Patient Safety
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding implementation of the WHO checklist:
a. Anaesthetists are responsible for implementing the checklist in theatre
b. Implementation of the checklist is an ideal student project
c. It is helpful to collect data, but it is important to only feed this back to the theatre team at the end of the implementation process
d. Where an item cannot be completed in your facility due to lack of resources, remove that item from the checklist
e. Staff are more likely to use the checklist if they understand how it improves patient safety - When conducting the WHO Checklist:
a. Good communication is important – use clear questions and direct them to an individual using their name
b. The WHO checklist is only for elective cases, not emergencies
c. It is important to maintain status and hierarchy during the checklist so that everyone knows who is in charge
d. Antibiotics are usually given after skin incision
e. Retained swabs, needles and instruments are an important recurring adverse event worldwide. Use of standardised packs and lists of instruments helps the counting process - When briefing and debriefing:
a. These steps are added before and after each surgical case
b. The briefing provides an opportunity for everyone to introduce himself or herself and identify their role
c. Surgeons are usually very busy so they do not need to attend the pre-list briefing
d. The briefing takes too long for it to be used routinely
e. There would be no reason to debrief a day that had run smoothly as there is nothing to learn
Key Points
- Adverse events in surgery are an important problem globally. Many are preventable.
- The WHO Surgical Safety Checklist has been shown to reduce surgical complications and improve communication and teamwork in the operating theatre.
- Key components to successful implementation of the checklist include senior administrative support, surgical buy-in, ensuring underlying processes of care are in place and the use of local champions.
- Modification to suit local practice, training, stepwise implementation and real time feedback on performance improves uptake.
INTRODUCTION
In 2008 the World Health Organization (WHO) introduced a surgical safety checklist applicable to all surgical teams to be used for every patient undergoing a surgical procedure. This tool has been implemented around the world, and encourages dialogue within multidisciplinary teams and the use of routine safety checks to minimize harm to our patients.
Example case:
An 18yr old girl, Ms X came to theatre for an urgent appendicectomy. When the operating staff called to the ward for Ms X, her nurse was busy with another patient. Another nurse helpfully gathered the notes and brought Ms X to the operating area. An anaesthetist, Dr A, had assessed Ms X on the previous shift and had given a brief handover to the current anaesthetist, Dr B. Dr B was approaching the end of a busy 12-hour shift, with emergency cases on the priority list. Having anaesthetised Ms X, Dr B was about to give antibiotics and noticed that the allergy box on the anaesthetic chart was left blank. She went to check the drug chart and saw Ms X had a severe penicillin allergy. The nurse was unaware of this allergy, Ms X did not mention it before induction and Dr A had forgotten to hand it over to Dr B. This was a near miss and could have been avoided if the allergies had been checked before induction of anaesthesia during the ‘sign in’ part of the surgical safety checklist.
Errors in surgery – the size of the problem
The WHO have estimated that 234M operations are performed annually around the globe1. A systematic review including over 74 000 patient records found a median incidence of in-hospital adverse events of 9.2% with approximately half of those events being operation or drug-related, and 43% deemed preventable2. In England and Wales, the National Reporting and Learning System (NRLS) reported 10 526 patients died or came to severe harm secondary to incidents in 2013-2014. Over 3000 of these incidents were related to treatment or procedure, or implementation of care and ongoing monitoring/review3. These figures, when extrapolated to the global number of surgeries conducted, are alarming and provide clear motivation to make surgery safer.
History of the WHO Surgical Safety Checklist
In 2002 the World Health Assembly urged countries to improve the safety of health care and monitoring systems. They requested that the WHO set global standards of care and provided support for countries to improve patient safety. As a result, WHO Patient Safety was formed, and focussed its energy on campaigns named Global Patient Safety Challenges. Following their first challenge, ‘Clean Care is Safer Care’, WHO launched ‘Safe Surgery Saves Lives’ and led by Professor Atul Gawande, published WHO Guidelines for Safe Surgery4. This set out 10 essential objectives for safe surgery from which the Surgical Safety Checklist was derived. (Figure 1)
The aim of this ‘WHO checklist’ was to give teams a simple, efficient set of priority checks to improve effective teamwork and communication and encourage active consideration of patient safety for every operation performed. WHO also wanted to ensure consistency in patient safety in surgery and introduce (or maintain) a culture that values patient safety5.
