General Topics
SELF-ASSESSMENT QUESTIONS
Before continuing, try to answer ‘true or false’ to the following statements. The answers can be found in the text.
- Methicillin-resistant Staphlyococcus aureus can cause necrotising fasciitis
- Pain is often a late sign
- Flucloxacillin is the first line antibiotic
- Blood cultures are positive in approximately 80% of patients
- Hyberbaric oxygen therapy can be useful in treating infections caused by clostridium species
SUMMARY
The term necrotising fasciitis (NF) refers to ‘severe inflammation of the muscle sheath that leads to necrosis of the subcutaneous tissue and adjacent fascia’1. It is associated with a significant risk of mortality and long-term disability from limb and tissue loss. It occurs frequently enough that physicians outside critical care are likely to be involved in the management of at least one patient with NF during their practice, but infrequently enough that familiarity with the disease will seldom be achieved.1,2
Early clinical suspicion, surgery and antibiotics are key to improving survival as rapid progression can occur leading to systemic toxicity, limb loss and death.3
Patients need integrated, interdisciplinary management involving surgeons, intensive care physicians and microbiologists. Close monitoring and physiological support in an intensive care unit is often essential.
This tutorial describes the diagnosis and common management of NF. Less conventional treatments, including intravenous immunoglobulins and hyperbaric oxygen, are also discussed.
INTRODUCTION
NF is often grouped alongside other severe soft tissue infections such as myositis and myonecrosis (gas gangrene). Necrotising soft tissue infections are associated with considerable mortality.1
The overall incidence of NF has been estimated as 0.24 – 0.4 per 100 000 adults. In older reports, inadequate culture techniques together with reviews and meta-analyses that fail to differentiate between the different types of NF make it difficult to attribute causation exactly.3
CAUSATIVE ORGANISMS
A wide spectrum of bacteria have been found to cause NF, which can be broadly classified into 2 groups:
Type I NF (also known as synergistic NF)
Type I NF is a mixed infection caused by anaerobic, aerobic and facultatively anaerobic gram-positive and gram-negative bacteria. Up to 15 different organisms can be cultured from the affected areas with an average of 5 pathogens in each wound. Most of the pathogens originate from the bowel flora e.g. E.coli, Pseudomaonasspp, Bacteroides spp and Vibrio spp. Risk factors include immune compromise, recent surgery and underlying abdominal pathology including malignancy.3, 4
Type II NF
Type II NF is usually monomicrobial and due to gram-positive organisms. The most common pathogen causing type II NF is group A β-haemolytic streptococcus either alone or in combination with other species, usually Staphlyococcus aureus. Cases of NF without a recognised precipitating factor are more likely to be due to streptococcal infection or, more recently, due to community-acquired methicillin-resistant Staphylococcal infection (CA-MRSA).1,11 Risk factors include lacerations, burns or any injury to cause a breach in the dermis and recent surgery, childbirth, intra-venous drug use and varicella infection have all been linked to type II NF. In cases with no clear portal of entry, it is likely to be due to haematogenous spread from the throat to a site of skin injury.3 Mortality in this group is very high, approaching 50%–70%.4
RISK FACTORS FOR DEVELOPING NF
General risk factors include age over 50 years, intra-venous drug use and chronic debilitating conditions such as diabetes, immune suppression, obesity and peripheral vascular disease.1 The possibility that non-steroidal anti- inflammatory drugs (NSAIDs) may contribute to the development or progression of NF has been raised. It has been suggested that NSAIDs may mask the early symptoms and signs of NF or that they may directly contribute to the pathogenesis of NF by inhibiting neutrophil function and augmenting cytokine release.3,5
PATHOPHYSIOLOGY
NF is an infection of the deeper tissues that results in progressive destruction of the muscle fascia and overlying subcutaneous fat. Muscle tissue itself is frequently spared because of its generous blood supply.6 Infection typically spreads along the muscle fascia and the overlying tissue can initially appear unaffected. It is this feature that makes NF difficult to diagnose without surgical intervention.
Whilst the underlying pathophysiology of Type I and Type II NF is the same the clinical manifestations of these conditions can differ depending on the causative organism. Type I NF is typically a slow process that evolves over days. It often follows abdominal surgery, a perineal infection or an ischiorectal abscess.3 Whilst type II NF progresses more rapidly. As mentioned earlier, the disease may have arisen without any obvious focus in a previously well patient. Approximately 50% of type II NF cases are associated with the exotoxin driven disease- toxic shock syndrome. This association of toxic shock syndrome and type II NF has been demonstrated to significantly increase mortality from 40% to 67%.3
Massive T cell proliferation and cytokine release can occur in type II NF via the effects due to the M protein of group A β-haemolytic streptococcus (GAS). This filamentous protein has anti-phagocytic properties and is an important virulence determinant as it can produce multiple pyogenic exotoxins that act as super-antigens. These cause a systemic inflammatory response syndrome (SIRS) which may progress to multi-organ failure.
