Regional Anaesthesia
KEY POINTS
- Regional anaesthesia is increasingly used in children as techniques are becoming safer through the use of ultrasonography, fluoroscopy, and tailored dosing regimens.
- Unlike in the adult population, paediatric regional anaesthesia is an integral part of analgesia rather than being used for anaesthesia.
- The safety of regional anaesthesia in children, including those under general anaesthesia, has been validated in large prospective databases.
- The landmark technique for caudal epidural in children has a high success rate but ultrasound can aid caudal epidural performance by helping identify aberrant anatomy, localising the dural sac, and reducing the rate of dural or intravascular perforation.
INTRODUCTION
The use of regional anaesthesia in children is increasing. It is becoming an integral part of intra- and postoperative analgesia. Regional techniques in children are mostly performed under general anaesthesia. Caudal epidural anaesthesia accounts for over half of regional techniques performed in children.
This tutorial will summarise the safety of regional anaesthesia in children, paediatric local anaesthetic toxicity, the use of ultrasound in paediatric regional anaesthesia, and an in-depth discussion on ultrasound-guided caudal epidural techniques and its related anatomy.
ANATOMY
The sacrum is defined as a triangle of bone formed by the fusion of 5 sacral vertebrae (Figure 1). At the apex of the triangle lies the sacral hiatus, through which caudal anaesthesia is performed.
The sacral hiatus is a midline defect in the dorsal surface of the sacral canal, formed due to failed fusion of the S5 laminae. This space is palpable by tracing a finger down the sacral crest to the natal cleft.
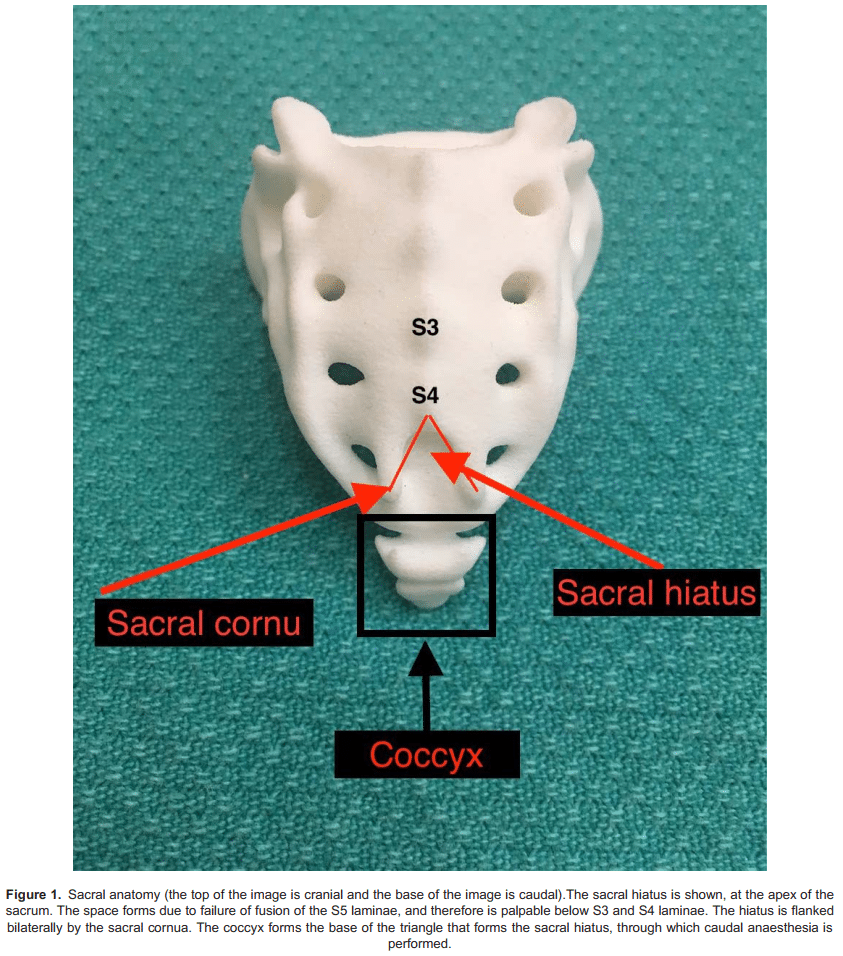
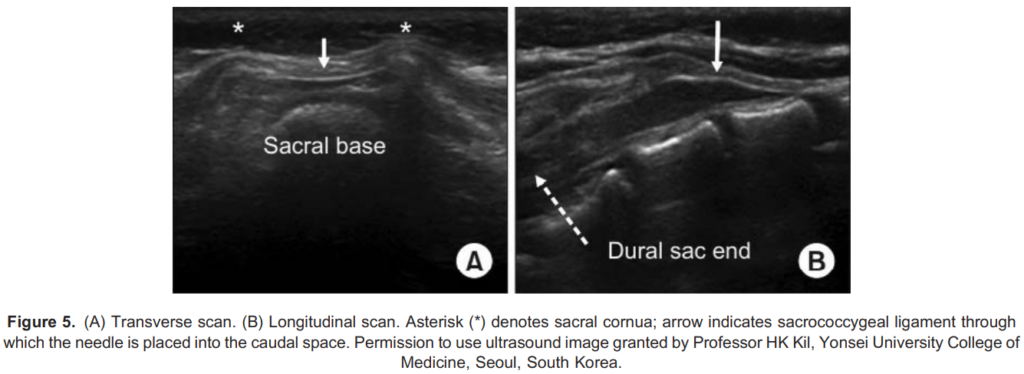
