Obstetrics Anaesthesia
QUESTIONS
Before continuing, try to answer the following true/false questions. If you are not sure, read the tutorial and try again. The answers can be found at the end of the article, together with an explanation.
- The hypothalamus and posterior pituitary are connected by a portal circulation
- The anterior pituitary secretes 6 peptide hormones including GH and ACTH
- ADH is synthesised in the posterior pituitary
- Dopamine inhibits prolactin release
- Pituitary macroadenomas are greater than 20mm in diameter
- GH inhibits gluconeogenesis
- The dexamethasone suppression test is used in making a diagnosis of acromegaly
- Cushing’s Disease is also known as Cushing’s Syndrome
- TRH from the hypothalamus stimulates TSH release from the anterior pituitary
- Glucocorticoids inhibit ACTH release from the anterior pituitary
INTRODUCTION
The pituitary is a complex endocrine gland that produces a variety of hormones, essential to survival. These hormones have both direct and indirect effects on other endocrine glands and cell turnover, growth and metabolism. Secretion of hormones by the pituitary is normally regulated by the hypothalamus and the two structures are closely linked by vascular and neural connections. Hyper- and hypo-secretion of pituitary hormones, most commonly due to pituitary tumours, can lead to various clinical conditions. Several of these conditions will be discussed later in this tutorial.
ANATOMY
The hypothalamus makes up part of the diencephalon of the brain and is situated below the thalamus where it forms the floor of the third ventricle. Directly below the hypothalamus lies the pituitary gland. The two areas are connected by neuronal tissue and blood vessels within the infundibular stalk.
The pituitary gland, which is also called the hypophysis, is situated in the middle cranial fossa. It is found in a depression of the base of the skull in the body of the sphenoid bone known as the sella turcica (Turkish saddle). This is located directly below the diaphragma sellae, a sheet of dura through which the infundibular stalk passes separating the optic chiasm from the anterior pituitary.
The pituitary gland has a diameter of approximately 8mm and is divided functionally and embryologically into two parts:
