General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Which of the following conditions can be managed using interventional neuroradiology?
a. Cerebral Aneurysm
b. Idiopathic Intracranial Hypertension
c. Acute thromboembolic stroke
d. Trigeminal neuralgia - The following monitoring requirements are advised for patients undergoing neuroradiological procedures:
a. Invasive arterial blood pressure
b. Central venous pressure
c. Urine Output
d. ACT - Appropriate anaesthetic management of patients having endovascular treatment of intracranial aneurysms includes?
a. Laryngeal mask airway
b. Arterial line
c. Attenuation of pressor response to laryngoscopy
d. Maintenance of high intracranial pressure (ICP)
INTRODUCTION
The scope of anaesthesia for neuroradiology is increasing, with a broad range of indications for increasingly complex and lengthy imaging and intervention. While anaesthetists are involved in managing anaesthetised patients for neurological imaging from early in their career, many of these cases can present challenges for even the most experienced consultants.
The evidence in favour of interventional neuroradiological techniques is growing, which will lead to an increased need for anaesthesia in increasingly complex patients [1]. It is therefore imperative that anaesthetists have a comprehensive understanding of the indications, techniques and potential pitfalls associated with this area of anaesthesia, in order to provide high quality care and maintain patient safety.
This tutorial aims to provide an overview of the key challenges and anaesthetic management of patients requiring anaesthesia for imaging and interventional neuroradiology (INR).
NEUROANATOMY AND PHYSIOLOGY
A sound understanding of basic neuroanatomy and physiology is important in order to appreciate the rationale behind the physiological management of patients presenting for imaging and intervention. For the purposes of this tutorial, this knowledge is assumed and can be revised elsewhere [2].
INDICATIONS FOR NEURORADIOLOGY
Patients requiring straightforward neuro-imaging may not require any anaesthesia at all. Local anaesthesia is an option for some interventional procedures. However, general anaesthesia can provide improved patient comfort, guaranteed patient immobility (and improved image acquisition), facilitation of manipulation of patient physiology and ability to manage complications swiftly. Neuroradiology procedures can be classified as diagnostic or therapeutic.
Diagnostic
Imaging in the form of CT or MRI may be performed urgently or electively to diagnose a multitude of neurological conditions and events, including intracranial haemorrhage, cerebrovascular accident and space occupying lesions (figures 1-3).
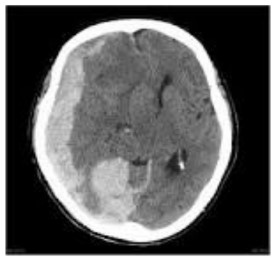
Figure 1. CT scan showing right subdural haemorrhage.
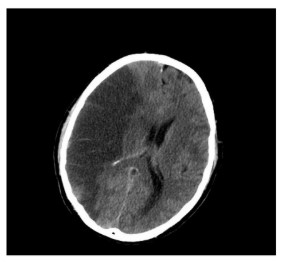
Figure 2. CT scan showing right MCA infarct.
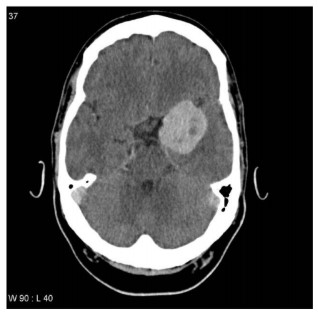
Figure 3. CT scan showing space-occupying lesion.
Anaesthetic support may be requested due to patients’ reduced level of consciousness, inability to lie still or inability to tolerate the investigation for psychological reasons. The highest quality images can be produced using digital subtraction angiography (DSA). With DSA, the neurovasculature is delineated using IV contrast and this image can be superimposed onto live, bone-subtracted fluoroscopy to allow radiologists to review the progress of catheters through the vasculature (figure 4).
