Paediatric Anaesthesia
KEYPOINTS
- Anaesthesia for children with complex cardiac disease in the remote environment of a cath lab can be challenging.
- Ketamine is a key drug for patients with impaired cardiac function.
- Recurrent chest infections are common in children with cardiac disease, especially in those with excess pulmonary blood flow. Respiratory infections should be treated before proceeding with elective cases.
- Fasting should be kept to a minimum and dehydration avoided.
- It is very easy for the child to get cold. Maintain normothermia with active warming.
INTRODUCTION
Diagnostic and minimally invasive interventions in the cardiac catheterisation laboratory (cath lab) are now routine in the management of children with congenital heart disease (CHD). Children may present at any age, including as neonates, for elective or emergency procedures. The majority of procedures require the child to be anaesthetised. It is important to understand the cardiac anatomy and physiology of each child, and the potential risks, benefits and complications of the interventions being undertaken. Effective communication within the team, and constant vigilance are essential when managing these complex cases. With good clinical care, most patients can be discharged to a cardiac ward at the end of the procedure, making treatment of children in the cath lab a realistic option for all patients, even in resource limited countries.
GENERAL PRINCIPLES
Equipment
Cardiac cath labs tend to be situated in remote areas of the hospital. In adults, procedures are often performed under local anaesthesia or sedation and as such, immediately available anaesthesia resources or equipment may not always be optimal. It is more common for children to require a general anaesthetic and therefore essential that the same standard of anaesthetic care is provided as elsewhere in the hospital. The anaesthetist must be familiar with, and check, the anaesthetic equipment pre-operatively.1
Basic monitoring is fundamental and must include as a minimum, working pulse oximetry, electrocardiography (ECG), and blood pressure (BP) monitoring. A working defibrillator with paediatric paddles or attenuated pads must be available. The operator should be trained to use this device.1
The cath lab is often cold and the patient may be exposed when setting up the case. It is easy for children to become hypothermic. This may promote arrhythmias and will delay recovery from anaesthesia. Active warming using an air blanket is ideal, and at minimum, the patient should be kept covered and the room temperature controlled to suit the patient needs. 1
Echocardiography (ECHO) is essential in the cath lab. It is used to diagnose or confirm the cardiac lesion, measure pressure gradients, check positioning of deployed devices and assess for cardiac injuries following an intervention (e.g. tamponade or new valvular regurgitation).
Pre-operative Investigations
All patients should have a full blood count pre-operatively. For interventional procedures a valid group and save should be available, and for higher risk procedures e.g. balloon dilatation of valves, blood should be cross-matched and immediately available. Blood sugar, electrolytes, renal function, and liver function should be checked as indicated locally, but particularly in patients with significantly impaired cardiac function or those with complex co-existing diseases. Thrombocytopenia may be seen in patients with severe hypoxia and compensatory polycythaemia. Cardiac catheterisation should ideally take place when the platelet count is over 100 x 109/L, which may require transfusion for this to be achieved.
A 12-lead pre-operative ECG is important, particularly in children with known arrhythmias, and acts as a reference should changes develop.
Fasting
Patients should be fasted pre-operatively, but the most common problem in all parts of the world is that clear fluids are withheld for too long. Prolonged starvation for fluids will make the child miserable, more likely to be hypoglycaemic, and if dehydrated, more difficult to cannulate. It is particularly important to avoid dehydration for patients with severe hypoxia who are polycythaemic as this increases the risk of thrombotic complications.2,3 Patients late on the list may become increasingly dehydrated during their wait and clear instructions of type and quantity of fluid allowed should be provided (See ATOTW Tutorial 352 for further information and recommended fasting times).