DIAGNOSIS
As already mentioned, NF can progress in an insidious manner and the diagnosis is often difficult to establish. It is important to always have a high index of suspicion as often by the time NF has been diagnosed the condition has progressed to late NF. Certain clues can be obtained from the history that point to a diagnosis of NF such as soft tissue injury, recent surgery, ulcers or illicit drug use.3 GAS NF may be secondary to haematogenous spread from throat infections, vaginitis, or close contact with impetigo. The diagnosis is particularly hard to make in general practice as often patients will present with non-specific signs. One fifth of patients have influenza-like symptoms characterized by fever and myalgia, while others may present with symptoms of gastroenteritis.3
Initial signs and symptoms usually include pain, swelling, erythema, pyrexia, and tachycardia. However it is only once the disease progresses that the typical more advanced skin changes are observed. These include tense oedema, pain disproportionate to skin changes and dusky blue/purple plaques that progress to haemorrhagic bullae and necrosis.1 Gas forming organisms and anaerobes may produce crepitus. Progression of these signs and symptoms is usually fast, particularly if GAS or Clostridium species are involved.
Common differential diagnoses include cellulitis, clostridial myonecrosis, deep vein thrombosis, gastro-enteritis, sunburn or an allergic rash. Self-administration of NSAIDs may mask the temperature or pain and anaesthesia produced by nerve destruction in late disease may be falsely reassuring.3 When the underlying infection ascends to the subcutaneous tissues the epidermal and dermal layers become oedematous, giving a ‘peau d’orange’ appearance with a ‘woody’ firmness of the skin.3
A definitive diagnosis of NF is established surgically with visualization of fascial planes and subcutaneous tissue in theatre. However, clinical clues and diagnostic tools should be used in combination to help make an early diagnosis.3
- The earliest clinical feature common to all types of NF is exquisite pain, often out of proportion to any visible skin changes.
- This is due to occlusion of perforating nutrient vessels with resultant nerve infarction.3
- Pain progresses until the nerves supplying the skin are destroyed resulting in anaesthesia of the affected areas.
LABORATORY INVESTIGATIONS
Blood cultures are positive in approximately 60% of patients with type II NF due to GAS but the yield is lower in patients with synergistic NF – around 20%.13
Other laboratory findings are usually non-specific and frequently demonstrate a high white cell count, high CRP, coagulopathy and raised creatine kinase. Patients are often hypocalcaemic due to calcium precipitation and fat necrosis.
Laboratory scoring tool for prediction of NF
The LRINEC (Laboratory Risk Indicator for Necrotising Fasciitis), illustrated below, is a tool developed to help distinguish NF from other types of soft tissue infections based on laboratory investigations.7 It was derived from a retrospective review that compared 89 cases of confirmed NF with 225 control cases of cellulitis or abscesses. Numerous variables were analysed, of which the most statistically significant indicators for NF infection were white cell count, CRP, haemoglobin, sodium, creatinine and glucose. The study concluded that a score of six or more should raise the suspicion of NF and a score of eight or more is strongly predictive of the disease.
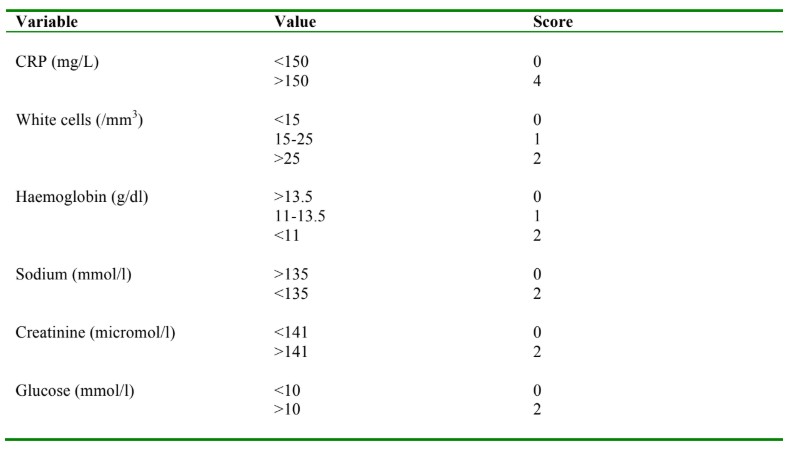
Table1. Laboratory risk Indicator for necrotising fasciitis (LRINEC)
Histology and microbiology
Frozen section biopsies that include the advancing edge and central necrotic areas have been found to be useful in diagnosing NF early. This method is often impractical however, as frozen sections are not readily available out of hours and the time taken to carry out, process and analyse the sample could be used for debridement in theatre. Instead, a bedside ‘finger test’ is recommended which involves infiltrating the area with local anaesthetic and making a small incision down to the deep fascia. The test is positive if there is a lack of resistance to finger dissection and lack of bleeding and contractile muscle function in addition to the release of ‘dishwater fluid’. This is an offensive, brown-coloured fluid that is released at the time of the finger test that is due to lysis of polymorphs and fascial necrosis with or without myonecrosis.1,3 All tissue samples should be gram stained and cultured aerobically and anaerobically. Histological examination of the tissue reveals vasculitis, thrombosed vessels, necrosis, polymorphonuclear infiltrate and microorganisms.3
Imaging
Radiological tests should never delay surgery if there is crepitus on examination or evidence of advancing soft- tissue infection. X-rays, computed tomography scanning (CT) and magnetic resonance imaging (MRI) are useful in diagnosing NF if there is subcutaneous gas present. This is a highly specific finding, but not very sensitive.