- The anterior pituitary (adenohypophysis)
- The posterior pituitary (neurohypophysis)
The anterior pituitary gland has an ectodermal origin, being derived from buccal epithelium known as Rathke’s pouch. The smaller posterior pituitary is derived from the floor of the third ventricle.
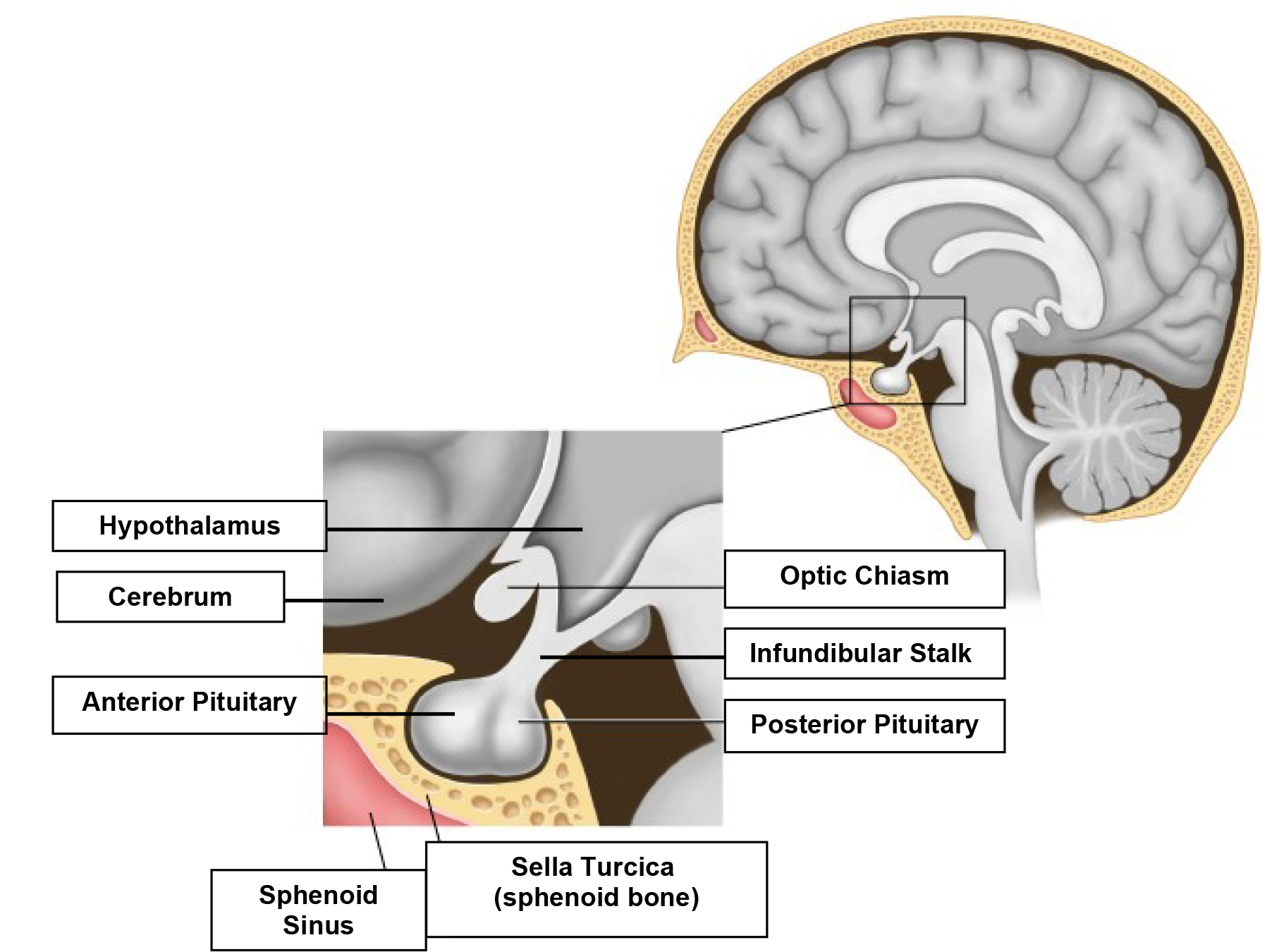
Figure 1: Diagram of the anatomical relations of the pituitary gland and hypothalamus
BLOOD SUPPLY
The hypothalamus receives blood from branches of the circle of Willis. The blood supply of the anterior pituitary is mainly from the superior hypophyseal artery and the posterior pituitary receives blood from the inferior hypophyseal artery, both of which are branches of the internal carotid artery. Some capillaries from the inferior hypophyseal artery pass through the pituitary stalk as portal veins which also supply the anterior pituitary with blood and connect the hypothalamus and anterior pituitary in a portal circulation. A portal circulation is one which connects two capillary beds but does not receive a direct arterial supply nor drain into a venous system. This complex vascular network means that the anterior pituitary can receive blood containing hormones from the hypothalamus as well as receiving substances which are circulating in the peripheral circulation. The venous drainage from the anterior pituitary gland passes into the cavernous sinuses and then into the petrosal sinuses and jugular vein.
ANTERIOR PITUITARY HORMONES
The anterior pituitary secretes 6 peptide hormones. Peptides are small proteins which are made up of chains of amino acids. The 6 anterior pituitary hormones are:
- Adrenocorticotropic Hormone (ACTH)
- Growth Hormone (GH)
- Follicle Stimulating Hormone (FSH)
- Luteinising Hormone (LH)
- Thyroid Stimulating Hormone (TSH)
- Prolactin (PRL)
POSTERIOR PITUITARY HORMONES
The posterior pituitary secretes 2 peptide hormones which are synthesised respectively in the supraoptic and paraventricular nuclei of the hypothalamus:
- Antidiuretic Hormone (ADH)
- Oxytocin
These hormones are then transported to the posterior pituitary from where they are released into the circulation to act on their target organs.