Team brief at the start of the list
As with all lists, it is good practice to start the list in the cath lab with a ‘team brief’.4 This is an opportunity for members of the team to introduce themselves, to review each case on the list and to confirm the list order (see ATOTW Tutorial 325). At Great Ormond Street Hospital we discuss the following at the start of each cath list:
- Review all patients on the list and discuss any specific patient concerns
- Confirm if cross-match is required/available
- Any special equipment required, including invasive monitoring
- Imaging requirements
- Antibiotic requirements
- Warming
- Who is operating and who is assisting
- Any other issues such as staffing or time
Anaesthetic technique
An anaesthetic technique should be chosen that enables a safe and rapid recovery of the patient and return to the ward post-operatively. This will be determined by local preference. Balanced anaesthesia with controlled ventilation, with or without use of a muscle relaxant is the usual technique of choice. Long-acting muscle relaxants such as pancuronium are best avoided. Spontaneous ventilation without intubation may be an option, and often used in resource limited countries for short cases where ketamine can be used as the sole agent. Deep volatile anaesthesia, particularly halothane, is best avoided in order to maintain haemodynamic stability. 5,6
It is essential to obtain secure vascular access, despite pressure from others to proceed. Failure to secure good vascular access may be regretted later if a serious adverse event occurs. In some children, vascular access can be achieved on the ward beforehand and will save valuable cath lab time. Alternatively, access can be achieved following induction of anaesthesia with an inhalational agent or ketamine (5-10mg/kg IM or 2mg/kg IV).
During catheterisation, direct cardiac and arterial pressure readings can be measured as part of the diagnostic work up and repeated to assess for post intervention changes. The fractional inspired oxygen concentration [FiO2] delivered can have significant effects on the pulmonary vascular resistance, and as such will affect pressure readings taken during catheterisation. Following a safe induction, aim to deliver the same oxygen requirement the patient would normally be exposed to (i.e. close to [FiO2] 0.21 if spontaneously ventilating in air), but do liaise with the cardiologist during the brief if in doubt. 5,6
Maintenance fluids
Isotonic maintenance fluids such as Ringer’s Lactate or Hartmann’s should be used for all patients. If there are concerns about hypoglycaemia (e.g. prolonged starvation), 50% glucose 10 – 50ml can be added to a 500ml bag of fluid to make a solution of 1-5% glucose/Ringer’s (or Hartmann’s). Blood glucose levels should be measured in this situation. There is no place for hypotonic maintenance fluids in modern paediatric anaesthesia practice. 7
Post-operative care
The child should be fully recovered before returning to the ward, ideally after a period of time in a staffed recovery room, with oxygen, suction and monitoring available. The vascular puncture site must be checked for bleeding prior to return to the ward, particularly where arterial access has been obtained. 5,6
SPECIFIC SITUATIONS
Pulmonary hypertension
Pulmonary hypertension (PH) is diagnosed when the resting mean pulmonary artery pressure (mPAP) exceeds 25 mmHg. Following induction of anaesthesia, FiO2 should be reduced to as close to room air as possible or the minimum FiO2 the patient can tolerate safely to allow PAP measurements. 100% O2 is then given for 10 minutes and PAP is measured again to assess for evidence of reversibility of PH, defined as a 15% reduction in mPAP versus mean blood pressure (MAP) [without a significant change in MAP]. FiO2 must be increased during testing if there is profound hypoxia or any ischaemic changes are seen. 8,9
Tetralogy of Fallot (TOF)
In many situations full diagnostic workup for patients with TOF is achieved using ECHO. In some centres, particularly where ECHO skills are not available, cardiac catheterization may be carried out to:
- Assess the course and severity of the right ventricular outflow tract (RVOT) obstruction
- Map the coronary blood supply and assess for an aberrant coronary artery crossing the infundibulum of the right ventricle (which could be jeopardised during surgery if not recognised)
- Identify major aorto-pulmonary collateral arteries (MAPCAs)
The major risk during cardiac catheterization of a child with unrepaired TOF is a hypercyanotic ‘spell’, particularly if the angiography catheter stimulates the infundibular area of the RVOT. The infundibular spasm is due to contraction of muscle bundles in the RVOT, particularly if the sympathetic tone is high (beta-1 adrenoceptor). This results in increased right to left shunting and profound hypoxaemia. The risk of spelling can be minimised by reducing sympathetic drive, including premedication, delivering balanced anaesthesia with opioid analgesia, and importantly avoiding ‘light’ anaesthesia. Epinephrine should be avoided as this will worsen any RVOT obstruction. 5,6,10
A cyanotic spell should be treated in the following way:
- Increase the FiO2
- Give an IV fluid bolus of 10ml/kg isotonic fluid, reassess and repeat if necessary. This will increase the cardiac output and pulmonary blood flow.
