal foramen ovale. The right wall is the free wall and it runs parallel to the sternum, and the tricuspid annulus and valve forms the inferior wall.
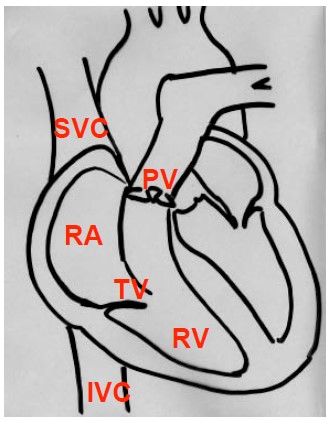
Figure 1: The heart
A further 3 openings can be identified into the ovoid shaped atrial chamber:
- the superior vena cava (SVC) which enters the atrium in the right anterior portion of the superior wall
- the inferior vena cava (IVC) which enters into the right posterior portion of the inferior wall
- the coronary sinus (CS), which collects all the venous return from the heart, empties into the infero-posterior portion of the right atrium, just above the tricuspid annulus (Fig 1)
The Sino Atrial Node (SAN), which is our natural pacemaker, sits at the junction between the SVC and the RA, and the Atrio- Ventricular Node (AVN) is adjacent to the CS.
Tricuspid Valve (TV)
It is the largest among the cardiac valves and has three thin leaflets: anterior, posterior, and septal each one associated to a papillary muscle. The septal leaflet of the Tricuspid Valve shares the fibrous skeleton of the heart with the Mitral Valve.
Right Ventricle (RV)
The right ventricle is an asymmetric chamber which resembles a crescent shape and it is wrapped around the right and antero-septal side of the left ventricle (LV). It has thinner walls then the LV and in healthy individuals reaches two-thirds of LV dimensions. One apex of the crescent is occupied by the TV and the other is formed by the Pulmonary Valve (PV). The inner cavity can be divided into an inflow and outflow tracts, anatomically separated by the Moderator Band. The septal wall of the RV is divided into a membranous portion basally and a muscular portion apically.
Pulmonary Valve (PV)
The PV separates the RV outflow tract from the Pulmonary Artery (PA). It is a semilunar valve, with three thin leaflets and it closely resembles the anatomical structure and the function of the Aortic Valve. Its function is to prevent the retrograde flow from the PA, but due to the lower pressure of the right circulation, its structure is thinner than the Aortic Valve.
Right Heart circulation
The main arteries perfusing the heart are:
- Left Anterior Descending (LAD)
- Right coronary Artery (RCA)
- Circumflex Artery (Cx).
The LAD and RCA both originate directly form the Aorta, just above the Aortic Valve (AV) and the Cx is a brunch of the LAD.
The majority of the RV is perfused by the Right Coronary Artery. After its aortic origin RCA travels in the atrioventricular groove, the groove between the RA and RV, and it wraps around it until it reaches the posterior wall and descends inferiorly to the cardiac apex. In the vast majority of the population (70 to 85%) perfusion of the posterior wall of the right ventricle is provided by the RCA, identifying what is called “right dominant perfusion”. In a smaller proportion (10%) the posterior brunches are supplied by the Cx (“left dominant perfusion”). In the remaining 10-15% the perfusion is provided by brunches of both arteries.
The RCA also perfuses the SAN and the AVN (the latter just in cases of right dominant perfusion). This clarifies the danger of a RCA occlusion, which can result in right cardiac failure, severe rhythm disturbance and even immediate death, as the SAN no longer has a blood supply.
The venous drainage of the right heart occurs via the Right Coronary Vein (also known as Small Cardiac Vein) which receives blood from the postero-inferior wall of the RV and RA, runs in the coronary sulcus between the RV and RA and opens into the CS or directly into the RA. Small accessory anterior veins can also be found in 20 to 45% of the population.
Septal abnormalities
Both the atrial and ventricular septum can face atypical embryonic evolution which results in septal defects. Several of these abnormalities are compatible with life and may not be diagnosed until adulthood.
