General Topics
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Samonella typhi:
a. Is a gram-positive rod
b. Is also known as Enteric Fever
c. Has an exotoxin
d. Is transmitted by ingestion of food or water contaminated by faecal matter
e. Is found in other animals apart from humans - Regarding diagnosis of typhoid fever:
a. The diagnosis of typhoid cannot be confirmed by clinical presentation
b. Definitive diagnosis can be made by isolation of bacteria from tissues
c. Punch biopsies of rose spots on skin are useful in diagnosis
d. The bacteria can be cultured from blood, urine & stool
e. Bone marrow culture is the least sensitive for diagnosis - Concerning treatment of typhoid fever:
a. Antibiotic therapy should only be based on specific laboratory testing
b. Fluroquniolones are the first- line antibiotic treatment
c. Ceftriaxone and azithromycin are included in treatment of drug resistant strains
d. Steroid therapy in conjunction with antibiotics has been shown to reduce mortality
e. Chronic carriers of typhoid require a 3-week course of antibiotics with amoxicillin, bactrim, or ciprofloxacin
Key Points
- Typhoid enteric fever is a bacterial infection transmitted by fecal-oral route exclusively through human hosts
- Gut perforation is a potentially lethal complication associated with an inflammatory response at Peyer’s Patches
- Definitive diagnosis requires successful culture from blood, stool, skin, or other infected site
- Prompt antibiotics therapy reduces mortality
- Preventative strategies include improved public water and sewage management
- Available vaccinations are currently too limited in efficacy and duration to be widely used in endemic regions
INTRODUCTION
Typhoid fever, otherwise known as enteric fever, is a bacterial infection of the gastrointestinal system that has long plagued humanity. Typhoid is in the family of Enterobacteria, and the genus of Salmonella. These gram-negative, facultative anaerobic bacilli are also flagellated, motile, and non-spore forming. Although the organism lacks an exotoxin to promote illness, it is strongly antigenic and causes an intense inflammatory response in tissues. Typhoid fever is associated with the Salmonella serotypes typhimurium (S. typhi) and paratyphimurium (S. paratyphi).1
While a wide variety of animals can be infected with Salmonella, only humans carry the S. typhi and S. paratyphi serotypes associated with typhoid fever. Thus livestock, household pets, and other animals are neither carriers nor vectors of typhoid fever.2 Humans acquire the disease from other humans through fecal-oral transmission, most commonly in the setting of contaminated water or food. It is not surprising that the greatest disease burden is found among the world’s poorest countries where water and sanitation services are the least robust. Non-typhoidal salmonellosis (NTS) can be spread by animals other than humans and presents as a milder gastroenteritis. NTS is beyond the scope of our discussion of typhoid fever and will not be elaborated on.1
EPIDEMIOLOGY
Typhoid is more common in urban than rural areas. Worldwide there are over 22 million cases with over 200,000 deaths each year, representing a 1-4% mortality rate.3,4 The disease burden of typhoid fever is lowest in the developed world and highest in resource-limited settings. North America and Europe have less than 10 cases per 100,000 people per year, while central and Southeast Asia have 10 times that amount achieving the highest rates in the world. The burden of the disease is difficult to estimate in African countries due to limitations in laboratory testing capacities. 4,5 Outbreaks are more frequent in low resource countries because they are associated with contaminated food and water and with fields fertilized by sewage, street vendors, uncooked fruits and vegetables, sick contacts, limited toilet access, and limited ability to wash hands.
PATHOPHYSIOLOGY
Once consumed, typhoid bacteria cross the epithelial layer of the intestinal wall. They are then quickly consumed by macrophages and transported to the aggregates of lymphoid tissue in the small intestine (Peyer’s patches) where the immune function of the gut is most concentrated. The typhoid bacteria alter host cell signalling and function in such a way that host cells ultimately promote the survival and replication of S. typhi and S. paratyphi.