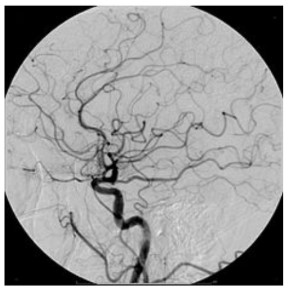
Figure 4. Cerebral angiography
Therapeutic
Interventional neuroradiology can be broadly defined as treatment by endovascular access for the purpose of delivering therapeutic drugs and devices [3]. The range of therapeutic interventions available is rapidly expanding, and includes:
- Endovascular treatment of cerebral aneurysms (coiling / embolisation)
- Endovascular treatment of AV malformations / fistulae (intracranial, dural, spinal)
- Pre-operative embolisation of cerebral and spinal tumours
- Embolisation for epistaxis
- Investigation and treatment (venous angioplasty/venous stenting) of Idiopathic Intracranial Hypertension (IIH)
- Sclerotherapy for venous angioma
- Balloon angioplasty for carotid stenosis and vasospasm
- Intra-arterial chemotherapy
- Treatment of acute thromboembolic stroke
- Stereotactic surgery
- Pain procedures
PATIENT ASSESSMENT
Thorough pre-operative patient assessment and planning is key to providing safe anaesthetic care. Patients presenting for emergency imaging may have a reduced level of consciousness necessitating emergency airway management and resuscitation. Patients presenting for interventional procedures can be confused, in pain, have multiple comorbidities, or be suffering from complications of the neurological insult itself.
History
Obtaining a history from the patient can be challenging due to communication difficulties and where possible, a collateral history should be sought. The history should include a standard anaesthetic assessment with specific questioning and review to focus on:
- Background history and chronology of recent events
- Neurological deficits and current GCS
- Symptoms of raised ICP
- In the presence of raised ICP, a higher mean arterial pressure (MAP) will be required to maintain cerebral perfusion pressure (CPP) (as CPP = MAP – ICP). Symptoms of raised ICP are therefore important, as the anaesthetist must maintain appropriate haemodynamic parameters at induction to maintain an adequate CPP. Furthermore, it must be recognised that ICP may vary significantly with altered clearance of CO2 and care should be taken to maintain a normal PaCO2 (4.5-5kPa)
- Previous neurosurgical procedures (and review of anaesthetic chart where possible)
- Allergy history and medication review
- It is useful to identify patients who may be at high risk from contrast induced nephropathy (elderly, diabetic, pre-existing renal insufficiency). Particular note should be made if contrast has been given previously without adverse reactions.
Importantly, there should also be a clear discussion with the patient regarding invasive monitoring, potential for critical care and anaesthetic risk. The details of the discussion must be clearly documented in the patient notes.
Examination
A focussed neurological examination and assessment of GCS must be made to establish the nature of any neurological deficits prior to induction of anaesthesia and prior to the neurosurgical intervention. Risk factors for SAH include hypertension and smoking so a cardiorespiratory assessment is also important. Acute aneurysmal disease is associated with cardiovascular dysfunction and the presence of arrhythmias or signs of cardiac failure should be identified and treated appropriately.
Investigations
A number of investigations need to be reviewed.
- Full blood count and coagulation testing
- A baseline haemoglobin is important in all patients. They may have bled and are at risk of further bleeding if undergoing a definitive radiological intervention. Furthermore, anticoagulation is a key factor in interventional neuroradiology and any underlying coagulation deficit must be identified.
- Check presence of a valid “group & save”
- If indicated, order blood products so that they are readily available.
- U&Es
- Electrolyte disturbances are common in patients with head injury and aneurysmal bleeds. They may also impact on the patient’s level of consciousness. Particular attention should be paid to the presence of hyponatraemia, which may indicate the presence of SIADH or cerebral salt wasting syndrome.
- Glucose
- Glycaemic control is important in all patients to prevent secondary brain injury. Recent evidence suggests intermediate goals for glycaemic control are appropriate in neurocritical care patients to prevent hypoglycaemia and optimise neurological recovery [4]. In our unit, target glucose levels are 6 – 10 mmol/L.
- ECG
- A recent ECG must be reviewed to assess for the presence of arrhythmias or myocardial ischaemia, which can be associated with SAH or traumatic brain injury.
- Imaging
- Any previous imaging should be reviewed. The World Federation of Neurosurgical Societies (WFNS) and radiological grade of SAH or severity of other diagnoses can allow a fully informed discussion with the patient and relatives to take place.
PERI-OPERATIVE CONSIDERATIONS
General Considerations
Remote Site Anaesthesia
Anaesthesia for neuroradiology is often performed in a remote site in which anaesthetic assistance may not be as immediately available as it might be in a theatre suite. Remote site anaesthesia is more challenging due to unfamiliarity with the isolated environment, available equipment, or indeed the procedure being undertaken, particularly if less experienced anaesthetists are involved. Anaesthetists should therefore seek to familiarise themselves with the environment and equipment and should ensure that relevant competencies are completed and that appropriate support is available before they start a case in the neuroradiology suite. Guidance on remote site anaesthesia is available from the RCOA (see
weblink).
