General Topics
KEY QUESTIONS
- What is a vapour?
- Define saturated vapour pressure (SVP)
- Define latent heat of vapourisation
- Does the Oxford Miniature Vapouriser have a temperature compensation device?
- What is different about the desflurane (Tec 6) vapouriser compared with standard plenum vapourisers?
- Why are these differences necessary?
- Define critical temperature
VAPOURS
For more than 150 years, the vapours of a variety of liquids have been the mainstay of general anaesthesia. In the mid to late 19th century chloroform and ether were used, with patients often breathing uncontrolled concentrations of the vapour, while today specially designed devices (vapourisers) allow us to deliver a very accurate concentration of all anaesthetics e.g. halothane, isoflurane, sevoflurane. To understand how these devices work, it is first necessary to understand what a vapour is and the properties of vapours.
If a glass of water (or any liquid) is left exposed to the atmosphere, the water will eventually disappear – we say that it has evaporated. The water has changed its physical state from a liquid to a gas. This happens because the molecules (or atoms) in the liquid all have varying amounts of kinetic energy (the energy of motion), and some have sufficient energy to break free of the liquid, enter the atmosphere and become a gas. If the temperature of this gas is below its critical temperature, it is known as a vapour (see appendix 1 for more details on critical temperature).
As molecules of a liquid enter the gas phase, the average energy of the remaining molecules falls and the liquid cools. The cool liquid then absorbs heat from the environment, the molecules speed up and more break free entering the vapour phase, a process clearly seen when ethyl chloride is sprayed onto the skin.. This process continues until all the liquid has turned to vapour. The heat energy required for this process is termed the latent heat of vapourisation (see below).
The higher the temperature of the liquid, the quicker it evaporates because the higher energy of the molecules means more can break free from the liquid. So on a hot day, the glass of water evaporates more quickly than on a cold day. Also, for a given ambient temperature, the closer a liquid is to its boiling point, the quicker it will turn to a vapour. This is because the kinetic energy of the molecules increases as they approach boiling point. Therefore at room temperature (20°C), a jar of ether (boiling point 35°C) will turn to vapour quicker than water (boiling point 100°C).
Now, what happens if we put the water in a jar with a sealed lid? The process of evaporation still occurs but molecules not only leave the liquid but also re-enter the liquid as they cannot escape from the jar. Eventually a state of equilibrium is reached where the number of molecules leaving the surface of the liquid is the same as the number re-entering the liquid (fig 1). Because the molecules in gaseous form have kinetic energy, they bounce off the walls of the container and exert a pressure that can be measured. This pressure is called the saturated vapour pressure (SVP) and like all pressures is measured in kiloPascals (kPa)