Artrial Septal Defects(ASD) (Fig 2)

Figure 2: interatrial septum and fossa ovalis
ASD identifies a patency in the septum between the two Atria resulting in a circulatory shunt of variable gravity. The main defects are:
- Ostium primum
- Ostium secundum
- Sinus Venosus Defect
- Coronary Sinus Septal Defect
Ostium Primum accounts for 15% of all ASDs. It sits just above the TV but frequently extends apically into the ventricular septum, becoming an AVSD. Most AVSDs are relatively large defects and significant shunt leads to right heart dilatation.
Ostium secundum is the most frequent ASD (75%) and results from continued patency of the pre natal fossa ovalis.
Sinus Venosus Defect is an abnormality at the entrance of the SVC or ICV into the atrium.
Coronary Sinus Septal Defect implies and unroofed coronary sinus which empties into the LA producing a right to left shunt.
Ventricular Septal Defects(VSD)
VSD accounts for 30% of congenital cardiac abnormalities and the majority (60%) result in spontaneous closure. The defects are identified according to their location on the septal wall and they can be differentiated into:
- Membranosus
- Inlet
- Muscular
- Outlet
Similar to atrial-septal communications, the consequence of the VSD depend upon the dimensions of the defect and the consequent haemodynamic change. Large defects will lead to significant left-to-right shunting (as SVR > PVR) resulting in pulmonary hypertension and right heart dilatation to the extent that the right circulation may reach higher pressures than the systemic circulation. The subsequent haemodynamic failure depends not only on the size of the VSD, but also the relative resistances in both the systemic and pulmonary vascular beds. When the defect is small and restrictive, the flow across the ventricular septum is usually limited. There is little or no functional disturbance since pulmonary blood flow is increased only minimally. When the defect is large and nonrestrictive, (approximately 75% of the diameter of the aorta) shunting across the septum is dependent on the ratio of the pulmonary vascular resistance (PVR) to systemic vascular resistance (SVR). Initially, SVR exceeds PVR, so that left-to-right shunting predominates. With long standing large, nonrestrictive VSD, substantial pulmonary overcirculation leads to pulmonary hypertension. When PVR equals or exceeds SVR, the shunting of blood form left- to right ceases, and right to-left shunting occurs. Irreversible vascular changes can develop with prolonged shunts of this magnitude, leading to the development of pulmonary vascular obstructive disease or Eisenmenger’s syndrome
RIGHT HEART PHYSIOLOGY
The right ventricle is coupled in series with the left ventricle, however it has its own unique physiology largely dependent on the low impedance characteristics of the pulmonary vascular bed.
Under steady state the stroke volume of both left and right ventricle are approximately the same, but because the right ventricle ejects its volume into a low resistance system it requires only one-fifth of the energy needed for the same stroke volume to be ejected from the left ventricle. As a result the right ventricle has a smaller muscle mass and is therefore ‘weaker’ than the left ventricle. Due to this relative ‘weakness’ the right ventricle cannot adapt acutely to large changes in pulmonary vascular resistance (afterload) and this is the main cause of right heart failure. Conversely, the right ventricle has better compliance than the left ventricle allowing for large changes in venous return (preload). The right heart contracts with a characteristic “peristaltic” pattern whereby the inflow tract contract 20- 50ms before the outflow tract.
