Intensive Care Medicine
QUESTIONS:
- What is the effect of a tracheostomy on anatomical deadspace and work of breathing?
- What are the advantages and disadvantages of early tracheostomy in cases where prolonged mechanical ventilation is predicted?
- Which patients should have dual cannula tracheostomy tubes inserted?
- When and why should tracheostomy tubes be routinely changed?
INTRODUCTION
Tracheotomy refers to the surgical opening of the trachea while tracheostomy refers to the creation of a stoma at the skin surface, which leads to the trachea.
The history of surgical access to the airway is largely one of condemnation. This technique of slashing the throat to save a life was known as semi-slaughter. However, once the technique was perfected as a last resort in largely hopeless cases of diphtheria, the opportunities it offered for medical heroism ensured its place in the surgical armamentarium, such that Fabricius could write in the 17th century, “This operation redounds to the honor of the physician and places him on a footing with the Gods”. Tracheostomy was performed in ancient Egypt and is one of the oldest surgical procedures.1 Chevalier Jackson described the principles of tracheostomy at the beginning of the 20th century.2
Tracheostomies may be temporary or permanent. A temporary tracheostomy may be used as a permanent tracheostomy however there will still be a communication between the pharynx and the lower airway via the larynx.
INDICATIONS FOR TRACHEOSTOMY
- Upper airway obstruction. This is no longer the most common indication for tracheostomy, owing to the improvement in designs of intubating laryngoscopes and alternative management strategies. Upper airway obstruction may be caused by swelling resulting from burns, anaphylaxis, trauma or infection or as a direct result of facial trauma or fractures.
- Prolonged ventilation. This is now the most common indication for tracheostomy, certainly in the intensive-care setting. A tracheostomy is more secure and decreases dead space compared to an oral endotracheal tube, which facilitates weaning from ventilation.
The timing of tracheostomy for this purpose is still controversial (see comments regarding the TracMan study later in the article). - To provide pulmonary toilet and/or to protect the airway. Tracheostomies may sometimes be performed for conditions associated with excessive tracheo-bronchial secretions requiring regular secretion clearance by suction. Examples are congestive cardiac failure, bulbar palsy, infections or neurological conditions where cough and swallow are impaired.
- As part of another procedure, for example, head and neck surgery.
EFFECTS OF A TRACHEOSTOMY
- The larynx is bypassed and so the patient is unable to phonate (unless a valved device is used; see later)
- There is decreased anatomical and respiratory dead space, decreasing the work of breathing
- There is loss of the humidification and filtration function of the nasal mucosa
- There is an increased risk of respiratory tract infection
- There is a redundant area above the tracheal opening and below the larynx in which mucus can accumulate and fall back into the lungs.
- A foreign body reaction can occur causing local inflammation.
TIMING OF TRACHEOSTOMY FOR PROLONGED VENTILATION CASES
The timing of tracheostomy remains an issue of debate. In a study of tracheostomy in mechanically ventilated adult ICU patients, Terragni et al found no statistically significant difference in the rates of ventilator-associated pneumonia with early tracheotomy (after 6-8 days of laryngeal intubation) versus late tracheotomy (after 13-15 days of laryngeal intubation).3
Meanwhile, a large, retrospective cohort analysis including nearly 11,000 critically ill patients evaluated the impact of tracheostomy timing on mortality. The authors found a slight overall improvement in survival in patients who underwent tracheostomy within the first 10 days of intubation.4
The TracMan study was carried out in the United Kingdom to assess the impact of early (day 1-4 of ICU admission) versus late (day 10 or later) tracheostomy.5 The study included 909 patients from 87 UK hospitals who were expected to stay 7 days or more in the ICU, between March 2006 and December 2008. Patients were randomised to early (n=455) or late (n=454) tracheostomy. Patient characteristics were similar across both groups, with respiratory failure the most common cause of admission to the ICU.
There was no significant difference in mortality between the early and late tracheostomy groups at 30 days (139 versus 141 deaths) or at 2 years post randomisation, with a 74% follow up rate. There was also no significant difference in ICU or hospital length of stay and no significant difference in antibiotic use. However, mean days of sedation were predictably reduced – to 6.6 days in the early group compared with 9.3 days in the late group.
At the study’s presentation, at the 29th International Symposium of Intensive Care and Emergency Medicine, the lead author stated the following: “If you had 100 patients requiring tracheostomy, doing it early results in 2.4 days less sedation overall, but you would perform 48 more, with 3 more procedural complications and no effect on mortality or ICU length of stay.”
TECHNIQUES FOR INSERTION
Tracheostomy may be performed using a percutaneous or an open surgical technique.
Percutaneous tracheostomy is performed by anaesthesiologists or intensivists, usually under fibreoptic bronchoscopic guidance. Open surgical tracheostomies are performed by ENT surgeons and in some countries, trauma surgeons. Percutaneous tracheostomy was first described in the late 1950s and 1960s but received widespread acceptance following introduction of commercial kits.
Two initial techniques were described – a serial dilatational technique described by Ciaglia et al6 in 1985 and a guidewire dilating forceps (GWDF) method described by Griggs and colleagues in 1990. In 2000, Byhahn et al modified the Ciaglia technique by introducing the Blue Rhino7 (Figure 1). This hydrophilically coated, curved dilator allows progressive dilatation of the tracheal stoma in a single step, reducing the risk of posterior tracheal wall injury, intraoperative bleeding and the adverse effect on oxygenation during repeated airway obstruction by sequential dilators.