Saturated vapour pressure is defined as “The pressure exerted by the vapour of a substance’s pure liquid (or solid) phase when, at a given temperature, it is in equilibrium with a plane surface of that substance.”
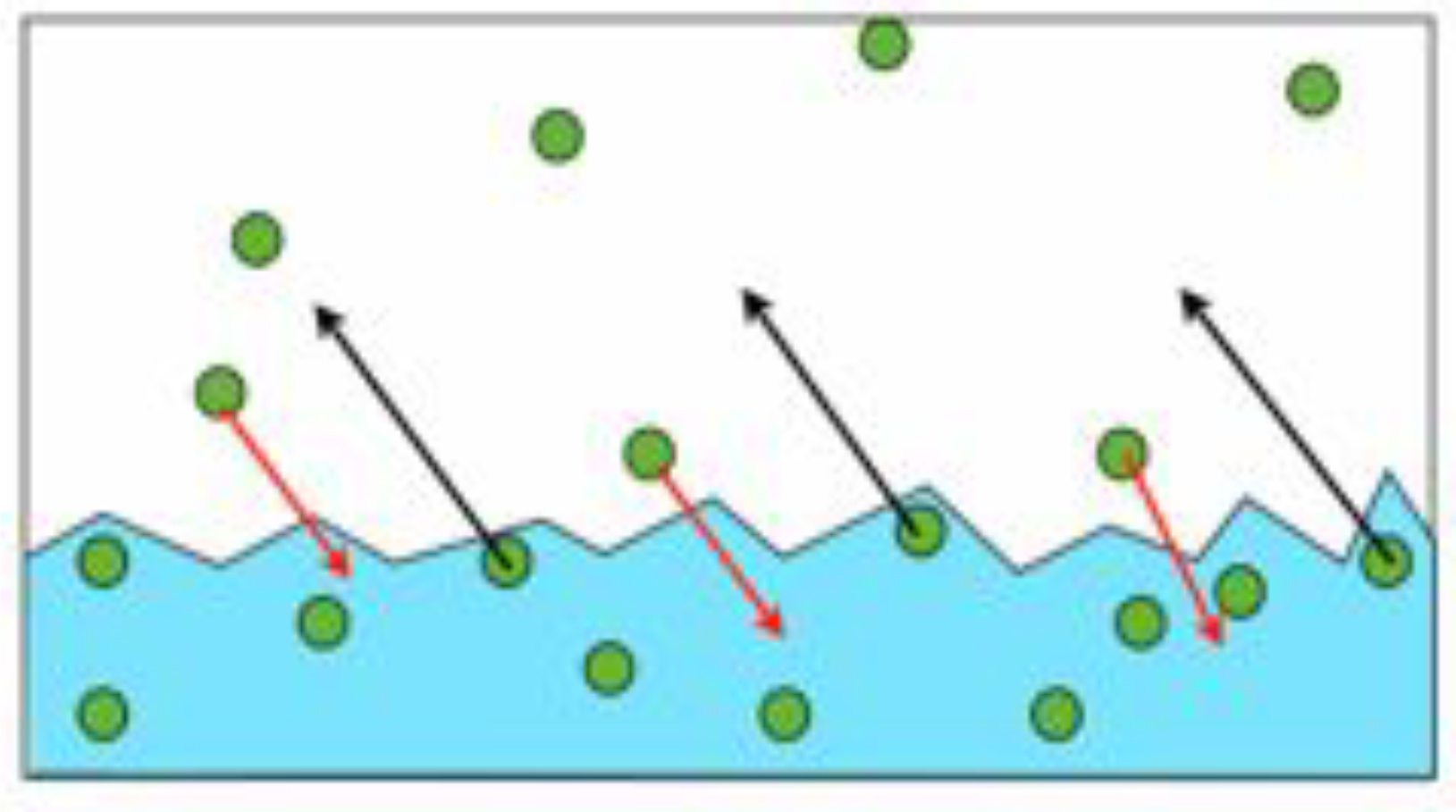
Figure 1. Molecules leaving and re-entering the surface of a liquid.
The SVP of a liquid is independent of the ambient pressure, but increases with increasing temperature. If we continue to heat a liquid, it eventually starts to boil and the SVP equals the ambient pressure (usually 101kPa). Therefore the boiling point of any liquid can be defined as the temperature at which its SVP equals atmospheric (or ambient) pressure.

Table 1 – The saturated vapour pressures of some common volatile anaesthetics at 20°C
So why is the SVP important to anaesthetists? The answer is that it allows us to generate known concentrations of vapour. Dalton’s law of partial pressures states that, in a mixture of gases each gas will exert a pressure, the same as it would exert if it occupied the same volume by itself. Alternatively, the partial pressure of a gas in a mixture is proportional to the volume it occupies in the mixture.
The implication of this is that if we generate a saturated vapour of a liquid in air and deliver the mixture at atmospheric pressure, we can derive the volume % occupied by the vapour in the gas mixture – i.e. its concentration.
For example if we consider our jar of water, the SVP of water at 20°C is 6.2kPa. If atmospheric pressure is 100kPa, the volume % (or concentration) of the vapour is calculated thus:
Concentration of water vapour = 6.2/100 = 6.2%
This is the same for any liquid. From table 1 we can see that it would be possible to create halothane vapour at a concentration of 32%, far greater than that needed for anaesthesia and potentially dangerous! To overcome this, modern vapourisers produce a saturated vapour, and then mix this with a known volume of a carrier gas (usually oxygen and either air or nitrous oxide), to produce a known, lower concentration of vapour using the principle above. Clearly this was not the case in very early vapourisers where there was little if any control over concentration.
VAPOURISERS
We will now discuss the operation of vapourisers – starting with the older devices, and moving on to the more sophisticated modern devices.
On October 16, 1846 at the Massachusetts General Hospital in America, William Morton gave the first public demonstration of anaesthesia using diethyl ether. He used a device of his own making consisting of a glass bulb with two orifices and containing sponges soaked in ether (fig 2.). One of the orifices was a mouth-piece through which the patient breathed in ether vapour, while at the same time drawing in air through the other. As the air passed over the sponges, more ether vapourises maintaining a steady supply of anaesthetic to the patient. Fortunately the low boiling point of ether means that vapour is produced quickly and at a sufficient concentration to maintain anaesthesia.