THE CONTROL OF PITUITARY HORMONE RELEASE
Anterior Pituitary Hormone Release
The hypothalamus secretes hypothalamic-releasing and inhibiting factors which pass through the portal circulation to act on the anterior pituitary. In response to these factors, there is stimulation or inhibition of the release of peptide hormones from the anterior pituitary into the bloodstream:

The anterior pituitary hormones and the hormones released from the target gland in turn exert a negative feedback influence on both the hypothalamus and the anterior pituitary.
Posterior Pituitary Hormone Release
The main stimulus to ADH release is an increase in plasma osmolality which is detected by osmoreceptors (water receptors) in the hypothalamus. Once plasma osmolality is returned to normal, a negative feedback mechanism inhibits further ADH release. The other important stimulus to ADH release is a reduction in the effective circulating volume (e.g. in haemorrhage). This is detected by baroreceptors (pressure receptors) in the aortic arch and carotid sinus.
Suckling is the main stimulus to the release of oxytocin which in turn stimulates the secretion of milk from the milk ducts.
SUMMARY OF THE PITUITARY HORMONES
ACTH
Site of Action: adrenal cortex
Effects: mainly stimulates the synthesis of glucocorticoids (steroid hormones involved in glucose synthesis) in the zona fasciculata. ACTH also stimulates the synthesis of mineralocorticoids (steroid hormones involved in sodium retention) in the zona glomerulosa. Glucocorticoids e.g. cortisol stimulate gluconeogenesis (glucose synthesis from amino acids and glycerol), promote the breakdown of fats (lipolysis) and damp down the inflammatory response. Mineralocorticoids e.g. aldosterone promotes sodium and water retention by the kidneys.
Release Stimulated by: CRH, stress (including surgery)
Release Inhibited by: glucocorticoids
GH
Site of Action: all body systems
Effects: protein synthesis, lipolysis, skeletal and fetal growth, gluconeogenesis, sodium retention and reduced sensitivity to insulin. Some of the effects of GH, including protein synthesis, skeletal growth and fetal growth are mediated by insulin-like growth factors I and II (IGF I and II). These are polypeptides with a similar structure to insulin and are produced by the liver in response to stimulation by GH.
Release Stimulated by: GHRH, exercise, hypoglycaemia, stress, glucagon, and dopamine
Release Inhibited by: somatostatin, GH
FSH
Site of Action: testes and ovaries
Effects: stimulates spermatogenesis in the male and ovarian follicle growth in the female
Release Stimulated by: GnRH
Release Inhibited by: oestrogen, testosterone (in men)
LH
Site of Action: testes and ovaries
Effects: testosterone secretion in the male, luteinisation of ovarian follicles and ovulation in the female
Release Stimulated by: GnRH, oestrogen
Release Inhibited by: oestrogen and progesterone (after ovulation), testosterone (in men)
TSH
Site of Action: thyroid
Effects: stimulates follicular activity and the synthesis and release of thyroid hormones (T3 and T4)
Release Stimulated by: TRH
Release Inhibited by: somatostatin, thyroid hormones
PRL
Site of Action: breasts
Effects: development of the breasts ready for lactation, stimulates milk production, reduces fertility
Release Stimulated by: PRLH, dopamine antagonists, suckling, prolactin (positive feedback mechanism), stress, exercise
Release Inhibited by: dopamine (also known as prolactin-inhibitory hormone)
ADH
Site of Action: distal tubule and collecting ducts in the kidney, blood vessels
Effects: water reabsorption in the kidney at the above sites, arteriolar vasoconstriction, synthesis of Factor VIII
Release Stimulated by: increased osmolarity of extracellular fluid, pain, haemorrhage, stress, thirst, activation of the renin-angiotensin system
Release Inhibited by: alcohol, reduced osmolarity of extracellular fluid
Oxytocin
Site of Action: breasts, kidneys
Effects: stimulates contraction of myoepithelial cells surrounding the milk ducts, therefore causing secretion of milk. Also stimulates contraction of the uterus and water retention by the kidney
Release Stimulated by: suckling
Release Inhibited by: dopamine
HYPER- AND HYPO-SECRETION OF PITUITARY HORMONES
Hyper- and hypo-secretion of pituitary hormones can lead to various clinical conditions which are shown in the table below. Deficiencies of FSH, LH and oxytocin do not result in particular clinical syndromes but will cause symptoms due to target organ failure.