In a pilot study of the WHO checklist implementation, Professor Gawande’s team prospectively observed over 3000 patients prior to the introduction of the checklist and nearly 4000 patients after checklist implementation, and measured the rate of surgical complication or mortality up to 30 days after surgery or until discharge6. The study included four hospitals in low- and middle-income countries and four hospitals in high-income countries and found the overall rate of death prior to introduction of the checklist was 1.5% and after checklist implementation fell to 0.8%. Inpatient complications were also reduced, from 11% pre checklist to 7% after the checklist was introduced. As a measure of adherence to the checklist, they identified 6 safety indicators, such as pre-incision antibiotics, swab counts and routine anaesthetic checks, and saw an increase in performance of these from 34.2% pre checklist to 56.7% post checklist. It is interesting that even with only 56% completing these 6 indicators, significant reductions in complications and death rates were seen. The checklist implementation team used team introductions, briefings and debriefings as part of the safety routine, which has also been formalised as part of the checklist strategy in the UK (see below).
By September 2014, the WHO team had identified 4132 institutions who had expressed an interest in using the checklist and 1790 institutions who were actively using the checklist in at least one operating theatre7. Seven years after introduction of the checklist, numerous studies have shown the benefit of the checklist, but observers, audits and trials have also reported common barriers to successful use of this patient safety tool. Key to successful implementation across all cultures, economies and specialties seems to be engagement of the whole team, through understanding the relevance and power of this tool in their setting.
Constituent parts of the checklist
There are three phases to the checklist:
- Sign in – before induction of anaesthesia, ideally with surgeon present, but not essential, verbally verify, review with the patient when possible:
- Patient identity
- Procedure and site
- Consent for surgery
- Operative site is marked if appropriate (involving left or right distinction)
- Pulse oximeter is on the patient and functioning
Review between anaesthetist and checklist coordinator: - Patient’s risk of blood loss. If >500ml in adults or >7ml/kg in children, it is recommended to have at least 2 large bore intravenous lines or a central line before surgical incision and fluids or blood available
- Airway difficulty or aspiration risk. Where a potentially high-risk airway is identified, at a minimum the approach to anaesthesia should be adjusted accordingly, emergency equipment must be accessible and a capable assistant should be physically present during induction. Symptomatic active reflux or a full stomach should also be handled with a modified plan
- Known allergies – all members of team need to be aware
- Anaesthesia safety checks complete (equipment, medications, emergency medications, patient’s anaesthetic risk)
- Time out – after induction and before surgical incision, entire team
- Each team member introduces him/herself by name and role
- Pause to confirm correct operation for correct patient on correct site. Anaesthetist, nurse and surgeon should all individually confirm agreement, plus the patient if awake
- Review anticipated critical events
- Surgical critical/unexpected steps, operative duration, anticipated blood loss
- Anaesthetic patient specific concerns, for example, intention to use blood products, co-morbidities
- Nurses confirm sterility of instruments and discuss equipment issues/concerns