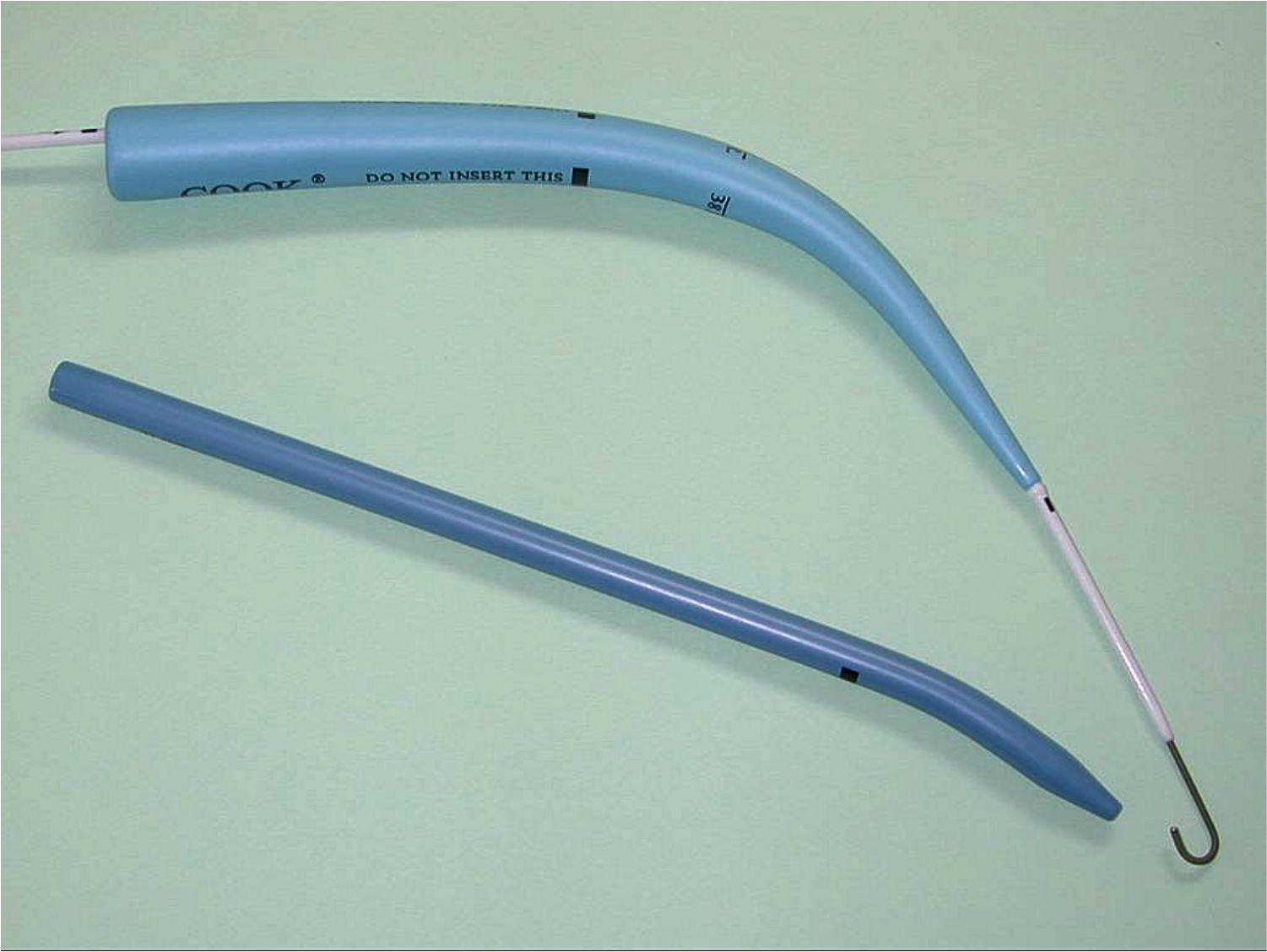
Figure 1. The Blue Rhino single stage dilator (Cook Medical).
Percutaneous Tracheostomy Insertion
Many commercial kits are available, all employing a Seldinger guidewire technique for tracheostomy tube insertion. Techniques may vary slightly, depending upon operator preference and experience but the basics principles of insertion are described below.
Percutaneous Tracheostomy Insertion – basic principles
- Contraindications for percutaneous tracheostomy are reviewed (see below).
- Consent should be obtained where possible – patients will frequently not be in a position to give consent. While the issue may be discussed with next of kin, the decision to proceed is made in the patient’s best interests.
- Platelet and coagulation assays are checked and the patient’s blood should be ‘group and saved’ as bleeding may be a complication of this procedure. Intensive Care Society (UK) guidance recommends the use of a pre-procedure ultrasound to confirm the absence of large blood vessels directly over the planned site of insertion.
- Nasogastric tube feeding should be stopped for at least two hours and the nasogastric tube should be aspirated to reduce any chance of aspiration of gastric contents.
- Equipment is assembled. This will include the percutaneous tracheostomy set, equipment for emergency reintubation, a fibreoptic bronchoscope, drapes and sterile skin preparation and a local anaesthetic solution containing a vasocontrictor such as 1% lidocaine with 1:200 000 epinephrine.
- The procedure requires two clinicians – one to operate the bronchoscope and one to perform the tracheostomy insertion – and assistance from a member of nursing staff familiar with the procedure and the environment to assist with the management of complications.
- Sedation, analgesia and muscle relaxation need to be administered, common examples being propofol, fentanyl and atracurium.
- The patient is positioned in a way that allows the neck to be extended. This can be achieved by using pillows or towels placed behind their shoulders. The patient should be fully monitored and ventilated with 100% oxygen during the procedure.
- The cuff of the patient’s endotracheal tube is deflated and the tube is withdrawn under direct vision until the tip is just distal to the patient’s vocal cords. This removes the endotracheal tube from the operative site. Alternatively, the tube may be exchanged for a laryngeal mask airway (LMA) providing that the patient can be ventilated safely using the device. This may not be the case in patients requiring higher a FiO2, high levels of PEEP or high inspiratory airway pressures.