Figure 2. Morton’s ether vapouriser (Reproduced with kind permission from Dr Rod Westhorpe, Australian Society of Anaesthetists).
Within a short period of time, John Snow in England introduced a similar inhaler, incorporating a vapourisation chamber into which liquid ether (and later chloroform) could be poured, connected to a facemask by a length of brass tubing.
Morton’s and Snow’s inhalers are called drawover vapourisers, with the gas (air) being pulled through the device by the patient’s inspiratory effort (see below for more modern types of drawover vapourisers). However, in both it was relatively difficult to control the concentration of anaesthetic vapour delivered to the patient as this depended on the SVP of the liquid used (chloroform is much lower than ether), the temperature of the liquid and the patient’s ventilation. This problem was partially resolved at the turn of the century with the development of the Schimmelbusch mask (fig 3).
Designed in 1890 by Carl Schimmelbusch, this hinge-framed mask holds a piece of lint or cloth. Ether or chloroform is dropped onto the material and the mask held onto the patient’s face. The anaesthetic evaporates and is drawn through the material and inhaled by the patient. The depth of anaesthesia could easily be altered by changing the rate at which the liquid anaesthetic is dropped onto the mask, or lifting the mask from the patient’s face.

Figure 3. The Schimmelbusch Mask
Use of the inhalers described above results in the patients breathing a mixture of air and vapour. In 1867, FE Junker developed a chloroform inhaler which, in many ways can be thought of as the direct predecessor of the modern “plenum” vapouriser. The Junker’s inhaler incorporated 2 rubber bags in series leading to a bottle which contained chloroform. The first bag had an attachment for oxygen to be administered as the carrying gas and acted as a reservoir of oxygen. Oxygen passed from this reservoir to a second bag which had a net around it. By manually squeezing this second bag, the anaesthetist was able to administer a positive inspiratory pressure to the patient. Gas from this second bag passed to a bottle, where it bubbled through liquid chloroform. The resultant mixture of oxygen and chloroform vapour passed to the patient via a facemask. The Junkers Inhaler required a positive pressure to drive the carrier gas (oxygen) through the vapouriser – a principle that can be seen in the “plenum” vapourisers used in modern anaesthetic practice.
The next major step forward in vapouriser design was the Boyles bottle – named after its inventor, Henry Edmund Gaskin Boyle, who worked as a Consultant Anaesthetist at St Bartholomew’s Hospital London in the early 1900s (fig 4). He also developed the first anaesthetic machine that bears his name.
The Boyles bottle is another early example of a plenum type vapouriser. Fresh gas enters the vapouriser, and passes either straight through a bypass channel, where it does not comes in to contact with anaesthetic vapour, or it passes into the vapourisation chamber which is filled with anaesthetic liquid. A plunger and cowl arrangement are used to increase the contact of the carrier gas with the surface of the anaesthetic, or even force the gas to bubble through the volatile. This increases the saturation of the carrier gas in the vapourisation chamber, and hence the vapour can be delivered at a pressure close to its SVP.

Figure 4. Boyle’s bottle
The saturated carrier gas leaves the vapourisation chamber and mixes with the gas which has bypassed the chamber. A valve mechanism and control lever controls the proportion of fresh gas which bypasses the vapourisation chamber – the splitting ratio. If the vapourisation chamber fully produces a fully saturated gas, the splitting ratio determines the concentration of vapour delivered to the patient (see below).
Problems with early vapourisers
The Schimmelbusch mask, the Junkers inhaler, and the Boyle’s bottle were widely used delivery devices. However a number of factors caused variation in the effectiveness of vapourisation, and thus the saturated vapour pressure and concentration of drug delivered to the patient.
The most important factor relates to cooling of the systems during use. Energy in the form of heat is required to change a substance from the liquid to the vapour phase. The heat energy required to convert one kilogram of a substance from liquid into gas form at a given temperature is called the latent heat of vapourisation. As a volatile anaesthetic drug evaporates the molecules with the highest kinetic energy leave the liquid state and take energy with them. This lowers the total energy held by the remaining liquid and thus reduces its temperature. As the SVP is dependent on temperature, a drop in the temperature of the system leads to a drop in the SVP of the volatile.