- Confirm prophylactic antibiotics where required, was given within the 60 minutes prior to skin incision. If not given and required, administer prior to incision. If >60 minutes, consider re-dosing the patient
- Essential imaging displayed as appropriate
- Sign out – during or immediately after wound closure, before moving the patient out of the operating room, whilst
surgeon still present- Confirm operation performed and recorded
- Check instrument, sponge/swab and needle counts are complete. Where numbers do not reconcile the team should be alerted and take steps to investigate
- Check surgical specimens labelled correctly
- Highlight equipment issues
- Verbalize plans or concerns for recovery and postoperatively, especially any specific risks
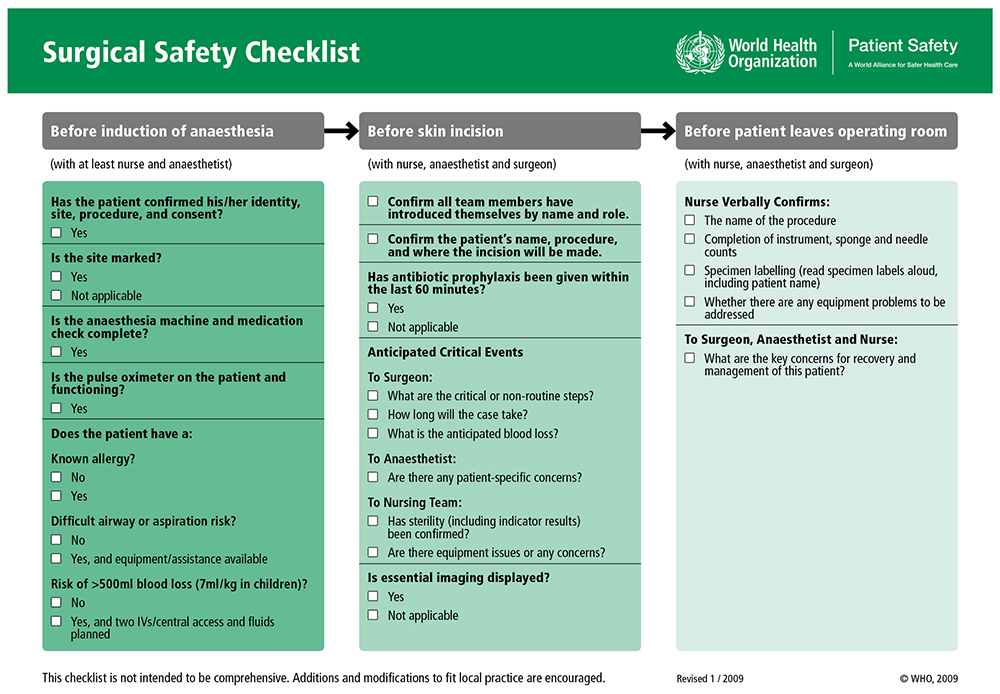
Figure 1. WHO Surgical Safety Checklist. Reproduced with permission of the World Health Organization
Implementing the checklist
The WHO issued an implementation manual in support of the checklist5. This gives detail on how each step should be conducted. The manual highlights the importance of leadership and institutional buy-in, and emphasizes that a department should practice using the checklist before introduction and should modify it so that it can be established within the normal operative workflow. Resources to help with implementation of the checklist are available on the WHO website: http://www.who.int/patientsafety/safesurgery/tools_resources/en/. Example videos from around the world can be seen on the SafeSurg website: http://www.safesurg.org/videos.html.
A single person should be responsible for checking the boxes on the list and this can be any healthcare professional in the operating team, often the circulating nurse. That nominated coordinator should prevent the team moving forward before each step has been addressed. Initially this could lead to tensions and resistance within the team, but only through consistently following the safety steps will the most common and avoidable risks be minimized.
Although facilities are encouraged to modify the checklist as needed, they are discouraged from removing safety steps simply because they cannot be accomplished. They also caution facilities from adding too many additional steps and creating an unmanageable, complex checklist. In England and Wales, the National Patient Safety Agency (NPSA) issued a patient safety alert in 2009. They launched a modified checklist for England and Wales with instructions to appoint a clinical lead within each organisation, ensure the checklist was completed for every patient undergoing a surgical procedure and that record of the checklist was entered into the patient notes8. A guide to modification of the checklist is available on the WHO website, as well as examples of modified checklists from around the world: http://www.who.int/patientsafety/safesurgery/local_adaptation/en/
Briefing and debriefing
The Patient Safety First Campaign was established to support implementation when the NPSA issued their alert informing England and Wales to use the checklist. Patient Safety First reported that some elements of the checklist could be more effective if incorporated into a briefing before the list starts. This is an opportunity to make a plan for the list, amongst all the team members, to anticipate and plan for any problems that can be foreseen. Any team member can lead the briefing, ensuring that everyone has introduced himself or herself and clarified their role and responsibilities for the list. An overview is taken of the list, highlighting any changes, equipment considerations, special requirements or safety concerns. All theatre team members should be present for the briefing and debriefing.