- The patient’s anatomy should be marked, including the intended level of insertion of the tracheostomy. The recommended level is between the second and third tracheal rings but should be adapted to the patient’s anatomy. Insertion near the cricoid cartilage and first tracheal ring is thought to increase the risk of subsequent tracheal stenosis whilst insertion at too low a level increases the risk of erosion of thoracic inlet great vessels.
- A full aseptic technique should be used including the use of gown, gloves, hat, and mask and eye protection. The patient’s skin should be prepared with an appropriate antiseptic cleaning solution and the operator should check the open kit.
- Following infiltration with the local anaesthetic and vasoconstrictor solution, a small transverse skin incision is made followed by blunt dissection with forceps until the tracheal rings are felt. Cannulation of the trachea can then be performed using the 14G needle and cannula provided in the kit. This may be confirmed by the ability to aspirate air through the needle but presence of the cannula in the trachea must be confirmed visually via the bronchoscope before proceeding further.
- These next steps should all be performed under direct vision via the bronchoscope. The guidewire is passed into the trachea where it should be able to move freely through the cannula. Depending upon the kit being used, there may be an intermediate rigid dilator, which is passed over the guidewire. Next, the curved dilator is passed over the guidewire, into the trachea, as far as the thick black line, in one smooth movement. An example of a curved dilator is the Blue Rhino (Cook Medical). Such dilators are coated in a lubricant that is activated by contact with saline or sterile water.
- The curved dilator is then removed and the tracheostomy tube, complete with introducer, is then inserted into the trachea over the guidewire.
- The introducer is removed, the tube cuff is inflated and the ventilation circuit is connected. Confirmation of correct placement is achieved by assessment of chest movement, auscultation and the use of capnography.
- The tracheostomy is secured using the supplied tapes and the procedure is documented in the notes.
- After the procedure, a chest X-ray is performed to exclude pneumothoraces. Ventilation settings can be returned to pre-operative values as soon as sedation and neuromuscular blockade allows.
CAUTIONS AND RELATIVE CONTRAINDICATIONS FOR PERCUTANEOUS TRACHEOSTOMY 8
- Emergency airway access
- Difficult anatomy
- Morbid obesity with short neck
- Limited neck movement
- Cervical spine injury – suspected or otherwise
- Aberrant blood vessels
- Thyroid or tracheal pathology
- Moderate coagulopathy
- Prothrombin time or activated partial thromboplastin time greater than 1.5 time the reference range
- Platelet count less than 50000 / mcl
- Significant gas exchange problems: e.g. PEEP > 10 cm H2O or FiO2 greater than 0.6
- Evidence of infection in the soft tissues of the neck at the insertion site
- Age less than 12 years.
Needless to say, these relative contraindications are subject to the experience and clinical judgement of the operator and are not set in stone.
CARE OF THE TRACHEOSTOMY
Changing tracheostomy tubes
Whilst changing a tracheostomy tube can be hazardous, failing to change one when required also carries risks. Guidance from the Intensive Care Society points out that recommendation regarding the timing of tube changes is inconsistent and not evidence based.8
It is recommended that tracheostomies without inner tubes be changed every 7-14 days, with the frequency decreasing as the stoma becomes better-formed and pulmonary secretions decrease. EEC guidance, from 1993, states that tracheostomies with inner tubes may be left in place for up to thirty days.
The first change should not occur within 72 hours of the tracheostomy being sited and ideally not for 7 days after a percutaneous insertion. This is to allow for the formation of a more reliable ‘track’ for the new tube to pass through. Emergency airway equipment, including a smaller tracheostomy tube, and emergency drugs should be immediately available during the change.
The tracheostomy tube may be changed over a soft suction or airway exchange catheter or soft tipped Ryle’s tube. The use of a rigid gum-elastic bougie for this purpose may increase the risk of creating a false passage (i.e. the new tracheostomy comes to lie next to rather than within the trachea). If a soft tipped Ryle’s tube or similar is used, it may be reassuring to see fogging within that tube with respiration. This will help to confirm that the exchange tube is in the airway and not in a false passage prior to passing the new tracheostomy tube. Alternatively, the track may be gently dilated with a gloved little finger.
There should be a low threshold for suspicion of erroneous placement if it is difficult to ventilate the patient. If difficulty is encountered in replacing the tracheostomy tube, the clinical need for a tube must be re-assessed. If in doubt, re-intubation with an oral endotracheal tube may be required.
Humidification
Cold and unfiltered air is an irritant when inhaled and can lead to increased production and viscosity of secretions. This can be uncomfortable for the patient as well as causing tracheal mucosal keratinisation.
The increasingly viscous secretions will be difficult to clear, causing sputum retention, atelectasis, impaired gas exchange and even life threatening blockage of the tracheostomy tube. It is therefore essential that inhaled oxygen is appropriately humidified using conventional techniques such as heat and moisture exchange (HME) filters or heated water baths.
Nutrition
It is conventional to feed intubated, ventilated patients enterally unless there is a good reason not to. This is usually via a nasogastric or nasojejunal tube but it may be possible for patients with tracheostomies to be fed orally. However, swallowing is still adversely affected by the presence of a tracheostomy tube, which has a tendency to limit normal movement of the larynx. In addition, the inflated cuff causes a sense of pressure in the upper oesophagus and the difficulty that occurs with swallowing may result in an increased risk of aspiration of food into the lungs.
Patients may be fed orally, with the cuff inflated or partially deflated, but staff must be alert to signs of aspiration, such as coughing, increased secretions and impaired gas exchange. It is prudent to commence with sips of water and some form of swallowing assessment.