Vapourisation leads to cooling of the delivery system which in turn decreases the SVP of the volatile and thus the amount of volatile delivered to the patient.
Problems encountered due to loss of heat during vapourisation (see below) within systems were well known to anaesthetists using the early vapourisers. With the Schimmelbusch mask, ice deposits frequently formed on the material from exhaled water vapour, thus reducing the surface area available for further vapourisation. Both the Junker’s inhaler and the Boyle’s bottle were glass – and thus had a low heat capacity and a tendency to cool rapidly during use. Many anaesthetists began to put the Boyle’s bottle in a warm water bath, which increased the heat capacity of the system and reduced the cooling of the liquid anaesthetic drug. The system remained far from perfect however, and consequently a number of temperature compensation features were developed and introduced in to modern plenum vapourisers – known as Temperature Compensated or “Tec” vapourisers.
Modern plenum vapourisers
A plenum vapouriser (fig. 5) has a high resistance to gas flow through it. Consequently it has to be used with a source of compressed gas. The patient’s respiratory effort would be inadequate to draw gas through the vapouriser. Modern plenum vapourisers are accurate and have many compensatory and safety features.
On entering the vapouriser the fresh gas flow (FGF) is split into two separate flows, a low flow that enters the vapourisation chamber and a high flow that enters a bypass channel. A series of wicks and/or baffles increase the surface area of the liquid anaesthetic drug, increasing the rate of vapourisation, to ensure that the gas leaving the vapourisation chamber is fully saturated with vapour.
In sevoflurane vapourisers this results in the production of a vapour concentration of 21%, far higher than needed for anaesthesia. This is therefore diluted by being mixed with the gas from the bypass channel; for example if 10% of the gas flow enters the vapourizing chamber, when mixed with the bypass gas (90%), the resulting concentration will be 2.1% (Why? Because the original volume of vapour will now be in ten-times the original volume – Dalton’s law). This separation of the gas flow through the vapourisers is called the splitting ratio and is controlled by a dial, usually calibrated to indicate the final concentration of anaesthetic vapour.
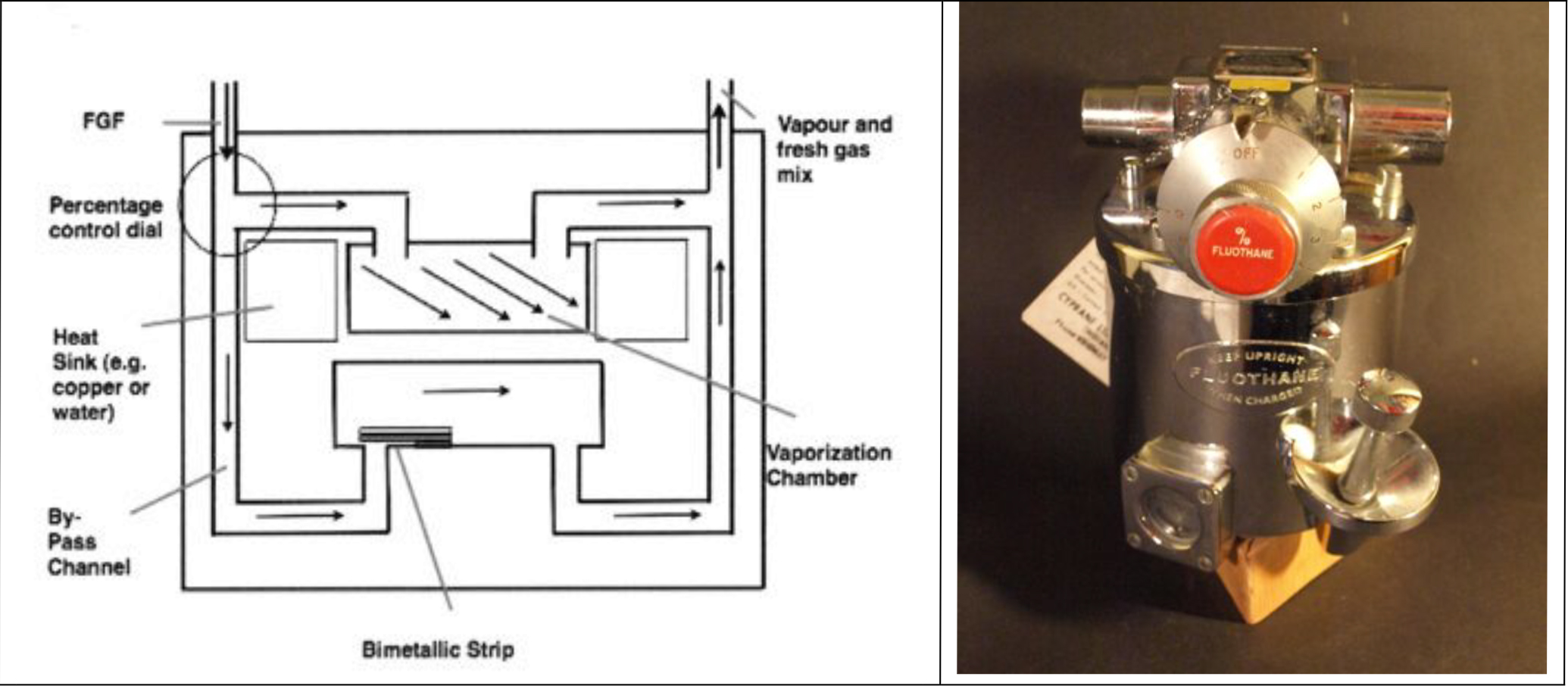
Figure 5. Diagram of plenum vapouriser showing bypass channel & bimetallic strip and Fluotec Mk2
As the drug evaporates the temperature within the chamber falls as previously discussed. This would cause the vapour pressure of the volatile drug in the gas leaving the chamber to fall – unless compensated for. Modern plenum vapourisers compensate in a number of ways. Firstly, they have a high heat capacity, and hence the temperature change caused by loss of latent heat is minimized. Many have heat sinks of water or copper which further increase the heat capacity, so that the temperature within the chamber stays as constant as possible.