The debriefing naturally occurs at the end of the list, before any team members have left the theatre or department. The purpose of this debrief is to reflect on the list and share perspective on tasks that went well and tasks that did not go well. This may include discussion of teamwork, the theatre atmosphere, errors or near misses, and a retrospective look at the briefing and use of the surgical safety checklist throughout the day. It is important to register successes, learning points, areas that require change or escalation and for this to be conducted in a non-threatening, open environment. Patient Safety First developed and promoted the ‘Five Steps to Safer Surgery’9 (Figure 2)
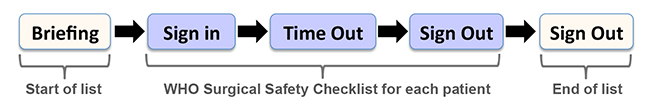
Figure 2: Five Steps to Safer Surgery
Barriers to implementation of the WHO checklist
Common themes that can hinder successful implementation of the checklist are listed in Figure 3. These barriers can be addressed to improve implementation outcomes9-11.
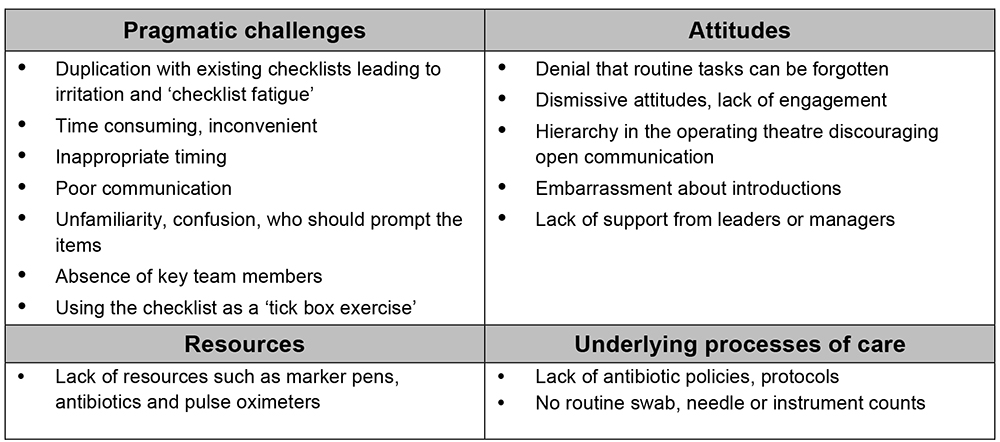
Figure 3: Table summarising barriers to checklist implementation
Top tips for successful implementation of the WHO Checklist
Implementation of the checklist can be a challenge, particularly when it is introduced as a new intervention, or top down mandate, or when the benefits are not well understood. After successful implementation, compliance can be one of the greatest challenges, either in terms of use of the checklist or completeness of the checks11. Below are pointers that may help to introduce the checklist and for it to be used effectively. These points are summarised below in figure 4.
Leadership
- Leaders in surgery, anaesthesia and nursing are very influential. It is important for leaders to embrace patient safety as a priority and to use the surgical safety checklist for their own cases. Senior members of staff should act as local champions on the ‘shop floor’, to support junior staff when they want to speak up or challenge an item, or simply to ask a question if they don’t understand something. These champions should be approachable, accessible and have skills in negotiation and persuasion. They need to create an honest, transparent culture and a baseline acceptance that we are all fallible and omissions can occur in any facility under anyone’s watch.
- It is important that the checklist is not mandated as a top-down chore for the staff, but that there is enthusiasm and engagement within the workforce, giving them good reason to engage. By using evidence from experience of near misses or adverse incidents, leaders can encourage transparency and honesty, and encourage teams to see the value of these routine checks.
- In addition to leaders and champions, it is important to engage administrative staff. New resources may be needed or simply a supply of paper for checklists in each theatre. Administrative support may also be required to ensure the antibiotic supply chain is established and that the proper equipment is available, including equipment to sterilise surgical instruments.
Implementation of team and staff training
- It is helpful to establish a local implementation team, with representatives from anaesthesia, surgery and nursing. This team should meet on a regular basis to plan introduction of the checklist.
- The implementation team should lead staff training, with in situ demonstrations, videos and coaching when they start to use the checklist. Training should be multi-professional, incorporating the whole team. This helps to flatten the hierarchy in theatre, and for many, it can be very revealing to see the world through the eyes of another.
- In addition to teaching sessions, it is helpful to raise awareness, for instance through posters, newsletters or computer screen savers.