The incubation stage of a typhoid infection is characterized by the replication and transfer of S. typhi and S. paratyphi from the Peyer’s patches in the gastrointestinal system, through the lymphatics, to the organs of the reticuloendothelial system including the lymph nodes, spleen, bone marrow, and liver. Once in the gallbladder, S. typhi and S. paratyphi are secreted back into the gastrointestinal tract. Having been previously exposed to the organism, the Peyer’s patches respond with an intense inflammatory reaction leading to congestion and clogging of the microcirculation and capillaries with release of lytic lysosomal enzymes and other inflammatory mediators.1,3 This results in varying degrees of necrosis and ulceration of Peyer’s patches of which the clinical manifestation is bleeding and perforation. The terminal ileum is the most common site of perforation, but perforation has also been reported to occur anywhere from the duodenum to the colon including the gall bladder and appendix. 3 (Figure 1)
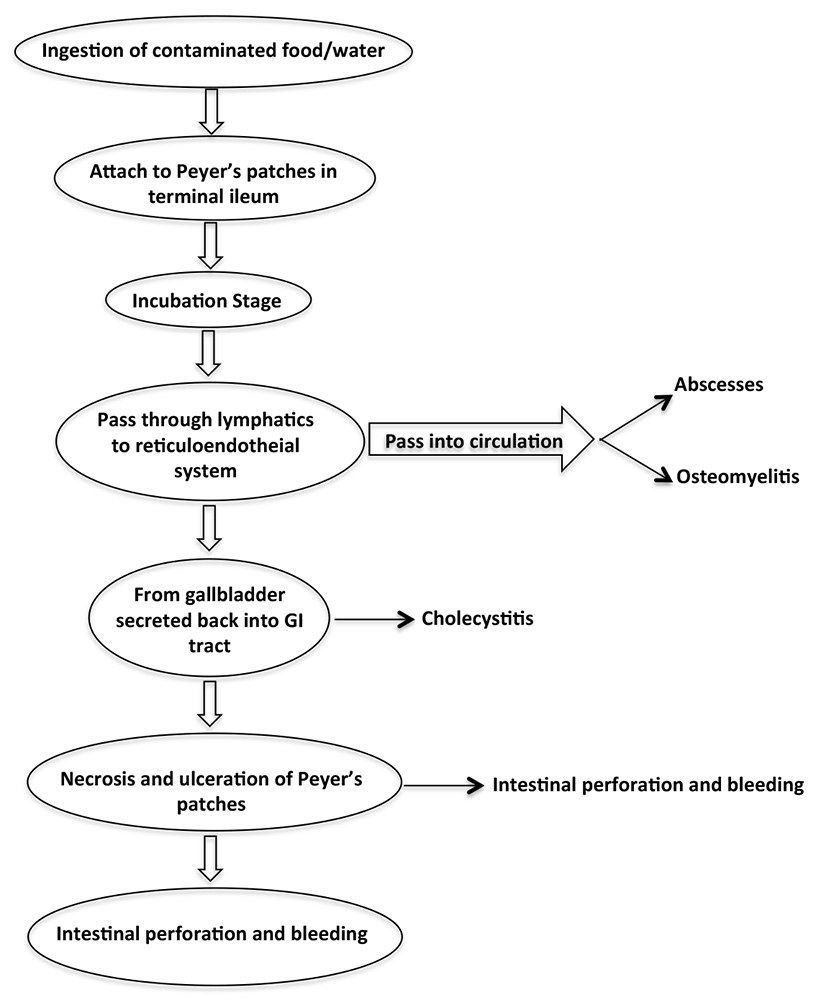
Figure 1. Aetiopathogenesis of typhoid intestinal perforation
Depending on the strength of the host’s immune system and the size of the inoculum, the incubation phase may last 3 days to 3 weeks.1 During this interval, a patient may have no symptoms or vague complaints of fever and abdominal pain. Once the bacterial load reaches a critical mass, an individual is said to have an active typhoid infection.
SIGNS AND SYMPTOMS
Although S. typhi is four times more common than S. paratyphi, in general, the clinical appearance of S. typhi and S. paratyphi infections are virtually indistinguishable. Signs and symptoms of the infection consist mostly of abdominal complaints including fever associated with frontal throbbing headache, nausea, vomiting, abdominal pain, anorexia, diarrhea, constipation, gastrointestinal bleeding and hepatosplenomegaly. Systemic complaints are also common and greater than 75% of patients report having flu-like symptoms. Neurologic problems include meningitis, Guillain-Barre syndrome and a delirium that features muttering and picking at clothes and imaginary objects. Disseminated intravascular coagulation, hemolytic uremic syndrome, renal failure, cardiac failure, and respiratory failure have all been reported as a consequence of severe infection. While no specific constellation of symptoms is pathognomonic of the disease, a transient skin rash, described as rose spots, can be biopsied to confirm the diagnosis. 1
The severity and duration of typhoid fever depends on several host factors including age, integrity of the immune system and GI tract, and alkalization of the stomach. It has been found that a more acidotic environment in the stomach is bactericidal, while a concomitant Helicobacter pylori infection, which increases gastric pH, promotes the disease. 1 In untreated cases that self-resolve, up to 4% of patients will become asymptomatic carriers and continue to shed the bacteria in urine and stool.