Radiation safety
Although there may be facilities in some neuroradiology suites to monitor the patient from a distance, administration of anaesthesia for neuroradiology will invariably involve exposure to ionizing radiation. Therefore, adherence to radiation safety guidelines is vital. Available guidance advises combining various types of shielding. These should include the wearing of lead aprons, thyroid collars and glass lead screens. Protective eyewear should also be considered, as recent evidence suggests that neuroanaesthetists can be exposed to a significant amount of scattered radiation, with an associated risk of cataract formation [5]. Female anaesthetists with undiagnosed early pregnancy are a specific risk.
Specific Considerations
Patient access, equipment and monitoring
The neuroradiology suite can also cause problems for anaesthetists because of poor lighting and restricted access to the patient. In particular, the need for movement of the scanner around the patient’s head means that the airway is not readily accessible. The MRI scanner is a unique environment and specific guidance is available [6].
Standard AAGBI monitoring (ECG, NIBP, SpO2, ETCO2, ETAA) is, of course, essential. Temperature monitoring and patient warming is important because procedures are often long and the ambient temperature cool. Furthermore, because the procedures can be very long, with the use of large volumes of IV flush and contrast as well as the potential for diabetes insipidus, urinary catheterization should be performed prior to the procedure. All monitors need to be visible, audible and, in the case of MRI, compatible with the surroundings.
In addition to non-invasive monitoring, arterial lines are necessary during most interventional neurosurgical procedures to allow for accurate BP management (and therefore maintenance of CPP) as well as access for sampling for intraoperative testing of coagulation, electrolytes and blood gas analysis. Deliberate hypertension may be required in the event of vessel occlusion and vasospasm, or hypotension to reduce blood flow in a feeding artery to AVM before glue injection.
Standard and difficult airway equipment should be checked and available. As airway access is restricted, it must be carefully secured, and long breathing circuits are required as the patient’s head is often distant from the anaesthetic machine.
Secure IV access is essential. In addition to administration of infusions and medication, in all interventional neuroradiological procedures there is potential for significant haemorrhage. Due to the severity of complications that can arise from extravasations of contrast from central lines, peripheral IV lines are preferred for contrast administration (unless the central line is confirmed to be power- injectable). Central access may be considered but is not always necessary. All lines will need extensions and this needs to be considered when giving IV bolus injections.
Anticoagulation
Anticoagulation is an important concern for the anaesthetist in the neuroradiology suite. Patients may have been anticoagulated pre-operatively with IV heparin subsequently requested during endovascular procedures to reduce the risk of vessel thrombosis. This necessitates ACT (activated clotting time) monitoring to achieve 2-3 x ACT (normal range 90 – 130 seconds). Aspirin (IV or NG) and clopidogrel may also be required. In difficult cases, intra-arterial abciximab, a potent glycoprotein IIb/IIIa inhibitor, can be effective for the treatment of thrombotic complications occurring during aneurysm coiling [7]. The potential for spontaneous bleeding must be recognised by the anaesthetist, and provisions made to manage this if it occurs.
Anaesthetic goals
The anaesthetist is responsible for maintaining and manipulating physiological parameters as well as enabling patient immobility, the management of anticoagulation and treatment of complications. Overall, the key anaesthetic goals are
- Haemodynamic stability
- Maintenance of adequate CPP
- Patient immobility
- Rapid management of complications
- Smooth, rapid emergence to ameliorate undesirable effects on intracranial and central venous pressure
- Analgesia
While there will be some variability in anaesthetic technique between different practitioners, the anaesthetic goals are the same, and a number of points will be considered at each stage of the anaesthetic.
Induction
At induction, the main aim is to maintain haemodynamic stability, CPP and avoid blood pressure surges, which would increase transmural pressure and compromise the patency of unsecured aneurysms. The choice of intravenous agent is at the discretion of the anaesthetists provided these goals are met. The choice of muscle relaxant will also depend on the need for rapid sequence induction. Although laryngeal masks may have a role in emergency airway management, their use for neuroradiology is not widely advocated. Often an opioid is used to obtund the pressor response to laryngoscopy, but alternatives include intravenous lidocaine (suggested dose 1.5mg/kg) or labetalol (suggested dose 20-50mg). Esmolol may also be used – it has the advantage of a faster onset and reduced duration of action, making it easier to titrate to effect. Following intubation, the endotracheal tube must be secured carefully because of restricted airway access as discussed.