Secondly, Tec vapourisers have a further temperature compensating device – the bimetallic strip, which is incorporated into the bypass channel. As the vapouriser cools, the metals in the strip shrink at differing rates, and the strip bends. This acts as a valve and alters the splitting ratio – increasing the amount of gas entering the vapourisation chamber as the temperature drops. This adds “extra” vapour to the gas flow leaving the vapouriser. The bimetallic strip is illustrated in fig 5.
Modern plenum vapourisers (fig. 6) have a number of safety features. The vapourisers are designed to be used with only one anaesthetic drug. The vapourisation chamber is filled by an agent specific “key filling system”, which prevents the wrong drug being used. The vapourisers are mounted on the back-bar of the anaesthetic machine, and the mounting system incorporates a lock which ensures only one vapouriser can be used at any one time. Other safety devices include; an anti-tip mechanism which will not allow volatile liquid in to the bypass channel even if the vapouriser is tipped upside down and a long inlet into the vapourisation chamber which prevents the bypass channel becoming contaminated by back flow from the vapourisation chamber during positive pressure ventilation.
The temperature compensation and safety features combine to make the Tec series of vapourisers reliable and safe. They are consequently the most commonly used vapourisers in anaesthetic practice.

Figure 6. Desflurane (left) and sevoflurane (right) vapourisers.
Drawover vapourisers
Drawover vapourisers are similar in design to plenum vapourisers, but have low resistance to flow. This allows the patient’s own respiratory effort to “draw” the fresh gas over the volatile drug, and obviates the need for a pressurised source of gas. This makes these vapourisers ideally suited to use in places where compressed gas supplies are unavailable. Popular draw-over vapourisers include the EMO and OMV vapourisers.
EMO Vapouriser
The EMO was created in 1952 by Epstein & MacIntosh of Oxford (fig. 7). It is heavier than other draw-over vapourisers and incorporates a bellows temperature compensation device and a water reservoir heat sink. It is used only with ether and will tend to give lower concentrations of vapour if used with a positive pressure ventilation, achieved by using the hand operated bellows seen in fig 7. This is a result of a decreased transit time through the vapourisation chamber, leading to less than full saturation of the carrier gas.

Figure 7. EMO and bellows
Oxford Miniature Vapouriser
The Oxford Miniature Vapouriser (OMV – fig. 8) is much more compact than the EMO. Consequently it is often used in remote locations and in the field – with equipment such as the tri-service apparatus. The vapour chamber comes in 2 sizes, 50ml and 100ml. The small size of the vapour chamber requires frequent top-ups.
The vapouriser is versatile and can be used reasonably accurately with many different volatile anaesthetics; the only requirement being a change of the dial scale. Two or more OMV vapourisers can also be used in series so more than one anaesthetic drug can be used simultaneously (usually halothane and trilene).