COMPLICATIONS
S. typhi has the capacity to affect virtually every organ system; as a result patients are vulnerable to a wide variety of complications. Intestinal perforation, occurring in 1-3% of cases is associated with the highest mortality. 1,3,4
Even when aggressively treated, mortality with perforation can be as high as 40%. In contrast there is only a 1% mortality rate with treatment in unperforated cases. Perforations can occur anywhere from the duodenum to the colon though the ileum is the most common site. Gall bladder perforation has also been reported. In children, there are frequently multiple perforations present.3 Anaesthesia in a child with typhoid intestinal perforation will be discussed in the next tutorial.
Additional complications include heart failure from myocarditis or endocarditis, liver failure from hepatitis or pancreatitis, renal failure from pyelonephritis or glomerulonephritis, respiratory failure from pneumonia, as well as disseminated intravascular coagulation, arthritis, and orchitis.1
While both genders can be infected equally, some evidence suggests that males suffer significantly more intestinal perforations than females. Typhoid fever can affect people of all ages, but the burden is heaviest among children aged 5-10 years. In endemic regions, children account for greater than 50% of intestinal perforation cases.5
In one published report of an outbreak in Uganda from 2007-2009 there were 577 cases with 249 intestinal perforations and 47 deaths resulting in an incidence rate of 8,092 cases per 100,000 people.4 While this likely does not represent the incidence rate of the population as a whole, it does emphasize that during an outbreak the burden of the disease can be tremendous.
DIAGNOSIS
The diagnosis of typhoid cannot be confirmed based on clinical presentation alone. Because the symptoms of typhoid fever are so variable, the differential diagnosis can be quite broad. Other infectious diseases such as malaria, HIV, hepatitis, GI viruses, and bacterial infections such as C.difficile or E. coli must be excluded. Suspicion of more common causes of fever such as malaria frequently delays diagnosis of typhoid. Malignancy, rheumatic processes, and inflammatory bowel diseases should also be considered.
Typhoid must be isolated from tissues in order for a definitive diagnosis to be made. Punch biopsies from rose spots on skin, or cultures from blood, urine, bone marrow, or stool are often adequate for diagnosis. Bone marrow cultures are the most sensitive at 55-90% and can grow S. typhi even in the setting of 5 days of antibiotic treatment. Unfortunately, a patient can have active typhoid and fail to grow the bacteria in culture.1
In such settings, Widal’s test has some efficacy and remains the most commonly used serologic test. It evaluates for agglutination between antibodies in the patient’s blood to the H (flagella) and O (somatic) antigens of S. typhi. The test is plagued by a high false positive rate as antibodies from other infectious disease such as dengue, malaria, and Nontyphoidal salmonella also cross-react with the S. typhi antigens. Previous immunization or exposure to the disease can also cause false-positives in acute disease. Poor commercial antigen preparation is responsible for both false positives and false negatives. Additionally, S. typhi carriers and others with low antibody production can have false-negative results in the setting of acute infection.6,7
In regions of the world where typhoid is endemic, laboratory resources are also very limited. PCR identification of S. typhi solves many of the shortcomings of cultures and Widal’s test, but these studies are expensive and require some of the most advanced technologies available. As a result of the laboratory limitations, practitioners must have a high index of suspicion for the disease and rely on the familiar constellation of symptoms previously described.