Studies have shown that increased thickness of the fascial layer can be associated with NF. However, this mirrors the results of clinical findings – high sensitivity but low specificity. The soft tissue swelling demonstrated may be due to recent trauma or surgery. CT has the additional advantage of identifying other sources of infection, particularly deep abscesses, however it usually requires the additional transfer of an often unstable patient to the radiology department.1,5
TREATMENT
Treatment of NF consists of early and aggressive debridement of necrotic tissue, together with broad-spectrum antibiotics and haemodynamic support. Treatment should be delivered by a multidisciplinary team of intensivists, microbiologists and surgeons, preferably including a plastic surgeon.3
Surgery
NF is a surgical emergency and warrants immediate surgical referral. Evidence has shown that when treatment is based on just antimicrobial therapy and support, mortality approaches 100%. Surgery is a vital means of source control. The goal of surgical intervention is to aggressively debride of all the necrotic tissue until healthy, viable (bleeding) tissue is reached.5
Tissue samples should be sent to the laboratory for gram staining and culture. The wound is then covered and the patient should be taken back to theatre 24 hours later for re-assessment and further debridement if necessary. Occasionally very aggressive resection including amputation of a limb is necessary to effectively stop the spread of infection.1
Antibiotics
Antibiotics are a vital adjunct to source control and may prevent the progression of septic shock when used early. In a widely quoted paper by Kumar et al effective antimicrobial administration within the first hour of documented hypotension was associated with increased survival in patients with septic shock. Furthermore, in this study which was not restricted to patients with NF, each hour of delay in administering antibiotics was associated with an average decrease in survival of 7.6%.8
Broad-spectrum antibiotics should be commenced immediately to include coverage of gram positive, gram negative and anaerobic organisms. GAS NF is almost always penicillin sensitive but high concentrations of the bacteria in the tissue results in most bacteria being in the stationary phase of the growth cycle, making antibiotics that target cell wall synthesis ineffective. Clindamycin is a lincosamide antibiotic that can switch off exotoxin production even in the stationary phase.3 In order to adequately cover for synergistic gram negative and gram positive NF an intravenous carbapenem, such as meropenem, should also be added. As cases of community acquired MRSA NF have increased over the years,11 antibiotics such as vancomycin or linezolid should be administered until MRSA has been excluded. Antibiotic regimes can subsequently be rationalised once culture results are known.
Broad spectrum antibiotics should be commenced immediately. A recommended regime is:
Clindamycin + meropenem + vancomycin
General supportive treatment
Patients with NF frequently develop septic shock and multi-organ failure. Other common complications include disseminated-intra-vascular coagulation, adult respiratory distress syndrome, acidosis and hypothermia. In addition to this patients will often have large areas of skin and subcutaneous tissue debrided at a time, resulting in a large ‘third space loss’ of fluid that is difficult to quantify. Patients should be managed in a critical care setting with close cardiac output monitoring, access to inotropic support and tight glycaemic control.
Intravenous immunoglobulin
Several studies have looked at the role of intravenous immunoglobulins (IVIG) in NF.1 These studies were small and difficult to compare due to different methodologies used. Therefore the exact benefits of IVIG remain uncertain. IVIG appears to be useful in GAS infections as they contain neutralizing antibodies that act against streptococcal antigens. In a controlled trial with 21 patients with streptococcal toxic shock syndrome (with or without necrotising fasciitis), patients who received the placebo had a 3.6-fold higher mortality rate than those who received IVIG.9 There is very little evidence of any benefit from using IVIG in gram negative sepsis. As a suitable powered study is unlikely to be performed in the future IVIG can be considered as an add-on therapy if it is available.
The dose of intra-venous immunoglobulin is:
2g/kg infusion with the option of a second dose if necessary after 24 hours.