The sacral hiatus is covered posteriorly by the sacrococcygeal ligament, a firm elastic membrane which is an extension of the ligamentum flavum. The hiatus is flanked bilaterally by the sacral cornuae. These are inferior articular processes of S5, which project in a caudal direction.
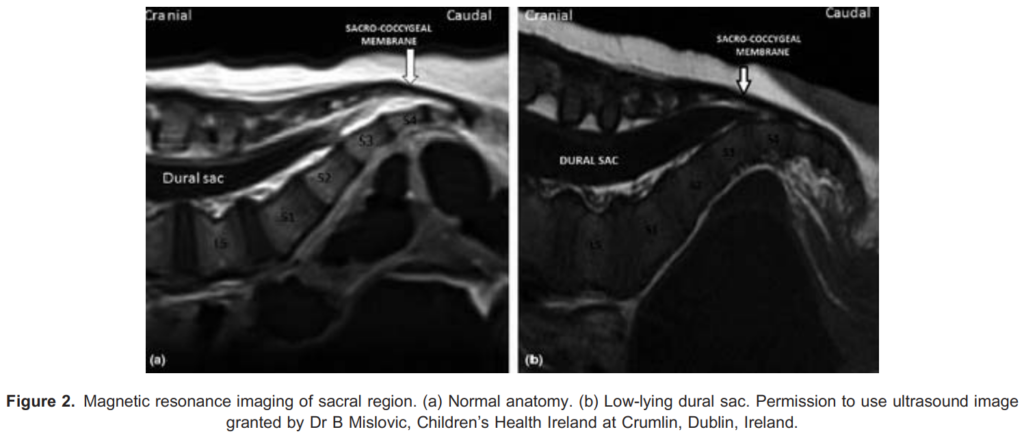
The sacral canal is triangular in shape, bordered by the dorsal aspect of the sacral vertebral bodies anteriorly and the corresponding laminae posterolaterally. It is a continuation of the lumbar spinal canal. The sacral canal contains the cauda equina, filum terminale, spinal meninges, sacral and coccygeal nerves, epidural fat, and venous plexus (anterolaterally). At birth the dura terminates at S4 and by 8 to 9 years of age terminates at the adult level of S2. The lower sacral spinal roots and the filum terminale pierce the dura at this point.
SAFETY AND COMPLICATIONS OF REGIONAL ANAESTHESIA IN CHILDREN
Regional anaesthesia is commonly performed in paediatric patients.1 Most blocks in children are performed under general anaesthesia or sedation. Prospective and retrospective safety studies support the notion that performing regional anaesthesia under general anaesthesia is safe practice.2 Large prospective databases have demonstrated the ability to perform regional anaesthesia in children with minimal risk of neurological damage.3
The Paediatric Regional Anaesthesia Network (PRAN) has prospectively collected data on over 100 000 paediatric regional blocks and has found a very low rate of complications.2 Reporting on the first 3 years of data registry, for single-injection blocks, they have reported a total adverse event rate of 3% for neuraxial, 2% for upper extremity, 1% for lower extremity, and 0.3% for other blocks (eg intercostal, paravertebral). There were no deaths or complications lasting longer than 3 months.