FEATURES OF TRACHEOSTOMY TUBES
The important features of a tracheostomy tube are as listed below:
- Diameter: The tracheostomy tube has an inner and an outer diameter. The size of the tracheostomy tube refers to the internal diameter (ID) and ranges from 5.0mm to 9.0mm in adult practice. The size quoted is for the outer tube, for single lumen devices, and the inner tube, for double lumen devices but only if the internal cannula is required for connection to a breathing circuit. See Fig 2.
- Cuff: The cuff reduces aspiration and leakage of air during anaesthesia and positive pressure ventilation. The tube can be changed to an uncuffed tube when mechanical ventilation is not required or when there is deemed to be minimal risk of aspiration. Whilst most patients can be weaned by simply deflating the cuff, it may still restrict airflow around the tube and changing to an uncuffed or smaller tube may help.
- Inner tube: The inner tube has the safety advantage of being easily and quickly removed to relieve life threatening obstruction due to blood clots or secretions. This is balanced by the slight reduction in internal diameter, which can result in an increased work of breathing and lengthened weaning. It is recommended that dual cannula tubes should be used whenever possible because of the safety advantage. See Fig 3.
- Fenestration: Fenestrations maybe be single or multiple and are sited at the site of maximum curvature of the tracheostomy tube. These aid phonation by allowing airflow through the fenestration into the larynx. The fenestration needs to be well placed for each patient’s anatomy in order to work well. Simply deflating a cuff may be an alternative approach in patients who do not require positive pressure respiratory support. See Fig 4.
- Flexibility: Flexible or reinforced tracheostomy tubes resemble reinforced endotracheal tubes and can be used in patients where a rigid tube may lie at an angle and cause abrasion or tube obstruction as its lumen abuts the posterior tracheal wall.
- Adjustable flange: The length of the tube from the tracheal lumen to the position of the stoma on the exterior can be adjusted in this variation of the tracheostomy tube. This is useful in obese patients or those with local tissue swelling, where the soft tissue depth is increased. See Fig 5.
- Subglottic suction: Some newer tracheostomy tubes include a subglottic suction port, the aim of which is to try and reduce the incidence of ventilator-associated pneumonia.
- Speaking valve: Speaking valves (like the Passy Muir valve) are one-way valves that are designed to be used with fenestrated tracheostomy tubes or unfenestrated tubes (with the cuff deflated). They allow inspiration but not expiration. Hence the expired air is forced through the larynx allowing the patient to phonate. See Fig 6.

Table 1. Table showing different types of commonly used tracheostomy tubes.
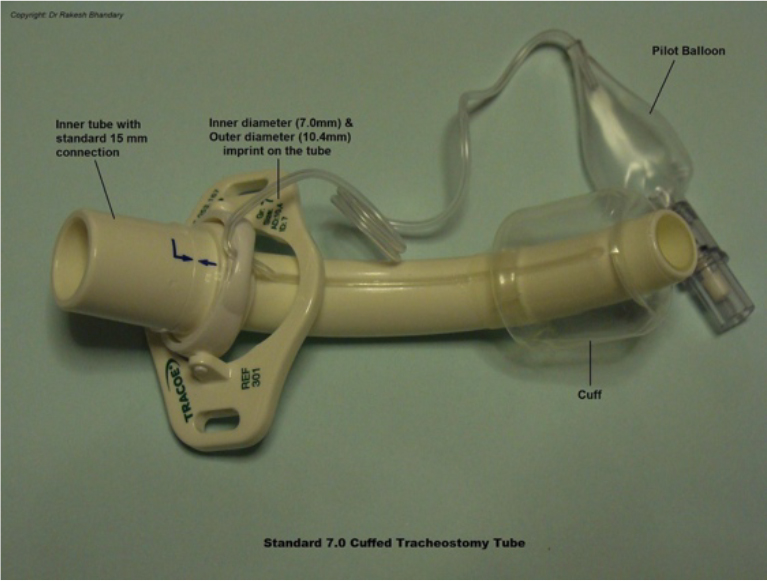
Figure 2. Figure illustrating a standard, single cannula, size 7.0 tracheostomy tube.
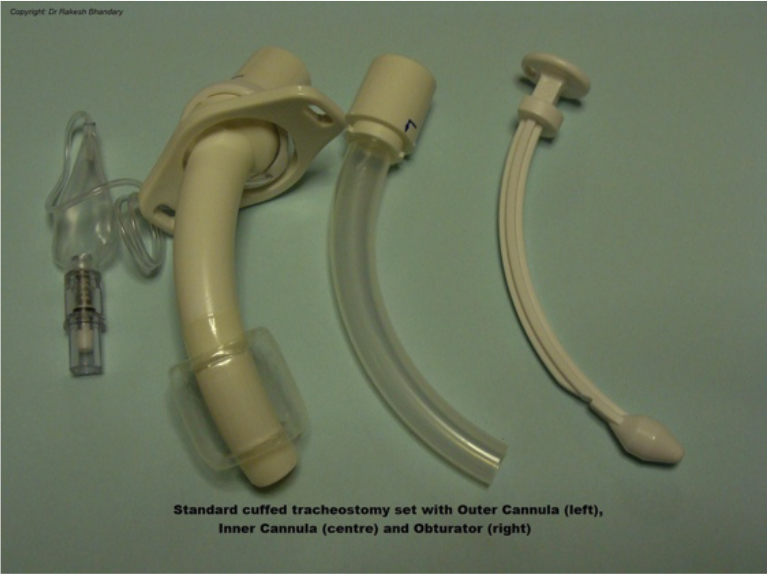
Figure 3. Figure illustrating a standard, dual cannula tracheostomy tube.