MEDICAL MANAGEMENT
Medical management is the preferred treatment for typhoid fever. Prompt diagnosis and initiation of appropriate antibiotic therapy is critical to treatment success and mortality reduction. The majority of uncomplicated cases do not require hospitalization. When possible, typhoid should be cultured and susceptibilities noted to help guide antibiotic therapy. If laboratory testing is not available, antibiotics should be selected based on regional susceptibilities. Fluroquniolones, such as ciprofloxacin and ofloxacin, are the first- line antibiotic treatment for S. typhi and S. paratyphi. Chloramphenicol, amoxicillin, and TMP-SMX (trimethoprim-sulfamethoxazole / bactrim) are all known to be efficacious against susceptible strains of typhoid. For drug resistant strains or empirical treatment, antibiotic coverage is broadened to include ceftriaxone and azithromycin. 1,5
Antibiotics can be administered PO or IV depending on the drug, and treatment courses rarely exceed 21 days. Steroid therapy with dexamethasone in conjunction with antibiotics has been shown to reduce mortality and is considered standard therapy for management of the disease. Chronic carriers of typhoid require a longer 4-6 week course of antibiotics with amoxicillin, TMP-SMX, or ciprofloxacin.1
Surgical intervention is only recommended in the case of intestinal perforation. If no perforation has been identified, surgery is not indicated as it could spread infection. In addition, it is difficult to identify which areas to resect, as the disease can be ubiquitous in the bowel.3
Surviving Sepsis
The recently revised Surviving Sepsis guidelines provide an excellent framework for managing cases of typhoid fever that have been complicated by intestinal perforation. The child or adult presenting for laparotomy due to typhoid perforation will almost always have concomitant septic shock. The definition of sepsis is a documented or suspected source of infection as well as symptoms of the systemic inflammatory response syndrome. These symptoms include altered mental status, tachypnea, fever or hypothermia, leukocytosis or leukopenia, tachycardia, (possibly bradycardia if the child is less than 1 year old) and systemic hypotension. There is also evidence of end organ dysfunction including an elevated lactate, creatinine, or a new coagulopathy.
Mortality in sepsis in minimized when the condition is promptly diagnosed and treated. The current surviving sepsis guidelines for adults recommends that within 3 hours of diagnosis cultures are drawn, empiric antibiotics are administered, a lactate level is measured, and a fluid bolus is given of 30 ml/kg. Within 6 hours the objective is to address hypotension and end organ perfusion by measuring a CVP and mixed venous saturation if available. Vasopressors can then be added to maintain a MAP 55-65mmHg with a goal of a CVP > 8mmHg or an SVO2>70%.
Hemodynamic goals are similar in pediatric patients, but there are some management differences in the Surviving Sepsis guidelines. A child in respiratory distress should initially be managed with high flow nasal cannula or CPAP. Fluid resuscitation should start with 20ml/kg of crystalloid or an albumin equivalent with a goal of maintaining capillary refill <2 seconds, normal blood pressures, baseline mental status, urine output of 1ml/kg/hr, and ultimately SVO2 of 70%. Empiric antibiotics should be administered within 1 hour of diagnosis. ACCM-PALS guidelines should be followed if the child is in shock and blood should be given to an initial goal of haemoglobin concentration of 10g/dl. Once stable, hemoglobin of 7g/dl can be allowed. [See ATOTW 278 on Paediatric Sepsis (2013)]
In both pediatric and adult patients, vasopressors should be added to maintain hemodynamic goals if shock is refractory to fluid boluses. Lung protective ventilation strategies should be employed. These typically involve low tidal volumes of 6-7ml/kg, higher respiratory rates, application of PEEP, and permissive hypercapnia. In children, if hypotension does not respond to fluid boluses and vasopressors, adrenal insufficiency should be considered and managed with steroids. 8
PREVENTION
Prevention is the preferred method of management of typhoid. Strategies include improved sanitation services, sewage disposal, water treatment services and early identification and minimization of outbreaks.
Vaccinations are available for several of the typhoid serotypes, but their effectiveness is limited. Additionally, there is no vaccine for S. paratyphi, which represents up to 25% of typhoid fever. The potential of vaccinations is limited because they are ineffective at preventing disease in the event of a large inoculum. Discretion is advised when food choices are made while traveling in a typhoid endemic area. Despite this limitation, vaccinations are still warranted as they will behave as an adjunct to treatment and limit the extent and duration of the disease.