Local anaesthetic toxicity is more likely to occur in children than adults because of low protein binding and decreased intrinsic clearance of the drug. Infants have lower levels of ∝1-glycoprotein and an immature cytochrome P450 enzymatic system than older children.4 This results in a higher systemic amount of the free unbound drug. Local anaesthetic toxicity results from peak plasma concentration of the free unbound form, not the total dose injected. It is therefore important not to exceed the maximum dosage recommendation.
SAFETY AND COMPLICATIONS OF CAUDAL BLOCKS IN CHILDREN
The majority of blocks performed in children are caudal blocks, with just less than half represented by peripheral nerve blocks; 18 650 children who received a caudal block were included in the PRAN study.
The overall estimated incidence of complications after caudal blocks was 1.9%.5 Patients who developed complications were younger, with a median age of 11 months. The most common complications were block failure, blood aspiration, and intravascular injection. Other known complications include hypotension, arrhythmias, and respiratory depression. Of all the reported complications, no child had any long-term sequalae.
In a prospective study by Ecoffey et al3 looking at morbidity during paediatric regional anaesthesia, including over 8000 caudal blocks, there were 6 dural punctures (with no postdural puncture headache), 1 patient with self-limiting thigh tingling 3 weeks after a caudal injection, and 1 case of cardiac toxicity manifest as a transient arrhythmia, (related to wrong dose given).
Use of a caudal catheter carries a slightly higher complication rate compared to noncatheter techniques (4.9%). The most common complications are catheter malfunction, block failure, infection, and vascular puncture. In the PRAN database there were no reports of persistent neurologic problems, serious infection, or local anaesthetic systemic toxicity.6
ULTRASOUND-GUIDED CAUDAL ANAESTHESIA
Ultrasound-guided caudal anaesthesia has several advantages over landmark-based methods. Although the landmark approach has a good success rate (above 96%),7 the use of the ultrasound appears to increase the success rate of a first puncture compared to the traditional approach.8
Real-time ultrasound monitoring of local anaesthetic spread allows for visual confirmation of correct placement. This is also beneficial in the teaching process of caudal blocks. The use of ultrasound also helps in directing the appropriate catheter length to be inserted.9
An extra layer of safety may be achieved through the use of ultrasound. As the paediatric caudal area is not ossified, it can be visualized using ultrasound guidance. This is advantageous as the dural sac can be at a depth of 1cm in neonates and a relative paucity of cerebrospinal fluid (CSF) may mean dural puncture is not clinically obvious. Abnormalities of sacral formation such as sacral dysgenesis can also be elucidated using ultrasound scanning prior to block administration as not all infants with sacral abnormalities have associated cutaneous stigmata.
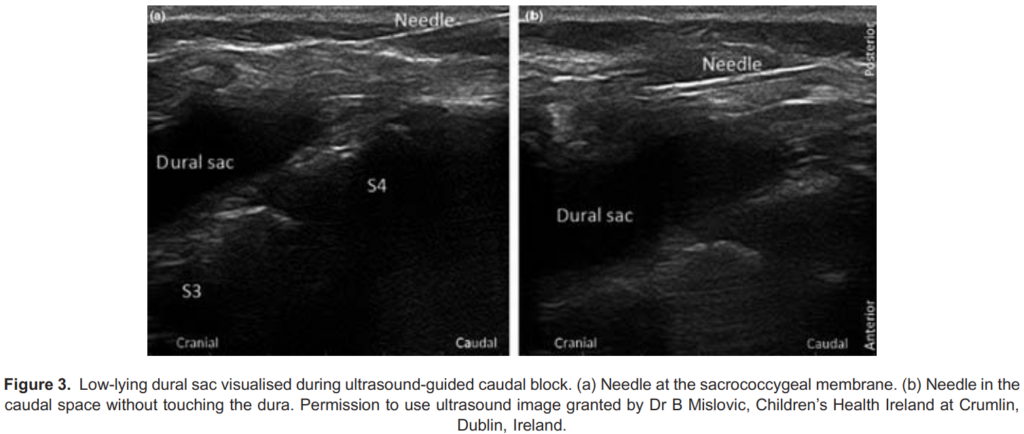
A low-lying dural sac can be visualised with the use of real-time ultrasound. This helps prevent inadvertent dural puncture and the risk of total spinal anaesthesia10 (see Figures 2 and 3). The dural sac is hyperechoic with the CSF being anechoic. Sometimes the cauda equina can be visualised as multiple hyperechoic lines within the sac.