- Phenylephrine (1 mcg/kg boluses) or norepinephrine infusion (0.05-0.1mcg/kg/min). These are pure alpha adrenoceptor agonists – they increase systemic vascular resistance (SVR) which reduces right to left shunting which in turn will increase blood flow through the RVOT.
- Beta-blockers e.g. esmolol (0.5mg/kg IV over 1 min, repeat if required, or infusion 25-300mcg/kg/min) or metoprolol (0.1 mg/kg IV over 5 min, max 5mg). This should relieve the infundibular spasm and reduce RVOT obstruction. 10

Single Ventricles
Children with single ventricle physiology (e.g. hypoplastic left heart syndrome, tricuspid atresia) require a series of palliative procedures for long term survival. After initial palliation in the neonatal period to secure systemic and pulmonary blood flow, further procedures are required to ensure adequate pulmonary blood flow as the child grows. The bidirectional Glenn (BDG) shunt is formed in infancy by anastomosing the superior vena cava (SVC) to the pulmonary artery, and at around 2-4 years of age, the total caval pulmonary connection (TCPC or Fontan circulation) is formed by anastomosing the inferior vena cava (IVC) to the pulmonary artery via an interposition graft. Venous return from the SVC and IVC then provides pulmonary blood flow in a fully ‘in series’ circulation with the single ventricle as the only pump. Successful TCPC completion requires the PAP to be low, as the driving pressure for pulmonary blood flow is the pressure in the SVC/IVC.11
A diagnostic cardiac catheter may be carried out as part of the workup for a TCPC. The anaesthetist may be asked to insert an internal jugular vein catheter to measure the pressure in the BDG to estimate the PAP. Ideally, children with single ventricle physiology for catheter procedures should be kept ‘well filled’ to facilitate venous return and hence pulmonary blood flow. It is very important to avoid prolonged starvation and dehydration in this patient group. During anaesthesia the patient should be intubated and positive pressure ventilation provided. Small tidal volumes (≤7ml/kg) should be set to avoid increased pulmonary vascular resistance (PVR) seen with large tidal volumes. Additionally, end tidal CO2 concentrations [EtCO2] should be controlled to avoid unwanted PVR changes from hyper or hypoventilation, and importantly, to ensure adequate venous return from the cerebral circulation into the SVC.
Transoesophageal echocardiography (TOE)
TOE is used to guide interventions, such as secundum ASD device placement, or to improve diagnostic information when trans-thoracic echocardiography is found to have lower diagnostic capability e.g. sinus venosus ASD. Tracheal intubation and controlled ventilation is required. A diagnostic TOE is generally a short procedure requiring only a brief period of intubation. An intermediate acting muscle relaxant to facilitate intubation e.g. atracurium could be used, or if unavailable, suxamethonium, which is frequently used in resource limited countries. Some centres may have the benefit of having rocuronium and sugammadex available. 5,6,10
INTERVENTIONAL PROCEDURES
Interventional procedures must only be conducted in centres where surgical back up is immediately available to assist with complications e.g. embolisation of a septal occluder device or significant trauma to a cardiac structure. 5,6,10,12
Blood should be cross-matched and immediately available for interventional procedures. Most interventions aim to either close an open or unwanted defect, or enlarge or create a defect. Interventional catheters have larger diameters than those used in diagnostic procedures and carry an increased risk of vascular injury.