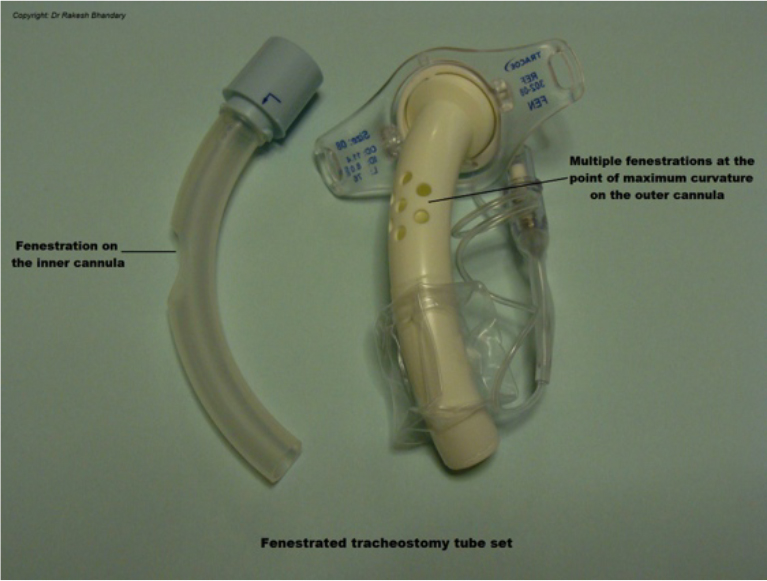
Figure 4. Figure illustrating a fenestrated dual cannula tracheostomy tube.
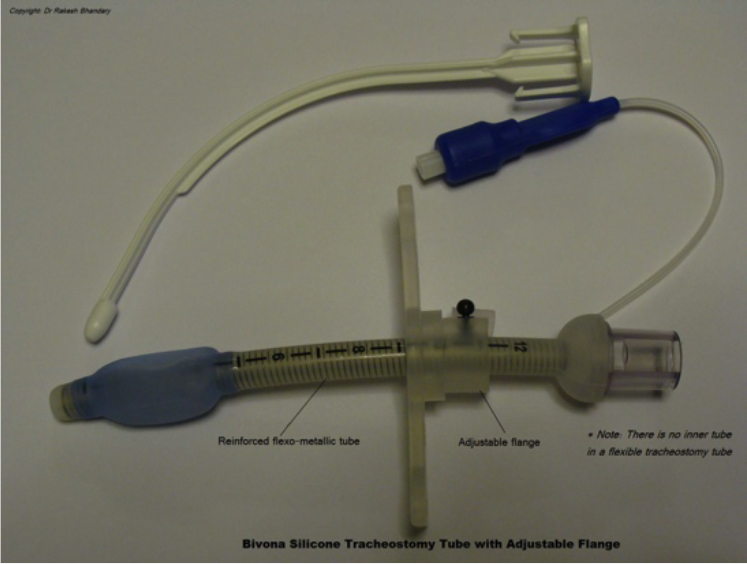
Figure 5. Figure illustrating an adjustable flange, flexible tracheostomy tube

Figure 6. Figure illustrating a Passy Muir speaking valve. This is inserted into the external orifice of the tracheostomy tube.
COMPLICATIONS OF TRACHEOSTOMY
Complication rates range between 4% and 31% for percutaneous tracheostomy and 6% to 66% for surgical tracheostomy.9 Kost in 2005 reported on the use of percutaneous tracheostomy in 500 consecutive intubated adults in the intensive care unit.10 When this procedure was performed in conjunction with bronchoscopy, she stated the complication rate as acceptably low (9.2%). No serious complications (pneumothorax, pneumomediastinum, death) occurred. The 2 most common complications were oxygen desaturation in 14 patients (defined as a drop [even transient] to less than 90%) and bleeding in 12 patients (when intervention was required to control the bleeding).
This is one of many studies that demonstrate a favourable complication rate for the percutaneous method compared to the surgical method.
The complications of tracheostomy can be grouped as immediate, intermediate and long-term and are listed below.