Despite these notable advantages, so far there has not been any decrease in morbidity and mortality found when using an ultrasound technique.5
INDICATIONS
Caudal epidural anaesthesia can be used in children for most surgeries below the umbilicus. This includes surgery of the perineum, anus, and rectum; inguinal and femoral; cystoscopy and urethral surgery; and surgery on the lower limbs. With increasing volume, the block can also spread to the upper abdomen and lower thorax, but this is less reliable.
CONTRAINDICATIONS
As with other neuraxial blocks, the relative and absolute contraindications should be considered. These include coagulation disorders, infection, and refusal to consent.
Any cutaneous abnormalities overlying the caudal region should ideally be investigated prior to performing a caudal block to exclude spinal cord pathology such as a tethered cord. This should be done by magnetic resonance imaging. However ultrasound imaging can aid detection of simple-to-detect spinal cord abnormalities, such as a low-lying dural sac (see Figures 2 and 3).
LOCAL ANAESTHETIC DOSING AND ADJUNCTS
The cephalad spread of caudal anaesthesia is more predictable and extensive in children than in adults. This is mainly due to increased impedance at the lumbosacral junction that will develop during puberty (due to increased fibrosis of fat, increased lumbosacral angulation, and increasing size of the pelvis).

Current recommendations suggest the use of the Armitage regimen for volume required, whilst avoiding the maximum dosage of local anaesthetic (2.5 mg/kg for levo-bupivacaine/bupivacaine, 2 mg/kg ropivacaine).11 In very small infants, a more dilute concentration may be required in order to attain the required volume. A high volume/low concentration block appears more efficacious than a low volume/high concentration block in terms of duration and quality.12
The Armitage regimen is illustrated in Table 1.
An alternative dosing regimen has been described as 0.1 mL/segment/year of age for 1% lidocaine or 0.25% bupivacaine.13 A 3-year-old requiring analgesia to the umbilicus (T10) would therefore need 0.1mL × 13 segments (5 sacral, 5 lumbar, 3
thoracic) × 3 years = 3.9 mL.
We advise slow administration of the drug and a test dose of 0.1 mL/kg of bupivacaine 0.25% with 1:200 000 epinephrine. (This equates to 0.5 μg/kg of epinephrine, to a maximum of 15 μg.) Elevation of the heart rate by > 10 beats/min or increase in systolic blood pressure by 15 mm Hg should be taken as indicative of systemic injection. T wave changes on the electrocardiogram occur earliest following intravascular injection, followed by heart rate changes, and lastly, by blood pressure changes. Vigilance is required to capture these changes as the electrocardiogram effects may be transient.