- The implementation team should consider whether to implement the checklist in one area first or to introduce the checklist unit-wide. One example of effective implementation in Washington State described initially piloting the checklist in a small number of operating theatres. Due to the publication in newsletters of ‘poster child’ success, the other theatres were impatient to wait for official rollout and the checklist spread spontaneously12.
- Where an item on the checklist is not a routine practice in your facility, for instance, a team brief or de-brief, or pre-incision antibiotics or counting surgical instruments, focused training in that area will be needed. These items can be introduced in a stepwise approach, mastering one new item for a period, before adding a second new item.
- Retained swabs, needles or instruments are the most commonly reported serious adverse events in surgery. Training should incorporate the safety impact of such tasks so that staff are given reason to perform them and to recheck the patient if the count is not correct. It will be difficult to complete a surgical instrument count if there is no standardised pack or formal instrument list. Through generation of formal packs and lists, and routine counting out of equipment when it is placed on the surgical trolley, the hazards of retained swabs, needles and instruments can be reduced. All staff groups need to understand the importance of new checks added to practice, to avoid one group finding this a disruptive, time consuming intervention.
- Timely administration of antibiotics at least 15 but not more than 60 minutes before knife to skin (including in caesarean section) is an effective intervention to reduce surgical site infection, and anaesthetists can make an important contribution to reducing this complication. It is important to establish local antibiotic protocols and to make sure that these are adhered to.
- It is useful to encourage teams to communicate clearly. Checks need to be performed out loud for all of the operating team to hear. Avoid leading questions (the antibiotics have been given haven’t they?); rather use specific communication to a named individual (Question: Dr X: have you given the antibiotics? Dr X Answer: Yes, the antibiotics have been given).
Timing of briefing and surgical checks
- The ‘Five Steps to Safer Surgery’ helps to highlight issues at the start of the day and enables early resolution to minimize negative impact on theatre safety and throughput. If your facility is going to adopt this approach, it is helpful to define a fixed time for the pre-list briefing to occur. This will avoid team members arriving casually at different times, and thus inefficiency and resentment whilst waiting for other team members.
- Staff need to free themselves up from distracting tasks when the checks are being completed, ideally asking for ‘a surgical pause’ or ‘a moment of silence’ to gather everyone’s attention. In addition to being attentive, all members need to be present. It is helpful for the sign out to be completed whilst the surgeon is closing the wound as this integrates the checklist into the surgical process and ensures the surgeon is still present in theatre.
Resources and documentation
- Where an item cannot be completed, for instance due to lack of skin marker pens, pulse oximetry or antibiotics, checklist coordinators should not tick the item dishonestly. Use regular audit to document this need and feed this back to the department on a regular basis. Contact your hospital administrator so that theatre resources can be improved.
- Some facilities have found it useful to record the checklist information on a whiteboard or laminated paper in theatre, to refer to during the case. With operating team members changing frequently, staff names particularly may be easily forgotten and the team may find it helpful to display each staff member’s name.
- Where the checklist is not part of the computer system, give each theatre a folder with multiple paper copies. Use of the checklist should be documented in the patient record, for instance, on the anaesthetic chart.
- Routine pre-anaesthesia safety checks and the use of a pulse oximeter are part of the WHO Standards for Safe Surgery, also the WFSA International Standards for the Safe Practice of Anaesthesia 201013. The Lifebox Foundation has been established to facilitate access to pulse oximeters in low- and middle-income countries where these are not available (www.lifebox.org); if you do not have access to a pulse oximeter, please contact Lifebox and make yourself known.
Data collection and feedback
- Data is a powerful way to drive change in practice, and is an essential component of any quality improvement project. This can be an informal or formal process, paper based or electronic, depending on your local situation. The Royal College of Anaesthetists has published a useful introduction to quality improvement14, and on-line courses are available through the Institute of Healthcare Improvement (IHI) (http://www.ihi.org/Pages/default.aspx)
- Data can be collected in the form of ‘process measures’ – for instance, audit samples of the patient records on a weekly basis to see if the checklist has been completed or if antibiotics have been given before knife to skin. Ask a member of the team to observe in theatre to see if the checklist is being done, or to check whether all items on the checklist have been completed.
- ‘Outcome measures’ such as surgical start times, reason for delays, adverse events, near misses, and postoperative infections have been used to support the introduction of the checklist. Patient stories are a powerful way to motivate teams.