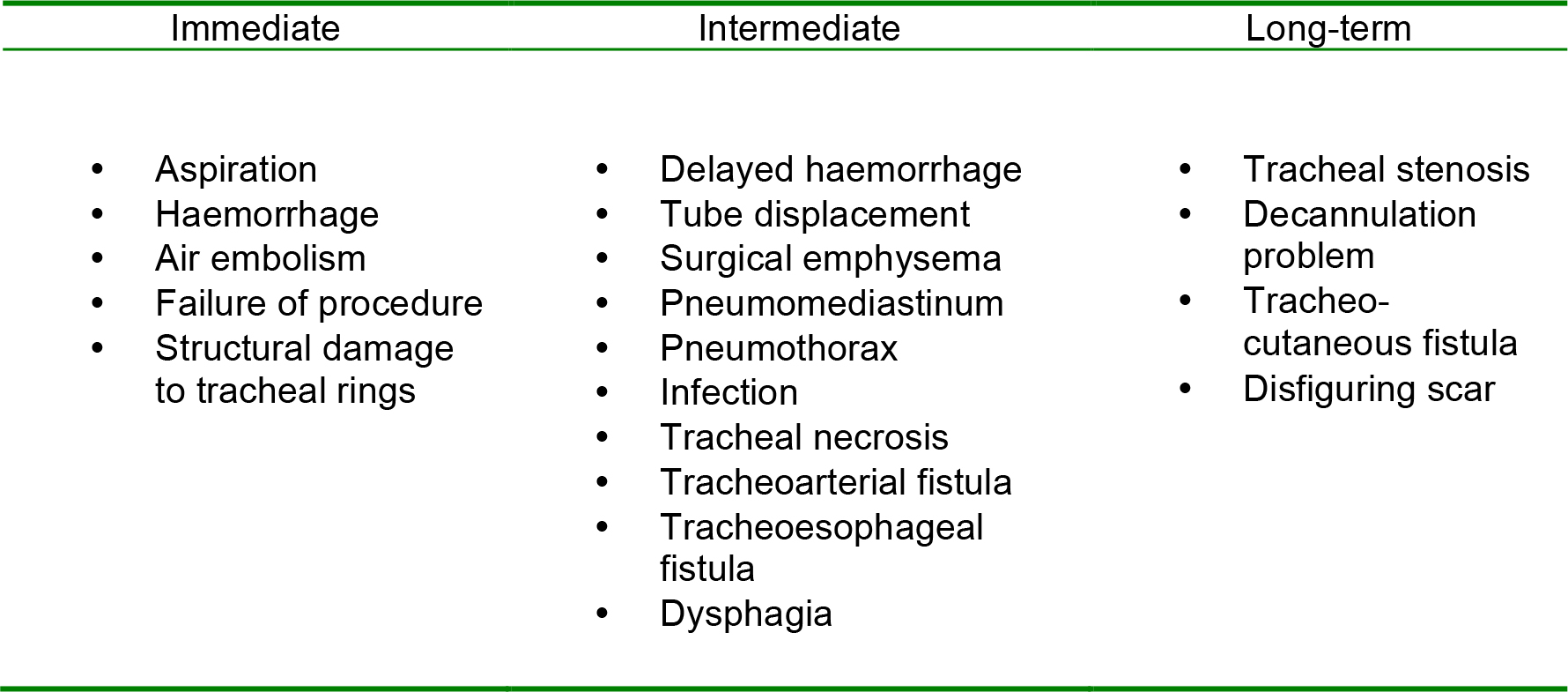
Table 2. Table of complications of tracheostomy
Immediate or early complications
Bleeding is the most common and the most commonly fatal complication of tracheostomy. The incidence is higher with an emergency procedure. Intraoperative bleeding is commonly due to cut edges of the vascular thyroid gland, anterior jugular vessels or inferior thyroid vessels; bleeding in the immediate post-operative period may be exacerbated by emergence from anaesthesia and hypertension. Vasoconstrictors infiltrated during the procedure may also be wearing off.
Although this may necessitate a return to the operating room, bleeding may be controlled with pressure, local packing – perhaps with dressings or Kaltostat soaked in dilute adrenaline, sutures or hypertension control.
Major bleeding can cause cardiovascular compromise of course, but may also cause respiratory difficulties, particularly if clots form and obstruct any part of the airway. In this situation, control of the airway should be achieved by conventional intubation, making sure that the cuff of the endotracheal tube is below the stoma. This may require an uncut tube. Surgical exploration is then necessary.
Other early, recognised complications include pneumothorax, which may result from direct injury to pleura, pneumomediastinum & injury to local structures like recurrent laryngeal nerve, cartilages & oesophagus.
Malposition of the tracheostomy is always possible but should, in theory, be minimised by the use of fibre-optic bronchoscopy for percutaneous insertions.
Intermediate complications
Delayed haemorrhage maybe due to displaced blood clots or ligatures, infective erosion into a blood vessels or rarely from a tracheo-innominate fistula, which may result from a long tube or low tracheostomy. As with an endotracheal tube, the tracheostomy tube may also cause tracheal mucosal necrosis at the level of the cuff.
The tube may also erode into the surrounding structures leading to tracheo-oesophageal fistula, pneumothorax or pneumomediastinum. Surgical emphysema may also be seen due to tight closure of tissue around the tube, tight packing material around the tube, or the false passage of the tube into pretracheal tissue.
Delayed complications
Tracheal stenosis may occur at the level of the stoma due to collapse of the cartilaginous ring or at the level of the tube cuff due to mucosal necrosis & fibrosis. Modern high-volume, low-pressure cuffs have reduced the incidence of tracheal stenosis.
A tracheal granuloma may develop or healing may be delayed leading to a persistent tracheo-cutaneous fistula or sinus. Sometimes, patients fail plugging trials or even decannulation for no apparent reason. Possibilities to consider include an obstructing granuloma previously held out of the way with the tube, bilateral vocal cord paralysis, fractured cartilage and anxiety. Evaluation should include fibreoptic laryngoscopy and bronchoscopy through the stoma.
Emergency management of a displaced or blocked tracheostomy tube
This complication can be fatal and it is important that those caring for patients with tracheostomies are alert to its clinical presentation and are familiar with a plan for its management.
The Royal College of Anaesthetists and Difficult Airway Society recently published the results of the National Audit Project 4 – Major Complications of Airway Management in the United Kingdom.11 In its Executive Summary, the authors made the following comment on the management of displaced tracheostomies:
“Displaced tracheostomy, and to a lesser extent displaced tracheal tubes, were the greatest cause of
major morbidity and mortality in ICU. Obese patients were at particular risk of such events and
adverseoutcome from them. All patients on ICU should have an emergency re-intubation plan.”
An example of an emergency management plan is illustrated in Appendix 2 of the audit’s report whilst another example is provided below. (Figure 7).
Both of these algorithms share some common themes. Recognising that this can become a rapidly fatal complication, emphasis is placed on recognising the clinical picture of a patient with a displaced or blocked tracheostomy tube and calling for senior help early.
Advice is given on how and when to attempt to replace the tracheostomy tube but if in any doubt, the tube should be removed and attempts should be made to maintain and secure the airway from above using a facemask, supraglottic airway devices and ultimately, oral endotracheal intubation.