A single caudal block will provide analgesia for 4 to 8 hours. This can be lengthened using adjuvants. These include preservative-free morphine (33-50 μg/kg), which can extend the block from 12 to 24 hours but has associated side effects, such
as pruritus, nausea, urinary retention, and respiratory depression, requiring appropriate postoperative respiratory monitoring. Clonidine is also used. This ∝2 adrenergic agonist is used in 1- to 2-μg/kg doses, although higher doses are associated with sedation, hypotension, and respiratory depression. This prolongs the block duration up to 12 hours. Furthermore, preservativefree ketamine has also been used. This can lengthen analgesia by up to 24 hours. That said, there is concern for potential neurotoxicity and as such is not currently recommended.11
The final method of augmenting block duration is by the insertion of a catheter. Due to the block’s anatomical location, a concern for infection exists but at least a 48-hour duration appears safe.14 A suggested regimen used in our institution would be levo-bupivacaine 0.125% running at 0.1 to 0.2 mL/kg/h (neonates or infants < 5 kg) or 0.1 to 0.3 mL/kg/h, with a maximum of 15 mL/h (children . 5 kg)
EQUIPMENT
- Standard monitoring: pulse oximetry, electrocardiogram, blood pressure
- Functional intravenous line in situ
- Chlorhexidine 0.5% (less neurotoxic than 2%) and sterile drape
- High-frequency linear array probe (8-14 MHz; allows for good resolution of caudal structures in children)
- 22 gauge intravenous cannula, or 22 gauge needle
- Local anaesthetic of choice, syringe (attached to a T piece if preferred; Figure 4A)
- Resuscitation equipment and medication
TECHNIQUE
- For a right-handed provider, place the patient in the left lateral position (hips and knees flexed) following monitoring and
induction of anaesthesia. Place the patient in the right lateral position if the provider is left-handed. - Prepare the field with sterilizing solution and a sterile drape.
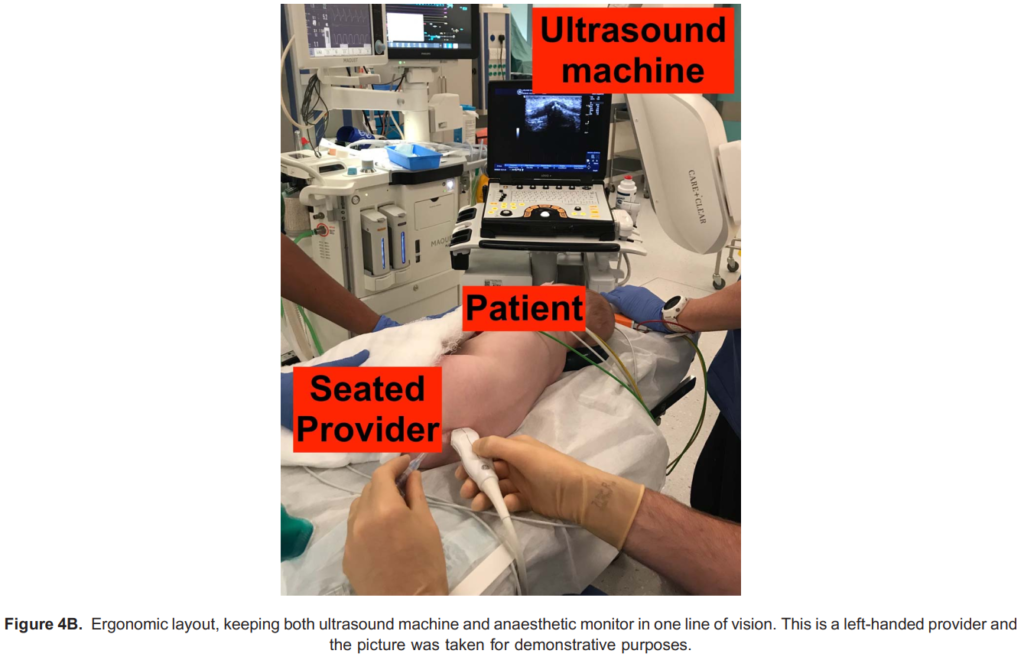
- Cover the ultrasound probe in a sterile cover. Ergonomic layout is suggested in Figure 4B.
- Initial scanning in the transverse plane allows for visualization of the midline and identification of the sacrococcygeal ligament between the 2 sacral cornua. The 2 cornua resemble the 2 eyes of a frog and thus are collectively called the frog-eye sign (Figures 5A and 6A).
- The probe is then rotated 90 degrees to acquire a longitudinal view (Figures 5B and 6B).
- The needle is advanced at a 20-degree angle with needle tip and length visualization (Figure 6C).