Maintenance
Both total intravenous anaesthesia (TIVA) and volatile anaesthesia maintenance are considered appropriate. Advantages of a TIVA technique – usually a propofol/remifentanil target controlled infusion – include haemodynamic stability and the avoidance of the potential for volatile-mediated uncoupling of cerebral metabolism and cerebral blood flow. A remifentanil infusion can also be used to maintain patient immobility without muscle paralysis, although atracurium (by intermittent boluses or infusion) can also be used for guaranteed immobility. TIVA should only be used by anaesthetists experienced and familiar with the technique. Sevoflurane is the volatile agent of choice as cerebral blood flow and metabolism coupling is maintained up to 1 MAC [8]. Nitrous oxide is best avoided due to the risk of exacerbating any potential venous air embolism. It is also a weak cerebral vasodilator increasing CBF and therefore ICP [9].
Post-operative analgesia should be planned according to the procedure performed. Angiography alone results in only minimal surgical stimulation. However, dural stimulation and tumour embolisation may be stimulating. This is usually managed using an infusion of remifentanil or boluses of other opioids such as fentanyl or alfentanil. These short acting opioids facilitate reliable neurological assessment in the post-operative period. Remifentanil is the most titratable of these and will wear off most rapidly.
Emergence
The aim is to wake the patient up in a smooth, rapid manner that allows for early neurological assessment. A number of techniques have been used. Anaesthetists may opt to extubate patients in a deep plane of anaesthesia and perform an ETT to LMA exchange or wake patients with a background remifentanil infusion in progress. Again, the choice of technique is less important than maintenance of physiological stability.
Recovery
Patients often require ongoing haemodynamic monitoring and BP management, and so may be managed post-operatively in a critical care area.
COMPLICATIONS AND THEIR MANAGEMENT
Complications during INR procedures can develop rapidly and must be managed swiftly. Calm, clear and concise communication between team members will facilitate systematic management of such events. A range of complications can arise (Table 1). The complications associated with most significant morbidity and mortality following SAH are re-bleeding and delayed cerebral ischaemia.

Table 1. Complications seen in Neuroradiology [3]
Haemorrhage
Haemorrhage may be spontaneous, iatrogenic or caused by anaesthetic factors (BP surges, inadequate depth of anaesthesia, poor control of BP at emergence) [10]. It may be identified by recognition of contrast extravasation or physiological changes (hypertension with or without bradycardia). Management involves rapid BP control, reversal of anticoagulation and measures to control a raised ICP. If bleeding cannot be controlled radiologically, the patient may have to be transferred to theatre for emergency craniotomy and clipping.
Vessel Occlusion
Occlusive complications can result from thromboembolic events, misplaced coils or vasospasm (discussed separately). In the event of vessel occlusion, management should involve direct lysis if possible, use of antiplatelet agents, and blood pressure manipulation to increase blood flow to ischaemic areas. Attempts should be made to remove misplaced coils [8].
Vasospasm
The term vasospasm is used to describe clinical and radiological changes – arterial constriction seen angiographically in association with neurological deterioration. The process is thought to be mediated by the presence of extravasated blood in the subarachnoid space. Traditionally, management has consisted of ‘triple H’ therapy; haemodilution (target haematocrit 30%), hypervolaemia and hypertension, based on the theory that vasospasm can be prevented or reversed by optimising cerebral blood flow. More recently, the focus of management has shifted to that of hypertension and euvolaemia. Appropriate systolic blood pressure targets are 160-180mmHg in secured aneurysms, and 140-160mmHg for unsecured aneurysms. A calcium channel blocker, nimodipine, is also used, and can be started prophylactically for a diagnosis of SAH with significant blood load. A typical regime is an infusion of 200 mcg/ml nimodipine at 0-10 mls/hr, or administer 60mg orally every four hours.
Delayed Cerebral Ischaemia (DCI)
DCI is a clinical syndrome of focal neurological or cognitive deficits, or both, occurring typically 4-12 days following aneurysmal SAH [11]. The term DCI encompasses a number of clinical entities including symptomatic vasospasm, delayed neurological deficit (DND – which occurs in the presence or absence of angiographic vasospasm), and asymptomatic delayed cerebral infarction [12]. The pathophysiology of DCI has, until recently, been attributed to vasospasm leading to tissue ischaemia, but an emerging body of evidence now suggests that DCI is likely to have a multifactorial aetiology. Triple-H therapy, magnesium and statins have all been used in the management of DCI, but nimodipine remains the only proven therapeutic intervention [13].