PITUITARY TUMOURS
Pituitary tumours are benign tumours which can be classified into microadenomas (< 10mm) and macroadenomas (> 10mm) or by the hormone(s) they produce.
Macroadenomas
These pituitary tumours usually present with symptoms due their mass effect; visual symptoms due to pressure on the optic chiasm or hypopituitarism due to destruction of the surrounding pituitary tissue and are usually non-secretory. These are managed in the first instance by medical treatment with dopamine agonists e.g. cabergoline or bromocriptine. Surgical resection used to be performed via a craniotomy, but this has now been superseded by endoscopic techniques via the transsphenoidal route. Where removal is incomplete, continued medical therapy or subsequent radiotherapy may be necessary.
Microadenomas
These tumours usually produce unregulated amounts of certain anterior pituitary hormones and as such present with symptoms secondary to hormone excess. The commonest pituitary hormone secreted by these tumours is prolactin (35%), followed by GH (20%) and ACTH (7%). Some tumours secrete a mixture of hormones, usually prolactin and growth hormone, whilst only approximately 1% produce LH, FSH or TSH. Excess secretion of PRL, GH or ACTH from a pituitary microadenoma can lead to hyperprolactinaemia, acromegaly or Cushing’s disease respectively. These conditions are outlined below.
HYPERPROLACTINAEMIA
Prolactin-secreting pituitary adenomas are one cause of hyperprolactinaemia (plasma prolactin > 390 mU/L). Many other conditions can also be responsible, particularly drug therapy (metoclopramide, chlorpromazine, haloperidol, oestrogens). A careful history must always be taken before a presumptive diagnosis of pituitary adenoma is made.
Clinical Features
In women, symptoms include weight gain, menstrual disturbance, infertility, depression and galactorrhoea (spontaneous secretion of milk from the breast). In men there may be impotence, reduced facial hair and galactorrhoea.
Diagnosis and Treatment
Diagnosis is made by measurement of plasma prolactin levels and pituitary MRI scan if a pituitary microadenoma is suspected (most likely if prolactin > 1000 mU/L). Microprolactinomas may be treated by transsphenoidal resection.
ACROMEGALY
Acromegaly is caused by over secretion of growth hormone. This is most commonly a result of hypersecretion by a pituitary tumour, less commonly it may be due to oversecretion of GHRH from the hypothalamus leading to over stimulation of the pituitary and excess GH secretion.
Most cases of acromegaly are diagnosed in patients aged 40-60 years with an approximately equal number of men and women affected. Characteristically, the disease is of slow onset and changes in physical features due to excess GH are often more apparent when comparison is made with the patient’s appearance in old photographs. Patients may complain that they find a need for an increase in shoe size!
Over secretion of growth hormone prior to puberty causes gigantism rather than acromegaly.
Clinical Features
Clinical features of acromegaly include an enlarged jaw (prognathism), prominent supraorbital ridges due to skull growth and kyphosis, increase in the size of hands and feet (these are not due to soft tissue enlargement – they are due to bony changes), soft tissue changes; enlarged nose, lips and tongue (macroglossia). There may also be a deepening of the voice, and development of coarse oily skin. Osteoarthritis, sleep apnoea, hypertension and diabetes mellitus secondary to the anti-insulin effect of growth hormone are also common. Enlargement of the heart (bovine heart) may cause heart failure.
As the name suggests, the main feature of patients with gigantism is extremely tall stature (giants). Characteristically there is often excess growth of the hands and feet (patients often have thick fingers and toes). Delayed puberty and visual disturbances including double vision may also occur.