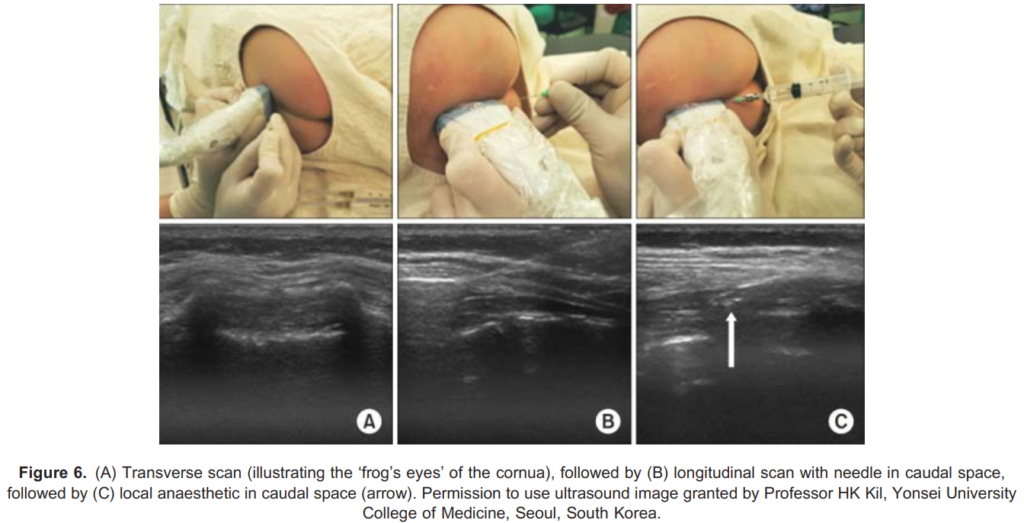
- A pop can be appreciated as the needle passes through the sacrococcygeal ligament.
- Once the needle is confirmed to be in the caudal space on the screen, carefully aspirate to confirm absence of CSF or blood.
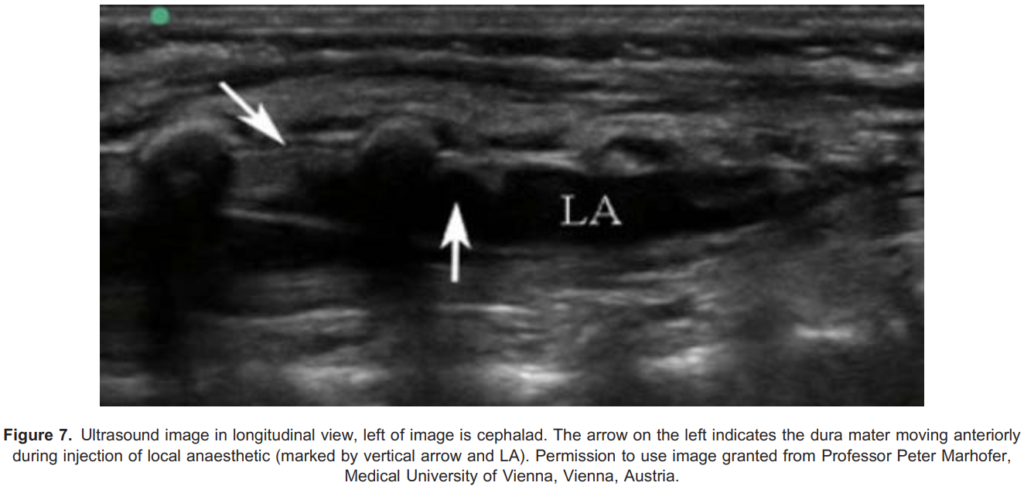
- The use of a saline bolus (0.1-0.2 mL/kg) can be performed to confirm correct positioning. If this is the case, real-time
visualization of cranial spread of the saline with anterior displacement of the posterior dura is seen (Figure 7). - Lack of dural displacement can be associated with intrathecal or intravascular injection requiring repositioning of the needle.
- A slow injection of the local anaesthetic can then be performed, often with a test dose of 0.1 mL/kg 0.25% bupivacaine with 1:200 000 adrenaline, observing for tachycardia and electrocardiogram changes. If the test dose did not elicit symptoms, inject the remaining bupivacaine dose as per the Armitage regimen.
TROUBLESHOOTING

TIPS AND TRICKS
- Injection of saline test dose spares local anaesthetic volume and ensures needle is in appropriate position.
- If the patient is very small, consider 0.125% bupivacaine to increase volume.