Contrast reactions
There are a number of risk factors for contrast-induced nephropathy that should be identified at pre-operative assessment. Advanced age, pre existing renal impairment, diabetes and hypovolaemia all increase risk. The anaesthetist has a role in managing fluid balance and cardiac output in order to minimise this risk. There is currently no good evidence to support N-acetylcysteine prophylaxis, and although IV sodium bicarbonate has been shown to reduce the incidence of nephropathy, mortality benefit has not been demonstrated and this is not used routinely [14]. All patients should have their renal function closely monitored following use of contrast in INR, and renal replacement therapy may be required if significant deterioration in renal function is seen.
INTERVENTIONAL MRI AND STEREOTACTIC SURGERY
The use of MRI in neurosurgery is increasing, with the development of intra-operative MRI (iMRI) techniques for tumour resection, epilepsy surgery and deep brain stimulation surgery [6]. Repeated scans during surgery help to improve the accuracy of lesion resection or device manipulation. It has been suggested that iMRI contributes to enhanced clinical outcomes, improved patient care, and possible economic savings if repeated surgeries can be avoided [15].
MRI is also used in stereotactic neurosurgery. A base ring and localising ring are applied to the patient’s skull under regional or general anaesthesia, and imaging (CT or MRI) is performed to facilitate ‘mapping’ of the area of interest. The ring will further restrict access to the patient’s airway and the anaesthetist must have full access to the ring at all times [10].
Anaesthesia in the MRI suite presents unique challenges for the anaesthetist that predominately relate to the issues of remote environment, need for specialised compatible equipment, restricted patient access and MRI safety concerns. Updated safety guidelines are available from the AAGBI (see weblink) and a comprehensive review of anaesthesia for MRI can be found in the reference section at the end of this tutorial [6].
EVIDENCE-BASED NEURORADIOLOGY
The body of evidence in favour of interventional neuroradiology for the management of neurological diagnoses is increasing.
- The International Subarachnoid Aneurysm Trial [1]
- Examined death or dependence at one-year following coiling versus clipping. In patients with ruptured aneurysms suitable for both treatments, coiling was more likely to result in independent survival at one year.
- Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) [16]
- Involved the random assignment of patients at high surgical risk to undergo either protected carotid artery stenting or carotid endarterectomy. The primary endpoint was the cumulative incidence of a major cardiovascular event at one year (a composite of death, stroke, or myocardial infarction within 30 days after the intervention or death or ipsilateral stroke between 31 days and one year. The study showed that stenting was not inferior to surgical endarterectomy
- A Randomised Trial of Unruptured Brain AVMs (ARUBA) [17]
- Medical management alone is superior to medical management with interventional therapy for the prevention of death or stroke in patients with unruptured brain arteriovenous malformations. The observational phase of this trial is continuing.
- The Cerebral Aneurysm Re-rupture After Treatment (CARAT) study [18]
- Demonstrated that the degree of occlusion after the initial treatment is a strong predictor of subsequent rupture, which justifies attempts to completely occlude aneurysms.
SUMMARY POINTS
Interventional neuroradiology is a rapidly expanding field and the anaesthetist plays a central role in the achievement of good patient outcomes. Anaesthesia for imaging and INR presents unique challenges, highlighting the importance of careful planning and patient assessment. The maintenance of peri-procedural physiological stability and recognition and management of potential complications is fundamental to providing sound anaesthetic care. Trainees should seek to learn from experienced anaesthetists and develop their competence and confidence in anaesthesia for neuroradiology.
- Careful patient assessment
- Maintenance of physiological stability
- Recognition and swift management of complications
ANSWERS TO QUESTIONS
Question 1: T, T, T, T
The indications for interventional neuroradiology are increasingly varied and a wide variety of neurological conditions can now be managed in the neuroradiology suite.
Question 2: T, F, T, T
Invasive arterial BP, UO and ACT are usually required for patients undergoing interventional procedures. Rapid management of blood pressure is important to manage physiological stability and maintain CPP. Large volumes of contrast may be used and there is the potential for patients to become insipid necessitating urinary catheterisation. Patients are usually anticoagulated and ACT monitoring will be required. A central line is not necessarily indicated.
Question 3: F, T, T, F
There is insufficient evidence to support use of LMA for patients undergoing interventional neuroradiological procedures. Procedures can be long, airway access restricted, and spontaneous ventilation does not allow for manipulation of ETCO2. However, the use of an LMA to facilitate smooth extubation and emergence is supported.