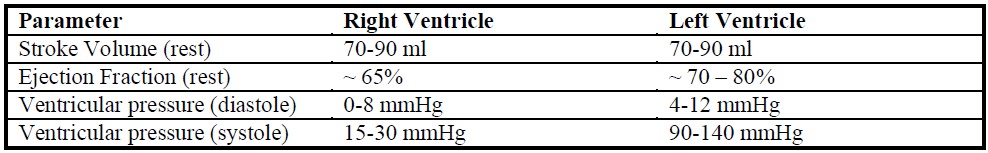
Table 1. Shows how a number of important parameters compare between the left and right ventricle (Ganong 1993)
The table clearly shows that right sided pressures are considerably lower than left sided pressures. Right ventricular pressure tracings therefore show an early peak and rapid decline in contrast to the more rounded left ventricular pressure tracing (Figure 3). Right ventricular isovolumetric contraction time is also shorter because right ventricular systolic pressure rapidly exceeds the low pulmonary artery diastolic pressure. The difference between the two ventricles can clearly be seen on pressure-volume loops (Figure 4). Unlike the rectangular left ventricular loop, the right ventricular loop is more trapezoidal with poorly defined isovolumetric periods. Uniquely, end systolic flow may even continue in the presence of a negative ventricular-arterial pressure gradient and pulmonary valve closure is known to occur well after the onset of right ventricular pressure decline. This is referred to as the “hangout interval” and is explained by continued momentum in the outflow tract. (Figures 3)
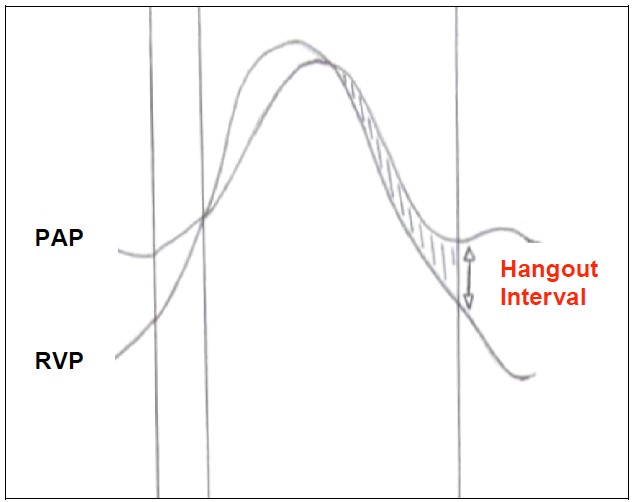
Figure 3. Right ventricular pressure tracing. PAP (pulmonary artery pressure) RVP (right ventricular pressure) Hangout interval, period of forward flow despite negative arterial-ventricular pressure gradient.
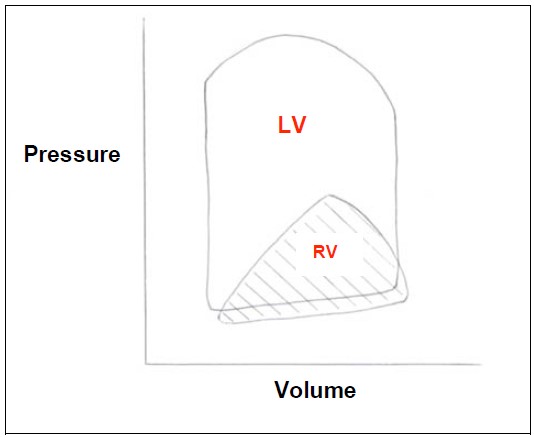
Figure 4. Pressure-volume loops for left and right ventricles.
In contrast to the left ventricle, perfusion of the right ventricle occurs during systole and diastole, with a systolo-diastolic coronary blood flow ratio higher than that of the left ventricle despite having half the oxygen consumption.
Preload
The preload of the right ventricle is determined by venous return and the distensibility of the right ventricular wall. As with the left ventricle the Frank-Staring mechanism is obeyed (Figure 5), however the right ventricle requires lower filling pressures to create the same ventricular output due pulmonary vascular resistance being considerably lower than systemic vascular resistance (Figure 6).
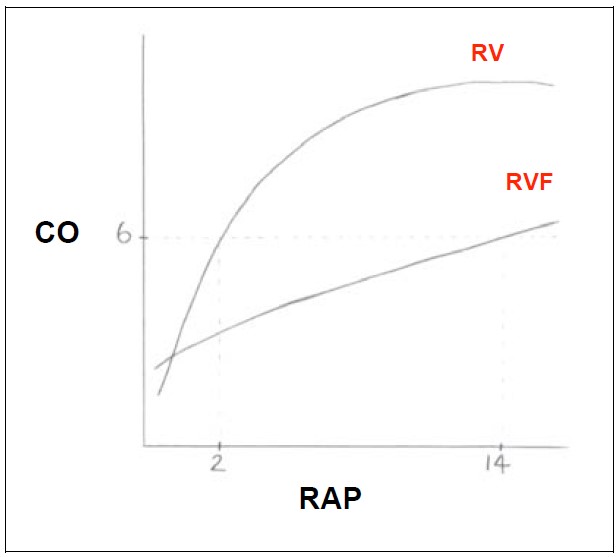
Figure 5. Frank Starling curve for the right ventricle (RV). CO (cardiac output), RAP (right atrial pressure) RVF (right ventricular failure)
The right ventricle compensates for changes in preload through stretching of the muscle wall (compliance) without loss of contractility (i.e. large increase in end-dyastolic volume without increase in end diastolic pressures). When the critical point on the Starling curve is reached the right heart fails due to exhaustion of these reserves.