Common interventions performed in the cath lab include:
- PDA closure
- ASD closure
- Pulmonary valvotomy
- Aortic valvotomy
- Atrial septostomy
- Vascular stents (coarctation, pulmonary vessels)
- Occlusion of collateral vessels
- Pulmonary valve insertion
Patent ductus arteriosus (PDA) closure
A patent ductus arteriosus (PDA) with left to right shunting leads to pulmonary overflow and signs and symptoms of congestive heart failure (Figure 1). Patients suffer from repeated chest infections and failure to thrive. Small isolated PDAs in infants and older children are amenable for device closure in the cath lab. Most anaesthetic agents reduce SVR and shunting across the PDA. Avoid high FiO2 in these patients (if possible), as O2 reduces PVR and increases pulmonary blood flow across the duct. We recommend a balanced anaesthetic technique with intubation and ventilation although it is also possible to manage such procedures using ketamine in a spontaneously breathing patient. 5,6,10,12
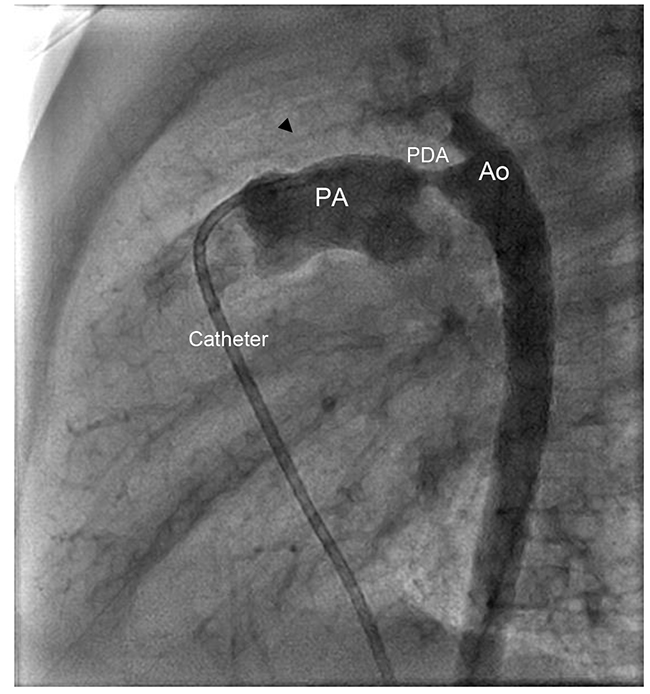
Figure 1. Cardiac Catheter Imaging: a patent ductus arteriosus (PDA: patent ductus arteriosus; PA: pulmonary artery; Ao: Aorta)
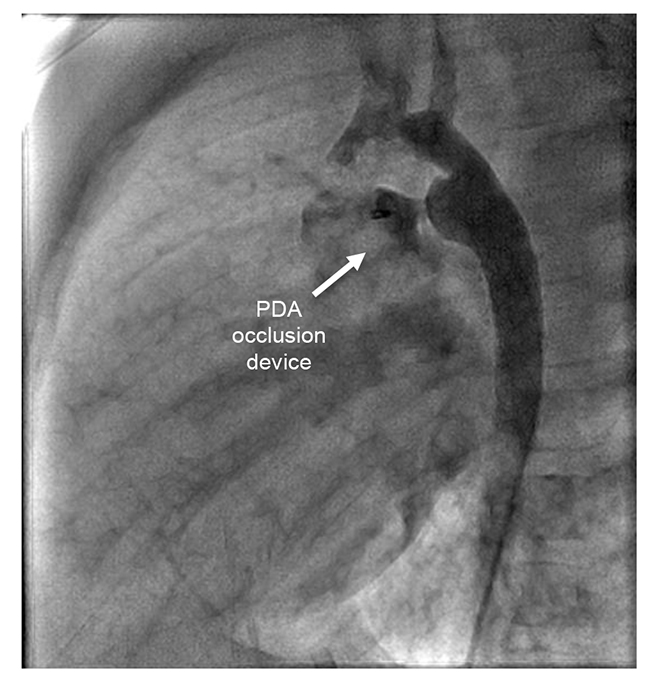
Figure 2. Cardiac Catheter Imaging: Post patent ductus arteriosus device closure
Atrial septal defect (ASD) closure
ASD can be closed percutaneously under angiographic and TOE guidance (Fig 3). Indications for ASD closure using a percutaneous device is based on clinical and ECHO findings. Specifically, assessment of size, anatomy and location of the ASD will determine if the procedure is feasible. Indications for elective closure include those with evidence of excess pulmonary overflow from a significant shunt [pulmonary flow [Qp] : systemic flow [Qs] ratio > 1.5) and patients with right ventricular overload. 5,6
Prophylactic intravenous antibiotics should be given within 30 minutes of the start of the procedure and the patient anticoagulated with heparin according to local protocol. If the ASD is deemed unsuitable for device closure following angiography, the heparin should be reversed with protamine, guided by the activated clotting time (ACT). Particular care must be taken to check for bleeding after the procedure if a large sheath has been used and the puncture site should be compressed for at least 10 minutes. ASD device closure is associated with arrhythmias from catheter manipulation, trauma to cardiac structures and air embolism. If embolisation of the septal occluder occurs, the device will need to be retrieved urgently by the interventional cardiologist, and may involve cardiothoracic surgery if this is not achievable percutaneously. 5,6,10,12
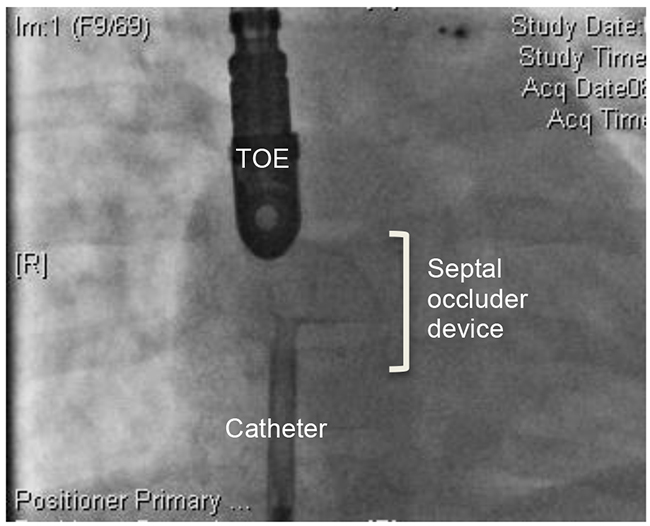
Figure 3. Cardiac Catheter Imaging: Atrial septal defect (ASD) occlusion guided by transoesophageal echocardiography (TOE). All images kindly provided by the Department of Cardiology, Great Ormond Street Hospital.