- The implementation team should feed this information back to the theatre team on a regular basis, ideally as ‘run charts’. A run chart is a simple plot of frequency of event (% patients with checklist completed, or antibiotics given) against time, so that the theatre team can see how they are performing each week or month. Consider comparing one theatre to another – competition is an effective driver for change. Use these results to stimulate discussion about why things work well, or to discover the barriers that prevent success.
- It is also important to present these data to the hospital administrative team (e.g. managers) so that recurring problems such as lack of resource or system issues can be addressed promptly. On the other hand, making them aware of improvement in patient outcomes will further incentivise management to endorse patient safety projects.
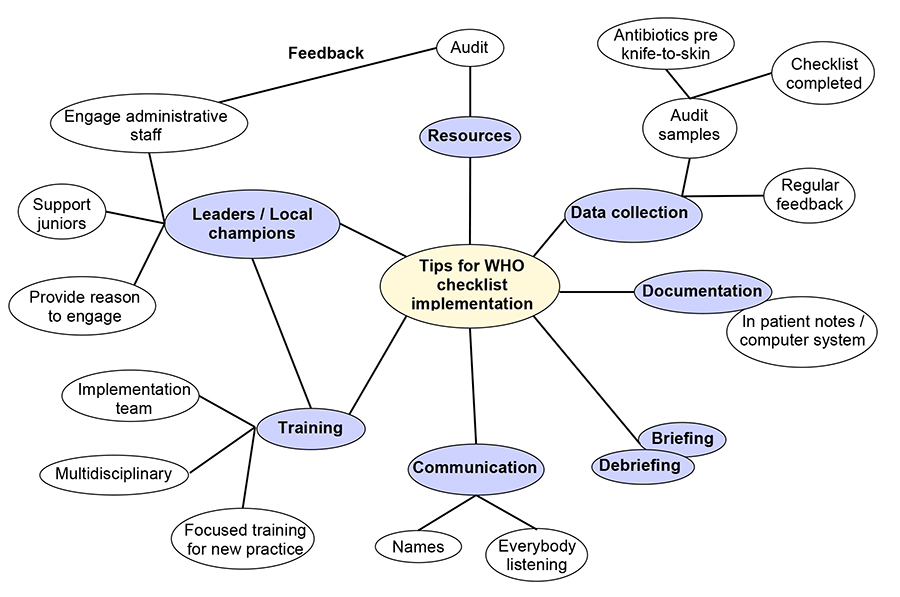
Figure 4: Diagram highlighting important steps in WHO checklist implementation
SUMMARY
Preventable harm occurs daily during surgery across the world. The WHO checklist was introduced as one means of reducing harm and improving patient safety in the operating theatre. With the benefit of hindsight, trials and audit, we have gained experience and identified the key factors that enable successful use of the checklist. These are senior multidisciplinary support, surgical buy-in, ensuring underlying processes of care are in place, and using local champions to enthuse and encourage staff.
The checklist needs to become part of routine surgical culture, even more so in an emergency or at the end of a long shift when simple tasks are easily forgotten. With consistent use, team members will become familiar with the checks, less embarrassed about using them, more time efficient, and break down the barriers to success. And ultimately, patient harm will be reduced.
ANSWERS
- a. False: Implementation of the checklist is a team effort. The team should represent everyone who works in theatre in order to get buy-in from all theatre personnel.
b. False: Experienced members of the theatre team who are committed to improving patient safety should lead the implementation process. Senior members of staff are very influential and need to be engaged; students are a very valuable resource and can help support the implementation process if they have support of the leaders in theatre.
c. False: Real time mentorship in theatre and continual feedback on progress of implementation is a powerful driver to influence change. It is useful to use regular observations and informal discussions on how things could be improved, rather than waiting until the end of an implementation period to evaluate the difficulties.
d. False: Safety steps should not be removed where they cannot be achieved. Examples include using a functioning pulse oximeter and administration of appropriate prophylactic antibiotics. Engage the administrative staff in the hospital so that all the items can be checked. This may also involve support from charities such as Lifebox.