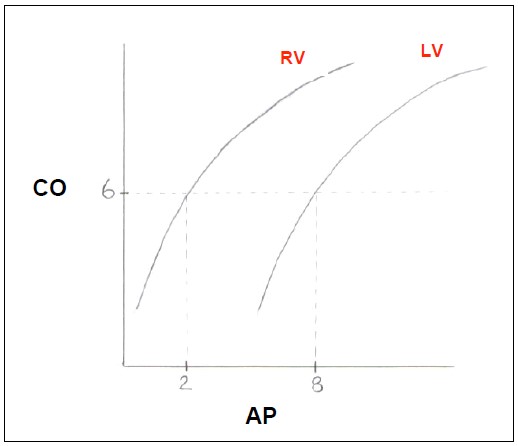
Figure 6. Comparison of right ventricular (RV) and left ventricular (LV) Starling curves. Note how the LV requires higher atrial filling pressures (AP) to produce the equivalent cardiac output (CO)
One must also not forget the influence of respiration. The mechanical work of breathing has a major impact on right heart haemodynamics. In a spontaneously breathing patient, during inspiration venous return (preload) is increased and during expiration it is decreased. This leads to fluctuations in stroke volume during the respiratory cycle. In contrast, during positive pressure ventilation venous return is reduced and this has been shown to result in a 10-15% reduction in cardiac output in normal subjects.
Afterload
Impedance of the pulmonary vascular bed represents the afterload of the right ventricle and remains low as long as the vascular bed can be recruited. Compared with the left ventricle the right ventricle demonstrates a heightened sensitivity to afterload changes. Consequently there is close correlation between pulmonary vascular resistance and right ventricular systolic function with a negative correlation between right ventricular ejection fraction and mean pulmonary artery pressure.
Other factors
Contractility, heart rhythm and ventricular interdependence are also important in right ventricular function. Atrial fibrillation and complete heart block are particularly disadvantages in right ventricular failure. It is also reported that approximately 20-40% of right ventricular contraction is provided from left ventricular contraction via the interventricular septum, therefore failure of the left heart can lead directly to dysfunction of the right heart. . Even the left ventricle can be penalised by a failing right ventricle via ventricular interdependence, whereby left ventricular filling is impaired by a left shift of the interventricular septum due to overstretch of the right ventricle within the shared pericardium.
PATHOPHYSIOLOGY
Right ventricular failure occurs when the right ventricle is unable to function properly despite fully activated compensatory mechanisms (Starling mechanism, ventricular hypertrophy, autonomic nervous system and hormonal changes). The result is systemic venous congestion, underfilling of the left ventricle, low cardiac output or cardiogenic shock.
There are multiple causes of right ventricular failure. The most important of these are summarised below.