Pulmonary Valvotomy
Significant pulmonary valve (PV) stenosis, defined as a pressure gradient across the PV greater than 50againmmHg is an indication for pulmonary valvotomy. The PV area may be very small or slit like and the right ventricular (RV) outflow is easily occluded when catheterised. During this time, the patient will become profoundly desaturated as the pulmonary blood flow is obstructed. The catheter should be withdrawn until the SpO2 normalises. A balloon tipped catheter is inserted under fluoroscopic control and the balloon inflated momentarily to split the commissure between the valve leaflets. A post procedure pressure gradient across the PV below 40 mmHg is usually acceptable. 5,6,10
Aortic valvotomy
Children with significant aortic stenosis (AS) will have left ventricular hypertrophy and may demonstrate evidence of reduced left ventricular function. SVR must be maintained in patients with AS to prevent coronary ischaemia. Aortic valvotomy via balloon dilatation is indicated if:
- Aortic valve (AoV) pressure gradient > 70 mmHg
- AoV pressure gradient > 50 mmHg with symptoms such as chest pain, or if there is evidence of ischaemia on ECG
During placement of the balloon tipped catheter across the valve, blood flowing through the left ventricular outflow tract may displace the balloon device away from the AoV. Rapid overdrive pacing can be used to reduce cardiac output during this procedure to reduce the cardiac output acutely and improve the success of the procedure. Severe aortic incompetence (AI) from over dilatation of the AoV can result in coronary insufficiency. 5,6,10,12
Coarctation of the aorta (CoA)
Severe coarctation of the aorta usually requires surgery in early infancy, but moderate degrees of coarctation or residual stenosis after initial surgical treatment can be treated in the cath lab. In suitable young patients, CoA is usually treated with balloon angioplasty. In older children, a stent may be placed across the coarctation if the anatomy is amenable. Complications include aortic rupture, aortic dissection, cerebrovascular events, femoral artery trauma, thrombosis and aneurysm formation. Cross matched blood must be available. 5,6,10,12
Older children often have upper limb hypertension so it is sensible to place the blood pressure cuff or an arterial line on the right arm to obtain a true measure of the blood pressure. Anaesthesia for treatment of aortic coarctation should aim to maintain haemodynamic stability, but not reduce arterial blood pressure excessively. In young children, inhalational anaesthesia with small doses of opioid e.g. fentanyl (1-2 mcg/kg) or ketamine (0.5mg/kg), is usually a safe combination. Total intravenous anaesthesia techniques may be suitable in older children.