Hyperbaric oxygen
Hyperbaric oxygen (HBO) is thought to increase the bactericidal effects of neutrophils since at low oxygen tensions killing mechanisms reliant on oxygen free radicals are less effective. HBO alone is also thought to be bactericidal for certain anaerobes, including Clostridium Perfringens as it switches off α-toxin production.3 It is for this reason that HBO is believed to be useful in treating synergistic infections.
A retrospective review using a nationwide database from the US identified 45,913 cases of NF, of which 405 were treated with HBO therapy. The study concluded that HBO treatment resulted in a statistically significant reduction in mortality (OR 0.49, 95% CI 0.29-0.83) but at a higher hospital cost and increased length of stay.2
When used to treat infections, HBO should be implemented early with two to three ninety-minute sessions at 3 atm. These high pressures maintain oxygen tension above 300 mmHg, which is sufficient to inhibit clostridial spore and exotoxin production.
Having said this there are few hospitals worldwide with easy access to HBO units with appropriate staffing and chambers large enough for patients needing intensive care facilities.3 It is anticipated that HBO will therefore remain an uncommon treatment for NF.
SUMMARY
NF is an infection of the deeper tissues that results in progressive destruction of the muscle fascia and overlying subcutaneous fat. It is a relatively infrequent but highly lethal infection. It is difficult to diagnose early and even more difficult to manage effectively.1 A variety of tools are available to aid the diagnosis of NF but a high index of suspicion and early surgical referral remains essential for both diagnostic confirmation and for effective treatment.
Broad-spectrum antibiotics should be administered as soon as possible then rationalised later once culture results are known. Acceptable regimes include clindamycin, a carbapenem, e.g. meropenem, and an agent with activity against MRSA e.g. vancomycin, linezolid.
The role of IVIG and HBO in the treatment of NF remains unclear due to the lack of randomized controlled trials. However if available, these therapies should be considered when surgical debridement, antibiotics and general supportive measures have proved unsuccessful.
ANSWERS TO SELF-ASSESSMENT QUESTIONS
- Methicillin-resistant Staphylococcus aureus can cause necrotising fasciitis – True
- Pain is often a late sign – False
- Flucloxacillin is the first line antibiotic – False
- Blood cultures are positive in approximately 90% of patients – False
- Hyberbaric oxygen therapy can be useful in treating infections caused by clostridium species – True
REFERENCES
- Anaya D, Dellinger E. Necrotising soft tissue infection: diagnosis and management. Clin Infect Dis 2007; 4: 705
- Soh CR, Pietrobon R, Freiberger JJ, Chew ST, Rajgor D, Gandhi M et al. Hyperbaric oxygen therapy in necrotising soft tissue infections: a study of patients in the United States Nationwide Inpatient Sample. Intensive Care Med 2012; 38:1143
- Morgan MS. Diagnosis and management of necrotizing faciitis: a multiparametric approach. Journal of Hospital Infection 2010. 75; 249 – 257
- Stevens DL, Bisno AL, Chambers HF, Everett ED, Dellinger P, Goldstein EJ et al. Practice guidelines for the management of skin and soft tissue infections. Clin Infect Dis 2005; 41: 1373 – 1406
- Stevens DL, Baddour L. Necrotising soft tissue infections. www.uptodate.com
- Gozal D, Ziser A, Shupak A, Ariel A, Melamed Y. Necrotising fasciitis. Arch Surg 1986; 121: 233
- Wong CH, Khin LW, Heng KS. The laboratory risk indicator for necrotising fasciitis score: a tool for distinguishing necrotising fasciitis from other soft tissue infection. Crit Care Med 2004; 32: 135 – 141
- Kumar A, Roberts D, Wood KE, Light B, Parillo JE, Sharma S et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006. Vol 34. 6; 1589 – 1596.
- Kaul R, McGreer A, Norrby-Teglund A, Kotb M, Schwartz B, O’Rouke K et al. Intravenous immunoglobulin therapy for streptococcal toxic shock syndrome – a comparative observational study. Clin Infect Dis 1999; 28:800 – 807
- Tibbles PM, Edelsberg JS. Hyperbaric oxgygen therapy. N Engl J Med 1996; 334: 1642 – 1648
- Miller LG, Perdreau-Remington F, Rieg G, Mehdi S, Perlroth J, Bayer AS et al. Necrotising fasciitis caused by community-associated methicillin-resistant Staphlyococcus aureus in Los Angeles. N Engl J Med 2005; 352: 1445 – 1453
- Martin A. Necrotising fasciitis – early recognition and treatment can minimize risk of fatalities. N Z Publ Health Rep 1994; 1: 9 – 10.
- Wong CH, Chang HC, Pasupathy S, Khin LW, Tan JL, Low CO. Necrotising fasciitis: clinical presentation, microbiology, and determinants of mortality. J Bone Joint Surg Am 2003; 85 –A:1454