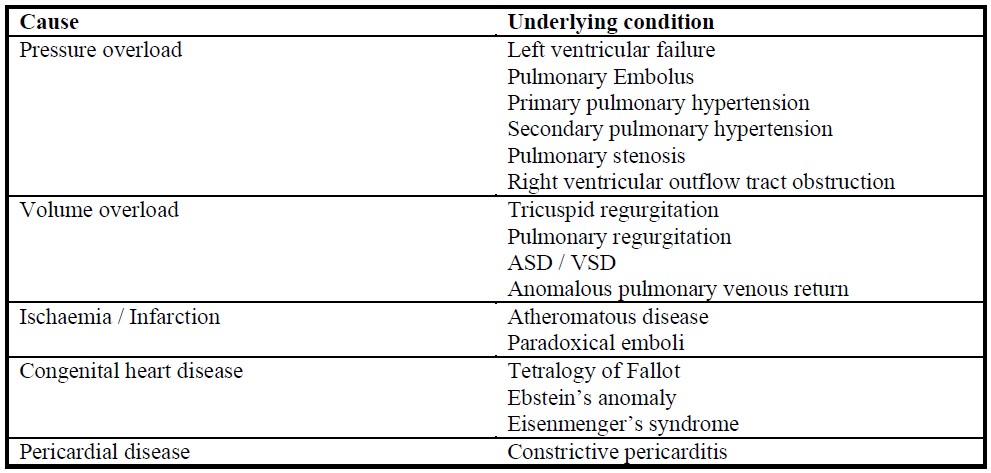
Table 2. Causes of right ventricular failure
Left ventricular failure
Chronic left ventricular failure is the commonest cause of right ventricular dysfunction. It occurs secondary to pulmonary venous hypertension and a fall in right ventricular contraction as the contribution from left ventricular contraction is lost (ventricular interdependence). It has been shown that the right ventricular ejection fraction represents an independent predictor of mortality in left ventricular failure.
Pulmonary embolus
This is the commonest cause of acute right ventricular dysfunction. The sudden increase in pulmonary vascular resistance (afterload) occurs via direct obstruction and hypoxic pulmonary vasoconstriction. A distinct pattern of right ventricular dysfunction ensues characterised by severe hypokinaesia of the free right ventricular wall with normal apical contraction. Mortality is closely related to the degree of right ventricular failure and haemodynamic instability.
Secondary pulmonary hypertension
Also known as cor pulmonale this is defined as right ventricular enlargement and hypertrophy secondary to pulmonary disease in the absence of left ventricular failure. The most common culprit is chronic obstructive pulmonary disease (COPD) followed by pulmonary fibrosis and adult respiratory distress syndrome. In COPD the rise in pulmonary arterial pressure is often mild but worsened by the degree of hypoxaemia due to hypoxic pulmonary vasoconstriction.
Ischaemia
Acute right coronary artery occlusion results in right ventricular free wall dysfunction. As a result right ventricular systolic function is depressed leading to a decrease in transpulmonary delivery of left ventricular preload and reduced cardiac output. The ischaemic right ventricle is stiff, dilated and volume dependent. This leads to a shift of the interventricular septum into the left ventricle. Consequently left ventricular compliance is also reduced, further decreasing cardiac output. The right ventricle becomes dependent on left ventricular contractile contributions to maintain residual function which is further complicated by the high incidence of arrhythmias associated with right coronary artery occlusion especially bradyarrhythmias.
Valvular heart disease
Right ventricular dysfunction is seen in both left and right valvular heart disease. Mitral stenosis and mitral regurgitation both lead to pulmonary hypertension. Pulmonary regurgitation leads to right ventricular dilatation. However, in aortic stenosis right ventricular systolic function is well maintained.
Congenital heart disease
The intricate and complicated physiology of congenital heart disease is outside the scope of this tutorial. However an atrio-ventricular septal defect alone or as part of a more complicated syndrome results in left to right shunting of blood and consequent volume overload. This is usually tolerated well if chronic but increases morbidity and mortality.
SUMMARY
- The right heart pumps in series with the left heart but it differs from the latter in shape and physiology.
- In the majority of people, the right heart is perfused by the RCA.
- Right atrial and ventricular defects can be asymtomatic and may not be diagnosed until adulthood.
- Due to the low pressures and high compliance of its chambers, the right heart can tolerate great changes to its physiology before decompensating.
- Hopefully this tutorial has demonstrated the key role that the right heart plays in cardiovascular haemodynamics and that it should no longer be simply considered as a passive conduit that returns blood to the left ventricle via the lungs.
Answers:
- b
- c
- a
Explanations to be found in the text.