e. True: Reporting and sharing stories of near misses or adverse incidents helps people to see how the checklist can be useful. Run charts of checklist completion rate can help people to see how they are doing with the ‘process’; audits of outcomes such as wound infections are more difficult to do, but can inspire a team to use safety checklists. - a. True
b. False: It is even more valuable to use the WHO checklist in an emergency as simple safety checks can easily be forgotten in a pressurised, urgent environment.
c. False: All staff members should feel able to raise questions and talk without fear or embarrassment. This can be encouraged by creating an open, non-hierarchal environment.
d. False: Antibiotics should be given 15- 60 minutes prior to the skin incision.
e. True: ‘Counting’ surgical swabs and instruments is an important part of modern surgical nursing. It is easier if there are standardised numbers of packs used (for instance, swabs are put on the surgical trolley in packs of 5) and a standard list of instruments so that they can be checked off at the end of the operation. - a. False: The briefing is held before the start of the list and debriefing at the end of the list rather than before and after every case.
b. True
c. False: A pre-list briefing can be used to pre-empt or trouble shoot equipment or safety issues and anticipate challenges for the list. The whole team should be present for pre-list briefing.
d. False: The briefing should take around 10 minutes, but will save delays throughout the day.
e. False: When a list has run safely, efficiently and uneventfully, it is useful to look at the team behaviours during that list that contributed to success. By verbalising what went well the team can actively take those positive strategies into their next list.
REFERENCES AND FURTHER READING
- Weiser TG, Regenbogen SE, Thompson KD, Haynes AB, Lipsitz SR, Berry WR, et al. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008;372(9633):139-44.
- de Vries EN, Ramrattan MA, Smorenburg SM, Gouma DJ, Boermeester MA. The incidence and nature of in-hospital adverse events: a systematic review. Qual Saf Health Care. 2008;17(3):216-23.
- National Reporting and Learning System. [cited 2015 21st July]. Available from: http://www.nrls.npsa.nhs.uk/resources/collections/quarterly-data-summaries/?entryid45=135410.
- Organization WHO. WHO guidelines for safe surgery: safe surgery saves lives 2009 [cited 2015 21st July]. Available from: http://www.who.int/patientsafety/safesurgery/tools_resources/9789241598552/en/.
- Organization WHO. World alliance for patient safety: Implementation manual surgical safety checklist 2008 [cited 2015 21st July]. First edition:Available from: http://www.who.int/patientsafety/safesurgery/ss_checklist/en/.
- Haynes AB, Weiser TG, Berry WR, Lipsitz SR, Breizat AH, Dellinger EP, et al. A surgical safety checklist to reduce morbidity and mortality in a global population. N Engl J Med. 2009;360(5):491-9.
- Centre for Geographic Analysis HU. Patient Safety: Surgical Safety Web Map 2014 [cited 2015 21st July]. Available from: http://maps.cga.harvard.edu/surgical_safety/.
- Agency NPSA. WHO Surgical Safety Checklist 2009 [cited 2015 21st July]. Available from: http://www.nrls.npsa.nhs.uk/resources/?entryid45=59860.
- Agency NPSA. ‘How to Guide’ Five Steps to Safer Surgery 2010 [cited 2015 25th July]. Available from: http://www.nrls.npsa.nhs.uk/EasySiteWeb/getresource.axd?AssetID=93286
- Tang R, Ranmuthugala G, Cunningham F. Surgical safety checklists: a review. ANZ J Surg. 2014;84(3):148-54.
- Aveling EL, McCulloch P, Dixon-Woods M. A qualitative study comparing experiences of the surgical safety checklist in hospitals in high-income and low-income countries. BMJ Open. 2013;3(8):e003039.
- Conley DM, Singer SJ, Edmondson L, Berry WR, Gawande AA. Effective surgical safety checklist implementation. J Am Coll Surg. 2011;212(5):873-9.
- Merry AF, Cooper JB, Soyannwo O, Wilson IH, Eichhorn JH. International Standards for a Safe Practice of Anesthesia 2010. Can J Anaesth. 2010;57(11):1027-34.
- Quality Improvement in Anaesthesia: Royal College of Anaesthetists; 2012 [cited 2015 3rd September] Available from: https://www.rcoa.ac.uk/content/searchcx=009352006448159467736%3Adf2oiygfg8q&cof=FORID%3A11&ie=ISO-8859-1&query=quality+improvement+in+anaesthesia