There are two forms of the S. typhi vaccine that are currently commercially available; however, neither form is available for children below 2 years of age. The live attenuated oral vaccine, Ty21a, is administered every other day for 3 doses. It has an efficacy of 67-80% and provides some cross protection against S. paratyphi. It requires a booster vaccination every 5 years, and is not generally given until the age of 6 years. The Vi CPS vaccine is an alternative parenteral form featuring the Vi polysaccharide of the S. typhi bacterial capsule. It can be administered to children as young as 2 years old but requires a booster every 2 years and it has an efficacy of 55-72%. Typhoid vaccination is recommended for persons visiting endemic or high- risk areas, but not for those who reside in them, and not for those with an acute infection. 9
SUMMARY
Typhoid fever is an infectious disease that exclusively affects humans and is transmitted by fecal oral routes. Though it typically results in gastrointestinal symptoms, typhoid enteric fever may present as a multi-systemic disease when infection is severe. Management strategies involve prompt diagnosis or a high index of suspicion, appropriate antibiotic therapy and early initiation of sepsis management. Typhoid intestinal perforation is a serious complication if prompt surgical intervention is not available.
There will be a tutorial on anaesthesia in a child with typhoid intestinal perforation to follow.
ANSWERS TO QUESTIONS
- Salmonella typhi:
a. False: They are gram-negative, facultative anaerobic bacilli.
b. True: Salmonella typhi is also known as Enteric Fever.
c. False: It does not have an exotoxin by which it produces the disease but by being strongly antigenic produces an intense inflammatory reaction to its presence in tissues
d. True: It is transmitted by ingestion of food or water contaminated by fecal matter.
e. False: It is only found in humans. - Regarding diagnosis of typhoid fever:
a. True: Symptoms of typhoid fever are highly variable with a broad range of differential diagnoses. Typhoid must be isolated from tissues in order for a definitive diagnosis to be made.
b. True
c. True: Punch biopsies from rose spots on skin, or cultures from blood, urine, bone marrow, or stool are oftenadequate for diagnosis.
d. True
e. False: Bone marrow cultures are the most sensitive at 55-90% and can grow S. typhi even in the setting of 5days of antibiotic treatment. - Concerning treatment of typhoid fever:
a. False: Antibiotics should be selected based on regional susceptibilities if specific laboratory testing is not available.
b. True: Fluroquniolones are the first- line antibiotic treatment for S. typhi and S. paratyphi.
c. True: For drug resistant strains or empirical treatment, antibiotic coverage is broadened to include ceftriaxone and azithromycin.
d. True: Steroid therapy with dexamethasone in conjunction with antibiotics has been shown to reduce mortality and is considered standard therapy for management of the disease.
e. False: Chronic carriers of typhoid require a longer 4-6 week course of antibiotics with amoxicillin, bactrim, or ciprofloxacin
REFERENCES AND FURTHER READING
- Longo D, Fauci A, Kasper D. Harrison’s principles of internal medicine: Salmonellosis. 18th Ed. New York, N.Y.: McGraw Hill Medical, 2012.
- Musher D, Musher B. Contagious Acute Gastrointestinal Infections. N Engl J Med,2004; 351: 2417-2427.
- Ukwenya AY, Ahmed A, Garba ES. Progress in management of typhoid perforation. Ann Afr Med 2011; 10(4): 259-265.
- Neil KP, Sodha SV, Lukwago L, et al. A large outbreak of typhoid fever associated with a high rate of intestinal perforation in Kasese District, Uganda, 2008–2009. Clin Infect Dis 2012; 54(8): 1091–9.
- Wain J, Hendriksen RS, Mikoleit M, et al. Typhoid Fever. The Lancet 2014; 385(9973): 1136-1145.
- Wain J, Hosoglu S. The laboratory diagnosis of enteric fever. J Infect Dev Ctries 2008; 2(6): 421-5.
- Olopoenia L, King A. Widal agglutination test – 100 years later: still plagued by controversy. Postgrad Med J 2000; 76(892): 80-4.
- Surviving Sepsis Campaign, International Guidelines for Management of Severe Sepsis and Septic Shock: 2012, http://www.survivingsepsis.org/guidelines/Pages/default.aspx (accessed on 9/24/2016)
- Slayton R, Date K, Mintz E. Vaccination for typhoid fever in Sub-Saharan Africa. Hum Vaccin Immunother 2013; 9(4): 903-906.