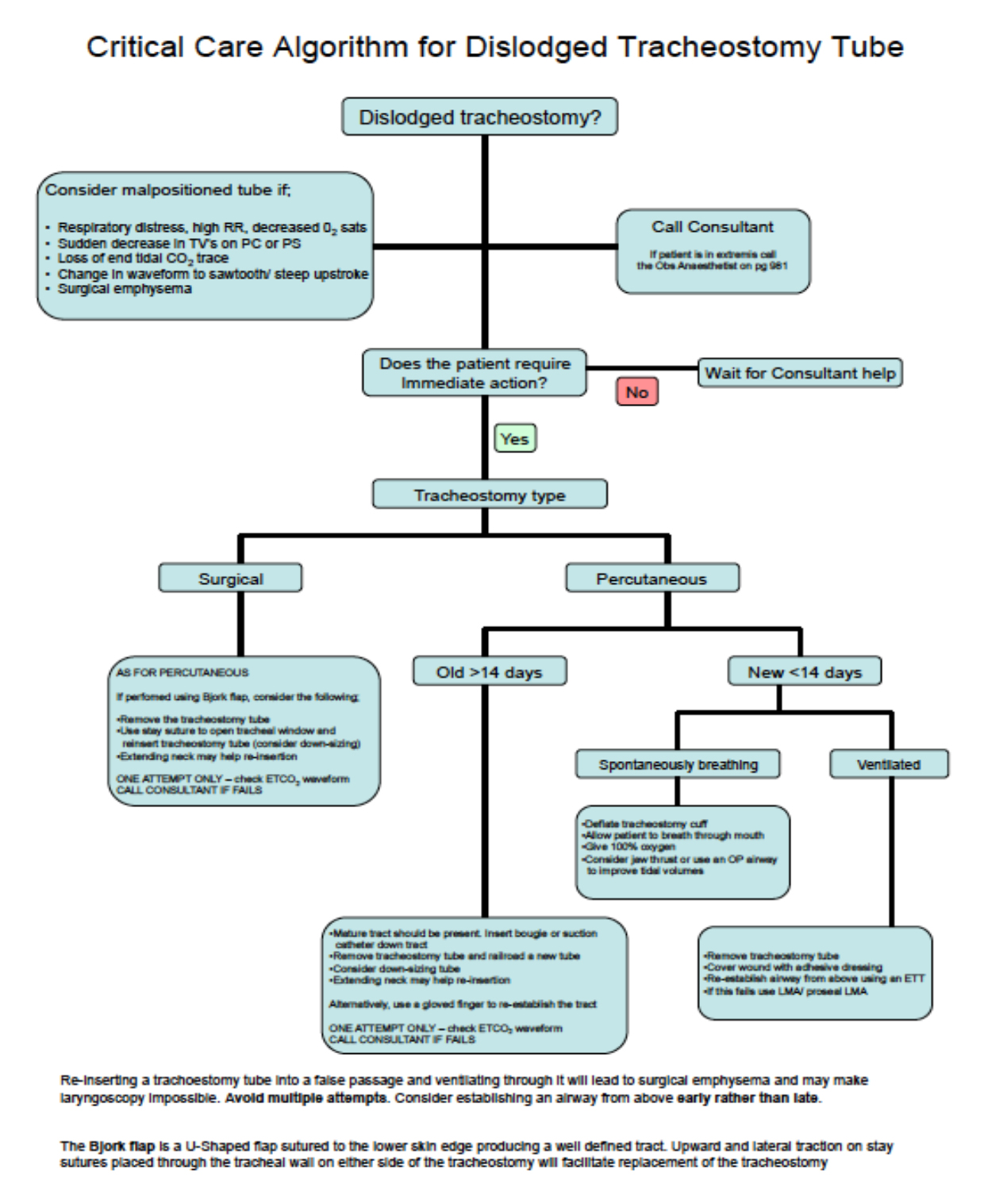
Fig 7. An algorithm for managing a displaced tracheostomy tube. Reproduced with kind permission of Dr Peter Ford, Dept of Anaesthesia, Royal Devon & Exeter NHS Foundation trust, UK.
WEANING AND DECANNULATION
The tracheostomy tube should be removed as soon as is feasible. Decannulation allows the patient to resume breathing through the upper airway and reduces dependence (psychological and otherwise) on the lesser resistance of the tracheostomy tube.
There are many ways of assessing adequate breathing around the tracheostomy tube. Patients can be trialled with increasing periods of cuff deflation. This allows patients to become re-accustomed to swallowing more normally and to having to clear their own secretions.
Alternatively, an occlusion cap may be used which completely blocks the tracheostomy tube. Of course this must be used with a fenestrated tube or an unfenestrated tube with the cuff deflated, and this greatly increases the work of breathing due to the increased airway resistance. It will be harder for patients to breathe in this situation than without the tracheostomy in place and this must be taken into account when interpreting the success or failure of such a trial.
Decannulation can be carried out when:
- The patient is not dependent on ventilatory support and has an adequate respiratory reserve (dead space will be increased without the tracheostomy tube)
- The patient is able to cough and swallow effectively and manage their own secretions whilst being able to protect their own airway
- The patient is able to cough and clear his / her tracheal secretions
- Patient can tolerate cuff deflation or capping of the tracheostomy tube.
Decannulation itself can be performed in the morning, with a rested patient and daylight hours in which to review their progress. The tube is removed and the stoma is covered with a semi-permeable dressing. The patient is encouraged to gently press over this defect with whilst speaking or coughing.
They should be subsequently be monitored for signs of respiratory distress. Equipment and expertise to re-secure the airway, either via the stoma or via oral intubation, should be available.