The OMV does not have a mechanical temperature compensation device but does have a heat sink containing antifreeze to allow its use in winter field conditions. Constant topping up of the vapour chamber with fresh anaesthetic at room temperature also helps to compensate for the drop in temperature due to vapourisation. If used with halothane the OMV’s dial may become stiff due to build up of thymol. This is easily taken care of by filling the vapour chamber with ether and working the lever back and forth to dissolve the thymol.

Figure 8. Oxford Miniature Vapouriser
TEC 6 Desflurane vapouriser
The final model of vapouriser discussed is the Tec 6 vapouriser, which is used solely for desflurane (fig. 6), and has a mechanism of action completely different to other vapourisers.
The boiling point of desflurane is 23.5°C. This means that it is almost boiling at a room temperature of 20 degrees. As a result, small changes in ambient temperature can cause large swings in SVP. This makes a “normal” plenum type vapouriser inaccurate and potentially very dangerous when used with desflurane.
The TEC 6 vapouriser has been developed specifically for desflurane, in order to overcome the difficulties associated with its low boiling point. The Tec 6 actively heats and pressurises the desflurane in the vapourisation chamber to 39 degrees and 2 atmospheres. Pure desflurane vapour is then injected directly into the fresh gas flow. This process is electronically controlled and operated by a calibrated dial. A transducer adjusts the amount of vapour injected in to the gas stream depending upon the fresh gas flow. Due to the heating, pressurisation, alarm systems and electronics it requires a source of electricity. It is agent specific and a warm-up period is necessary prior to use. It is very accurate at low flows and can be filled during use.
SUMMARY
The use of volatile anaesthetic drugs requires specialised equipment to deliver a known steady partial pressure of drug to the patient. A number of physical factors potentially introduce inaccuracy in to this process, and vapourisers have been developed with mechanisms to compensate for this inaccuracy.
ANSWERS TO KEY QUESTIONS
- A substance is said to be in its vapour phase vapour when it exists as a gas, below its critical temperature.
- Saturated vapour pressure is the pressure exerted by the vapour of a substance’s pure liquid or solid phase when, at a given temperature, it is in equilibrium with a plane surface of that substance.
- The Latent Heat of Vapourisation is the heat energy required to convert a substance from a liquid into a gas form without changing its temperature.
- No, it does have a heat sink but no temperature compensation c.f. EMO.
- The TEC 6 vapouriser for Desflurane heats and pressurises the volatile to 39 degrees and 2 ATM respectively. The correct amount of pure volatile is then electronically injected into the carrier gas, depending upon the fresh gas flow.
- This is because the boiling point of Desflurane is 23.5 degrees centigrade and small fluctuations around room temperature would lead to large changes of SVP.
- Critical Temperature is the temperature above which the gas of a particular substance cannot be liquefied no matter how much pressure is applied to it.
APPENDIX 1: Critical temperature
The terms gas and vapour are often used interchangeably, but they are quite distinct. If a vapour is compressed by increasing the ambient pressure, it will return to the liquid state. However, a temperature exists, above which it will not become a liquid however much it is compressed. This temperature is called its critical temperature and above this the substance exists as a gas. Critical temperature is therefore defined as “the temperature above which a substance cannot be liquefied irrespective of the pressure applied,” so strictly speaking a gas is a substance above its critical temperature. There are two good examples of the difference in anaesthesia; cylinders of nitrous oxide and oxygen. A cylinder of nitrous oxide, until almost empty, behaves just like our jar of water with the lid on.
The cylinder contains liquid nitrous oxide and above it is nitrous oxide vapour at its SVP at the ambient temperature. The critical temperature for nitrous oxide is 36.5°C, and while the cylinder contents are kept below this the liquid and vapour remain. In an oxygen cylinder, things are very different. The critical temperature for oxygen is -119°C, so at normal ambient temperature the cylinder contents are always a gas and never liquefy irrespective of the pressure. It is important to remember that although in most environments nitrous oxide exists as described, in tropical climates where the temperature may exceed 36.5°C the contents of the cylinder would be a gas.
Not surprisingly, each liquid at its critical temperature has a specific SVP, and this is termed its critical pressure. For nitrous oxide at 36.5°C, the critical pressure is 7300kPa; much higher than the pressure normally see in cylinders at around 20°C. Not surprisingly, the critical pressure for oxygen (at -119°C) is only 5000kPa.