Diagnosis and Treatment
Characteristic clinical features and noticeable changes in comparison with old photographs should lead to further investigation through the following:
- Oral Glucose Tolerance Test and GH measurement – patient is given a bolus of glucose (usually 75 g in 300mls water). Plasma glucose and GH levels are measured at 0 and then 30 minute intervals up to 120 minutes. In a normal patient there would be suppression of GH release; in acromegaly there is no suppression
- Pituitary MRI Scan
Treatment is usually via transsphenoidal surgery to remove the tumour +/- radiotherapy. Somatostatin analogues e.g. octreotide or dopamine agonists e.g. cabergoline are also used with limited success in some patients who are unfit for surgery.
CUSHING’S DISEASE
Cushing’s disease is adrenal hyperplasia caused by excess secretion of ACTH from a pituitary tumour. (NB. Cushing’s syndrome is adrenal hyperplasia due to excess ACTH secretion from any source, not just the pituitary gland). Cushing’s disease is most commonly diagnosed between 30-55 years and affects women more frequently than men. Untreated, the disease carries a 50% mortality at 5 years.
Clinical Features
These include weight gain and central obesity (characteristic ‘lemon on sticks’ appearance), thin skin, easy bruising, abdominal striae, proximal myopathy, poor wound healing, depression, hypertension and hyperglycaemia.
Diagnosis and Treatment
- Dexamethasone suppression test – dexamethasone 2mg is given orally four times daily for 2 days. Plasma and urinary cortisol are measured at 0 and 48 hours. In Cushing’s Disease there is no or only partial suppression of cortisol
- Pituitary MRI – if the above test is positive, a pituitary MRI will help to identify if a pituitary tumour is the source of ACTH secretion or if one should look for an ectopic source e.g. abdominal carcinoid tumour, small cell lung cancer
Treatment of a pituitary tumour is via transsphenoidal surgery. Drugs e.g. metyrapone can be used to reduce plasma cortisol.
Summary
- The anterior pituitary and hypothalamus are linked by a portal circulation
- The anterior pituitary secretes 6 hormones: ACTH, GH, FSH, LH, TSH and PRL
- The posterior pituitary secretes ADH and oxytocin
- Regulation of pituitary hormone release is via a negative feedback system
- Pituitary tumours may be microadenomas (< 10mm diameter) or macroadenomas (> 10mm)
- Tumours may present with symptoms due to a mass effect or hormonal hyper or hypo-secretion
- Hyper-secretion of hormones by pituitary tumours can lead to hyperprolactinaemia (excess PRL), acromegaly (excess GH) and Cushing’s Disease (excess ACTH)
ANSWERS TO QUESTIONS
- F – The hypothalamus and anterior pituitary are connected by a portal circulation
- T – The anterior pituitary secretes 6 peptide hormones including GH and ACTH
- F – ADH is synthesised in the supraoptic nucleus of the hypothalamus and secreted by the posterior pituitary
- T – Dopamine is also known as prolactin-inhibitory hormone
- F – Pituitary macroadenomas are greater than 10mm in diameter. Microadenomas are less than 10mm diameter
- F – GH is a diabetogenic hormone and stimulates gluconeogenesis
- F – The dexamethasone suppression test is used in the diagnosis of Cushing’s Disease
- F – Cushing’s Disease is adrenal hyperplasia secondary to excess ACTH from a pituitary tumour. Cushing’s Syndrome is adrenal hyperplasia due to excess ACTH from any source
- T – TRH from the hypothalamus stimulates TSH release from the anterior pituitary
- T – This is part of a negative feedback system
REFERENCES and FURTHER READING
- M. Longmore, I. Wilkinson, E. Török. The Oxford Handbook of Clinical Medicine. Fifth Edition: 300- 316. Oxford University Press
- C. Pinnock, T. Lin, T. Smith. Fundamentals of Anaesthesia. Second Edition: 497-505. Cambridge University Press
- R. S. Snell. Clinical Anatomy for Medical Students. Sixth Edition: 700. Lippincott Williams and Wilkins
- M. Smith, N. P. Hirsch. Pituitary Disease and Anaesthesia. Br J Anaesth 2000; 85: 3-14
- Diagram (with modification) from www.pituitary.me.uk