Balloon Atrial Septostomy (BAS)
The main indication for atrial septostomy is to improve mixing between systemic and pulmonary circulations in neonates with dextro-transposition of the great arteries (d-TGA) and insufficient oxygenation, prior to corrective surgery. Neonates with TGA usually present with hypoxia and acidaemia. Those with better mixing, e.g. a patent ductus arteriosus (PDA) have a better chance of survival. BAS may also be used to improve mixing in other life threatening cyanotic CHD e.g. pulmonary atresia with intact ventricular septum, tricuspid atresia, and hypoplastic left heart syndrome (HLHS). A balloon-tipped catheter is passed across the foramen ovale from the right atrium to the left atrium and pulled back with the balloon inflated to create the septostomy. The procedure is carried out under ECHO guidance with or without sedation, and is well-tolerated in skilled cardiology hands. 5,6,10
Ventricular septal defect (VSD) closure
VSD device closure is complex and requires general anesthesia with tracheal intubation and TOE guidance. There is a high risk of cardiovascular instability, and long operating time and should only be undertaken in experienced centres. 10
Cardiac tamponade
Cardiac tamponade impairs venous return and leads to a reduction in cardiac output (CO). Maintaining heart rate is crucial to avoid rapid decompensation. Vasoactive drugs (atropine and epinephrine) should be prepared in advance. Children with severe tamponade are very sick and ketamine is the anaesthetic drug of choice. Maintenance of preload is crucial in tamponade. Spontaneous breathing is better tolerated than positive pressure ventilation as this may impede venous return further, increase RV afterload, and further reduce CO. After draining the tamponade (usually using a catheter) a sudden return in pulmonary blood flow may precipitate pulmonary oedema. Invasive monitoring is essential for these high-risk cases. 5,6,10,12
Electrophysiology (EP) Studies
Patients who have symptomatic cardiac arrhythmias due to pathological conduction pathways can be treated successfully in the cath lab with radiofrequency ablation or cryoablation of the abnormal conduction pathways. Patients will have stopped their anti-arrhythmic drugs prior to the study, but anaesthesia suppresses endogenous sympathetic drive, and provocation of the arrhythmia during the procedure can be more difficult. There is a fine balance to ensure the patient is fully anaesthetised, not too deep under the anaesthetic but importantly not aware. On table arrhythmias e.g. ventricular fibrillation or sustained ventricular tachycardia, whilst uncommon, may occur and must be managed with defibrillation. Supra-ventricular tachycardias may respond to adenosine, but must be cardioverted if the patient is compromised. General anaesthesia with tracheal intubation is necessary for EP studies as these are long procedures with multiple large intravascular catheters. Propofol based total intravenous anesthesia is a popular technique. Sevoflurane with judicial use of opioids is an alternate option. Dexamethasone limits the size of the radiofrequency ablation ‘burn’ and so should not be used as a routine antiemetic in these patients. 5,13
COMMON COMPLICATIONS SEEN IN THE CATH LAB
Anaesthesia related complications
Patients may have limited pulmonary and cardiovascular functional reserve. Respiratory depression and hypoxia occur quickly following induction of anaesthesia, especially in neonates and infants. Adequate monitoring of breathing is essential throughout the case.
All anaesthetic agents are direct cardiovascular depressants. With ketamine, its direct myocardial depressant activity is offset by its stimulatory effect via catecholamine reuptake inhibition. However, ketamine is associated with emergence delirium. This can be ameliorated with midazolam in older patients. Opioids such as fentanyl are an excellent option as part of a multi-modal technique but are commonly associated with post-operative nausea and vomiting and anti-emetics should be used routinely in older children. 5,6
Environment related complications
The cath lab is a cold environment and hypothermia can develop easily, especially in neonates and infants. Active warming should be used, with temperature recorded throughout the case. Hypothermia will delay recovery from anaesthesia and may lead to arrhythmias. 5,6
Procedure-related complications
Vascular access with large sheaths can compromise distal perfusion and may result in poor perfusion or even ischaemia of the lower limbs. Arrhythmias occur frequently and are mostly due to the presence of intracardiac catheters. These usually resolve with catheter withdrawal. Other causes include electrolyte disturbances, hypercapnia or coronary air embolism. Contrast is nephrotoxic and may precipitate an allergic reaction, and occasionally intractable coughing. It is important to limit the contrast load as far as possible.