WEBLINKS
http://www.rcoa.ac.uk/document-store/anaesthetic-services-remote-sites
http://www.aagbi.org/sites/default/files/magnetic_resonance_unit_2010.pdf
WEBLINKS FOR IMAGES
http://radiopaedia.org/articles/subdural-haemorrhage
http://ookaboo.com/o/pictures/picture/25148954/CTscan_of_the_brain_with_an_MCA_infarct
http://radiopaedia.org/cases/meningioma-sphenoid-wing-typical
http://commons.wikimedia.org/wiki/File:Cerebral_Angiogram_Lateral.jpg#filelinks
REFERENCES and FURTHER READING
- Molyneux, A.J., Kerr, R.S., Yu, L.M., Clarke M., Sneade, M., Yarnold, J.A., & Sandercock, P. International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. The Lancet 2005:366(9488):783-785.
- Pinnock, Colin A., Ted Lin, Robert P. Jones, and Tim Smith, eds. Fundamentals of anaesthesia. Cambridge University Press, 2002 (page 441)
- Varma, M. K., Price, K., Jayakrishnan, V., Manickam, B., & Kessell, G. Anaesthetic considerations for interventional neuroradiology. British Journal of Anaesthesia 2007;99(1),75-85.
- Kramer, Andreas H., Derek J. Roberts, and David A. Zygun. Optimal glycemic control in neurocritical care patients: a systematic review and meta-analysis. Critical Care 2012;16(5):R203.
- Anastasian, Z. H., Strozyk, D., Meyers, P. M., Wang, S., & Berman, M. F. Radiation exposure of the anesthesiologist in the neurointerventional suite. Anesthesiology 2011;114(3), 512-520.
- Reddy, U., White, M. J., & Wilson, S. R. (2012). Anaesthesia for magnetic resonance imaging. Continuing Education in Anaesthesia, Critical Care & Pain 2012;12(3):140-144.
- Brinjikji, W., McDonald, J. S., Kallmes, D. F., & Cloft, H. J. (2013). Rescue treatment of thromboembolic complications during endovascular treatment of cerebral aneurysms. Stroke 2013;44(5):1343-1347.
- Schulenburg E, Matta B. Anaesthesia for interventional neuroradiology. Current Opinion in Anesthesiology 2011;24:426-432
- Allman, Keith, and Iain Wilson, eds. Oxford handbook of anaesthesia. Oxford university press, 2011.
- Dorairaj IL, Hancock SM. Anaesthesia for interventional neuroradiology. Continuing Education in Anaesthesia, Critical Care & Pain 2008;8(3):86-89
- de Rooij, N.K., Rinkel, G.J., Dankbaar, J.W., & Frijns, C.J. Delayed Cerebral Ischemia After Subarachnoid Hemorrhage: A Systematic Review of Clinical, Laboratory, and Radiological Predictors. Stroke 2013 44(1), 43-54.
- Washington, Chad W., and Gregory J. Zipfel. Detection and monitoring of vasospasm and delayed cerebral ischemia: a review and assessment of the literature. Neurocritical care 2011;15(2):312-317.
- Rowland, M. J., Hadjipavlou, G., Kelly, M., Westbrook, J., & Pattinson, K.T.S. Delayed cerebral ischaemia after subarachnoid haemorrhage: looking beyond vasospasm. British journal of anaesthesia 2012;109(3),315-329.
- Patel, S., and Appleby, I., Anaesthesia for interventional neuroradiology. Anaesthesia & Intensive Care Medicine 2013;14(9):387-390.
- Barua, E., Johnston, J., Fujii, J., Dzwonczyk, R., Chiocca, E., & Bergese, S. Anesthesia for brain tumor resection using intraoperative magnetic resonance imaging (iMRI) with the Polestar N-20 system: experience and challenges. Journal of clinical anesthesia, 2009;21(5):371-376.
- Yadav, J. S. Protected carotid-artery stenting versus endarterectomy in high-risk patients N Eng J Med 2004;351:1493-1501
- Mohr, J.P., Parides, M.K., Stapf, C., Moquete, E., Moy, C.S., Overbey, J.R et al. Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicentre, non-blinded, randomised trial. The Lancet 2014;383(9917):614-621
- Johnston, S.C., Dowd, C.F., Higashida, R.T., Lawton, M.T., Duckwiler, G.R., & Gress, D.R. Predictors of Rehemorrhage After Treatment of Ruptured Intracranial Aneurysms The Cerebral Aneurysm Rerupture After Treatment (CARAT) Study. Stroke 2008;39(1) 120-125.