PERMANENT STOMA, TRACHEO-OESOPHAGEAL PUNCTURE AND PROSTHETIC SPEECH VALVE
Even though complex laryngectomies are carried out in hospitals providing ENT services, some patients may present themselves to hospitals that do not offer this service. As a result, these patients may present to staff that are less familiar with permanent stomas.
The basic options post-total largyngectomy for speech rehabilitation include an artificial larynx, oesophageal speech and tracheo-oesophageal speech. Tracheo-oesophageal speech provides the advantage over the other two options that air supply for speech is pulmonary, phonation sounds natural, and voice restoration occurs within 2 weeks of surgery.
During total laryngectomy, a surgical fistula is created between the oesophagus and the trachea – a primary tracheo-oesophageal puncture (TEP). Alternatively TEP maybe performed a few weeks or even months after total laryngectomy – secondary TEP. The TEP is kept patent in the immediate postoperative period using a Foley’s self retaining catheter or feeding tube, which has the added benefit of enabling enteral feeding. Two to three weeks post-operatively, an appropriately sized Bloom-Singer valve is inserted.
A Bloom-Singer valve is a hollow, 16- or 20-French, silicone tube that has a one-way flap valve positioned within its proximal tip. The valve serves two purposes; first, it allows patient to phonate by allowing pulmonary air to pass through the valve, into the pharynx and out of the mouth and second, it prevents saliva and oral secretions from being aspirated into the tracheo-bronchial tree from the pharynx. To phonate, the patient inhales air through the permanent stoma, occludes the permanent stoma with the thumb and exhales. The occluded stoma diverts air through the Bloom-Singer prosthesis and up the oesophagus to the mouth. Vibration of opposed mucosal surfaces along the oesophagus and pharynx produces a variably husky or hoarse quality voice that is articulated by the tongue, lips, and teeth into intelligible speech.
The following complications maybe seen with a Bloom-Singer valve:
- Candida infection in and around the prosthesis
- Leakage through the valve due to defective one-way valve
- Peri-prosthetic leakage
- Occlusion of the prosthesis
- Inadvertent displacement and aspiration
What to do if a patient presents with displaced Bloom-Singer Valve
Two main problems are encountered if a patient presents with a displaced Bloom-Singer valve prosthesis. First, oral secretions may be aspirated into the tracheo-bronchial tree and secondly, the TEP may be seal spontaneously, warranting another surgical procedure. If these patients present to a hospital that does not provide ENT services, a self-retaining Foley’s catheter, equivalent in size to the B-S valve, can be introduced through the TEP into the oesophagus under local anaesthetic spray. The balloon is inflated with 3 ml of air, gently retracted and taped to the side of the neck, while awaiting inter-hospital transfer. If the valve has been aspirated, it can be removed using a fibre-optic bronchoscope.
Answers to questions:
- Bypassing the pharynx and larynx reduces anatomical deadspace. Work of breathing is reduced as a result of breathing through a shorter ‘tube’.
- The timing of tracheostomy in cases of predicted prolonged mechanical ventilation is still controversial. The TracMan study demonstrated a reduction in days of sedation but this was not translated into a reduction in mortality, hospital stay or ICU stay.
- Dual cannula tracheostomy tubes allow staff to quickly, cleanly and safely change tubes for the purposes of cleaning and relieving obstructions caused by clots and secretions. For this reason, they should be used for all patients, unless there is a good reason not to. One example of such a reason might be the use of an adjustable flange, flexible tracheostomy tube in patients whose neck anatomy precludes the use of a standard tube to achieve a ‘good fit’.
- Tracheostomies without inner tubes should generally be changed every one to two weeks. Those with inner tubes can generally be left in situ for up to 30 days. The inner tube can be removed and either cleaned or replaced, depending upon the manufacturer’s recommendations. Tubes are generally changed electively for hygiene purposes.
References:
- Frost E. Tracing the tracheostomy. Ann OtolRhinolLaryngol 1976; 85: 618-24
- Jackson C. Tracheostomy. Laryngoscope 1909; 19: 285-90
- Terragni P, Antonelli M, Fumagalli R, Faggiano C, Berardino M, Pallavicini FB et al. Early vs late tracheotomy for prevention of pneumonia in mechanically ventilated adult ICU patients: a randomized controlled trial. JAMA. Apr 21 2010;303(15):1483-9
- Scales D, Thiruchelvam D, Kiss A, Redelmeier DA. The effect of tracheostomy timing during critical illness on long-term survival. Crit Care Med. Sep 2008;36(9):2547-57
- TRACMAN study. http://www.pslgroup.com/dg/2361ee.htm
- Ciaglia P, Firsching R, Syniec C. Elective percutaneous dilatational tracheostomy. A new simple bedside procedure; preliminary report. Chest. Jun 1985; 87: 715-9
- Byhahn C, Wilke H, Halbig S, Lischke V, Westphal K. Percutaneous tracheostomy: Ciaglia blue rhino versus the basic Ciaglia technique of percutaneous dilational tracheostomy. AnesthAnalg. Oct 2000; 91:882-6
- Standards for the Care of Adult Patients with Temporary Tracheostomy. Standards and Guidelines. The Intensive Care Society, July 2008
- Trottier SJ, Hazard PB, Sakabu SA, Levine JH, Troop BR, Thompson JAet al. Posterior tracheal wall perforation during percutaneous dilatational tracheostomy. Chest 1999; 115:1383-9
- Kost KM. Endoscopic percutaneous dilatational tracheotomy: a prospective evaluation of 500 consecutive cases. Laryngoscope. Oct 2005;115:1-30
- Cook T, Woodall N, Frerk C. Major Complications of Airway Management in the United Kingdom. National Audit Project 4. The Royal College of Anaesthetists and the Difficult Airway Society. March 2011.