Other less common complications include embolisation of the device, catheter fracture, valvular trauma, cardiac tamponade, vascular trauma, and bleeding at femoral access sites. Most complications are dealt with in the cath lab but occasionally these more severe complications require emergency surgery. 5,6
RADIATION SAFETY
It is important for anyone who works in the cath lab to be aware of radiation safety and the risks posed to patients and healthcare workers. 5,14 National guidance on radiation safety should be followed. All girls of childbearing age should have their pregnancy status checked. The radiation dose to patients and staff should be minimized as much as possible. The factors that affect radiation dose are distance, barriers and exposure time:
- Distance: Radiation dose is reduced with increased distance from the source. The anaesthetist may be exposed to high doses of radiation if they remain close to the patient during screening.
- Barriers: Protective clothing such as lead aprons, thyroid collar and protective eye glasses must be worn. The threshold exposure for cataract formation may be reached in a few years without appropriate precautions.
- Time: Screening using fluoroscopy greatly increases the exposure hazard for the cardiologist and the anaesthetist. The maximum radiation exposure recommended for medical workers is 5rem/year. Ideally exposure should be much lower than this. Exposure during cine fluoroscopy may be particularly high so it is important to wear protective lead and stay as far as possible from the radiation source during screening.
SUMMARY
- It is important to understand the individual cardiac anatomy and physiology of each child to deliver the safest and most effective balanced anaesthetic in the cath lab.
- Liase with the cardiologist to understand their requirements for each case which will guide any anaesthetic measures necessary (e.g. ventilate with room air). Ensure any measures taken will not compromise patient safety. As with all cases, communication is key.
- Increasing diagnostic and interventional techniques are possible in the cath lab today, and like other remote anaesthetic sites, the full range of safety checks, including blood availability, must be in place for any unwanted complication might occur.
REFERENCES AND FURTHER READING
- Association of Anaesthetists of Great Britain and Ireland. Recommendations for standards of monitoring during anaesthesia and recovery 2015. Anaesthesia 2016; 71: 85-93
- Anderson H, Zaren B, Frykholm P. low incidence of pulmonary aspiration in children allowed intake of clear fluids until called to the operating suite. Paediatr Anaesth. 2015 Aug;25(8):770-7
- APA consensus guideline on perioperative fluid management in children: V 1.1 September 2007
- Organization WHO. WHO guidelines for safe surgery: safe surgery saves lives 2009 [cited 2017 1st April]. Available from: http://www.who.int/patientsafety/safesurgery/tools_resources/9789241598552/en/
- Qureshi S, Pushparajah K, Taylor D. Anaesthesia for paediatric diagnostic and interventional cardiological procedures, Contin Educ Anaesth Crit Care Pain 2015;15(1):1-6.
- Lam J, Lin E, Alexy R et al. Anesthesia and the pediatric cardiac catheterization suite: a review. Pediatric anesthesia 2015: 25(2)127–134.
- National Institute for Health and Care Excellence. Intravenous fluids therapy in children. (Clinical guideline NG29.) 2015.
- Tempe DK. Perioperative management of pulmonary hypertension. Annals of Cardiac Anesthesia 2010; 13(2):89-91.
- Goyal P, Kiran U, Chauhan S et al. Efficacy of nitroglycerin inhalation in reducing pulmonary arterial hypertension in children with congenital heart disease. British Journal of Anesthesia 2006; 97(2): 208-14.
- Reddy K, Jagger S, Gillbe C. The anaesthetist and the cardiac catheterisation laboratory, Anaesthesia 2006; 61:1175-1186.
- Feinstein JA, Benson DW, Dubin AM, et al. Hypoplastic Left Heart Syndrome: Current Considerations and Expectations. J Am Coll Cardiol 2012; 59 Suppl S:S1–S42.
- Thangavel P, Muthukumar S, Karthekeyan et al. Anaesthetic Challenges in Cardiac Interventional Procedures, World Journal of Cardiovascular Surgery, 2014, 4, 206-216.
- Ashley E. Anaesthesia for electrophysiology procedures in the cardiac catheter laboratory. Contin Educ Anaesth Crit Care 2012; 12(5):230-236.
- Taylor J, Chandramohan M, Simpson K. Radiation safety for anaesthetists. Contin Educ Anaesth Crit Care Pain 2013; 13(2):59-62.