- Positioning is key. Have the ultrasound machine in direct line of vision, sit down, resting your arms on the bed and ensure the patient is well flexed, using an assistant if possible.
SUMMARY
Regional anaesthesia use in children is increasing and has shown to be safe by large prospective databases, even when performed under general anaesthesia. Over half of all blocks performed in children are caudal epidurals. Caudal epidurals have a large array of indications, essentially any surgery below the umbilicus, and the block can be extended to include upper abdominal and lower thoracic incisions. The landmark technique is very successful in children because of easily palpable landmarks. However, ultrasound can aid aberrant anatomy identification, localisation of the dural sac, and reduction in dural perforation.
ACKNOWLEDGEMENTS
The authors would like to thank Dr B Mislovic, Professor Peter Marhofer, and Professor HK Kil for the creation of the ultrasound images and permission for their use in this article. The 3-dimensional printed sacrum was supplied by Dr Elaine Ng, at SickKids.
REFERENCES
- Shah RD, Suresh S. Applications of regional anaesthesia in paediatrics. Br J Anaesth. 2013;111(suppl 1):i114-124.
- Polaner DM, Taenzer AH, Walker BJ, et al. Pediatric Regional Anesthesia Network (PRAN). Anesth Analg.2012;115(6):1353-1364.
- Ecoffey C, Lacroix F, Giaufre´ E, Orliaguet G, Courre`ges P, Association des Anesthe´sistes Re´animateurs Pe´diatriques d’Expression Franc¸ aise (ADARPEF). Epidemiology and morbidity of regional anesthesia in children: a follow-up one-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists (ADARPEF). Paediatr Anaesth. 2010;20(12):1061-1069.
- Mazoit J-X, Dalens BJ. Pharmacokinetics of local anaesthetics in infants and children. Clin Pharmacokinet. 2004;43(1):17-32.
- Suresh S, Long J, Birmingham PK, De Oliveira GS. Are caudal blocks for pain control safe in children? An analysis of 18,650 caudal blocks from the Pediatric Regional Anesthesia Network (PRAN) database. Anesth Analg. 2015;120(1):151-156.
- Walker BJ, Long JB, De Oliveira GS, et al. Peripheral nerve catheters in children: an analysis of safety and practice patterns from the Pediatric Regional Anesthesia Network (PRAN). Br J Anaesth. 2015;115(3):457-462.
- Kao S-C, Lin C-S. Caudal epidural block: an updated review of anatomy and techniques. BioMed Res Int. 2017;2017:9217145.
- Ahiskalioglu A, Yayik AM, Ahiskalioglu EO, et al. Ultrasound-guided versus conventional injection for caudal block in children: a prospective randomized clinical study. J Clin Anesth. 2018;44:91-96.
- Ponde VC, Bedekar VV, Desai AP, Puranik KA. Does ultrasound guidance add accuracy to continuous caudal-epidural catheter placements in neonates and infants? Paediatr Anaesth. 2017;27(10):1010-1014.
- Mislovic B. Successful use of ultrasound-guided caudal catheter in a child with a very low termination of dural sac and Opitz–GBBB syndrome: a case report. Pediatr Anesth. 2015;25(10):1060-1062.
- Suresh S, Ecoffey C, Bosenberg A, et al. The European Society of Regional Anaesthesia and Pain Therapy/American Society of Regional Anesthesia and Pain Medicine recommendations on local anesthetics and adjuvants dosage in pediatric regional anesthesia. Reg Anesth Pain Med. 2018;43(2):211-216.
- Hong J-Y, Han SW, Kim WO, Cho JS, Kil HK. A comparison of high volume/low concentration and low volume/high concentration ropivacaine in caudal analgesia for pediatric orchiopexy. Anesth Analg. 2009;109(4):1073-1078.
- Schulte-Steinberg O, Rahlfs VW. Spread of extradural analgesia following caudal injection in children. A statistical study. Br J Anaesth. 1977;49(10):1027-1034.
- Kost-Byerly S, Tobin JR, Greenberg RS, Billett C, Zahurak M, Yaster M. Bacterial colonization and infection rate of continuous epidural catheters in children. Anesth Analg. 1998;86(4):712-716.



