General Topics
KEY POINTS
- Hyperthermic intraperitoneal chemotherapy (HIPEC) is a technique which involves infusing chemotherapy heated to 42°C-43°C into the peritoneal cavity.
- It is usually performed following extensive cytoreductive surgery (CRS).
- In certain malignancies, it is associated with an improvement in the median 5-year survival.
- Marked intraoperative physiological changes due to the temperature and large-volume fluid shifts pose significant anaesthetic challenges.
- The use of chemotherapeutic agents in the operating theatre poses potential safety issues.
- Patients need to be carefully monitored for postoperative complications.
INTRODUCTION
Hyperthermic intraperitoneal chemotherapy (HIPEC) is a highly concentrated, heated chemotherapy treatment that is delivered directly to the abdomen during surgery. HIPEC combined with cytoreductive surgery (CRS) has developed over time as an effective multimodal treatment option for selected patients with peritoneal surface malignancies. Traditionally, these types of malignancies were considered incurable conditions suitable for palliation only. HIPEC with CRS improves the median survival as well as the 5-year survival rate for these patients. The technique itself involves infusing chemotherapy heated to 42°C-43°C into the peritoneal cavity following macroscopic resection of visible tumour. This procedure can lead to physiological and potentially pathological changes with implications for perioperative care.
HISTORY
The management of peritoneal malignancies has progressed and advanced over the decades as CRS, HIPEC, and the perioperative care of these patients have evolved over time (Table 1).

Table 1. Timeline of Developments in Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy1
SURGICAL INDICATIONS
Indications are shown in Table 2.
PATIENT SELECTION
Appropriate selection of patients in order to achieve complete cytoreduction is key to a successful outcome (Table 3). Good baseline health without significant cardiac or respiratory disease and age under 70 are ideally required, however, not absolutely indicated. Patients presenting for CRS and HIPEC should have no disease progression on chemotherapy whilst awaiting surgery. Scoring systems such as the peritoneal cancer index described by Sugarbaker and the more recent peritoneal surface disease severity score are used to assess degree of peritoneal disease, which is used to help prognosticate outcome.4
PREOPERATIVE ASSESSMENT
CRS combined with HIPEC is an invasive abdominal surgical procedure with additional intraoperative temperature changes and fluid shifts over and above conventional laparoscopic or open surgical approaches. The pathophysiological effects associated with this procedure can precipitate organ failure in some patients. It is imperative to perform a detailed preoperative assessment in patients undergoing this procedure. The cardiac risk in these patients is similar to the risk of other major

Table 2. Surgical Indications for Cytoreductive Surgery (CRS) and Hyperthermic Intraperitoneal Chemotherapy (HIPEC)2,3

Table 3. Selection Criteria for Hyperthermic Intraperitoneal Chemotherapy (HIPEC) and Cytoreductive Surgery (CRS)
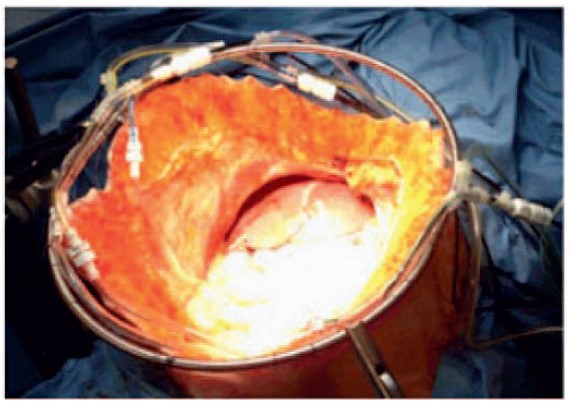
Figure 1. Open technique. (From Halkia et al, ‘‘Peritoneal carcinomatosis: intraoperative parameters in open (Coliseum) versus closed abdomen HIPEC’’5; used with permission.)
abdominal surgery. Thorough assessment of the patient’s cardiac and pulmonary systems is essential to assess their ability to respond to the physiological challenges faced intraoperatively. Echocardiography and dynamic cardiac assessments may be required. Full blood count, coagulation studies, and electrolyte, urea, and creatinine levels should be obtained. It is also important to review the nutritional status of the patient and to measure a preoperative albumin level. Estimation of the glomerular filtration rate may help identify patients at risk of an acute kidney injury associated with the HIPEC.3
SURGICAL TECHNIQUES
The procedure is divided into 3 stages:
- Exploration: The initial step may begin with an exploratory laparoscopy to assess for disease progression or proceed directly to opening the abdomen through a full-length midline laparotomy incision and evaluation of the peritoneal cancer.
- Cytoreduction/debulking: In the debulking, or cytoreduction, phase of the procedure, the surgeon will remove the visible/ macroscopic tumour. This may also involve organ removal.
- Chemoperfusion: Here the abdominal cavity is rinsed with a heated chemotherapy solution.
Cytoreductive surgery can vary from an isolated omentectomy to complete resection of multiple abdominal organs including the gastrointestinal tract, pancreas, spleen, liver portions, gallbladder, ovaries, and uterus. Peritonectomy is also commonly performed and this can be associated with increased intraoperative blood loss. The HIPEC can be delivered in one of two ways—the open (or Coliseum) or closed techniques.5
The open technique (Figure 1) ensures optimal distribution of heat and cytotoxic solution due to manual stirring of the abdominal contents; however, the technique may be compromised due to heat loss (requiring an increase in the temperature of the perfusion fluid, thus exposing the bowel to the risk of scald injuries) or suboptimal exposure of the anterior parietal wall as well as the risk of spillage of cytotoxic drugs.5,6
The closed technique (Figure 2) prevents heat loss associated with the open technique, minimizes drug spillage, and increases drug penetration, but homogeneous distribution of the perfusion fluid may not be achieved.5,6 The duration of the HIPEC
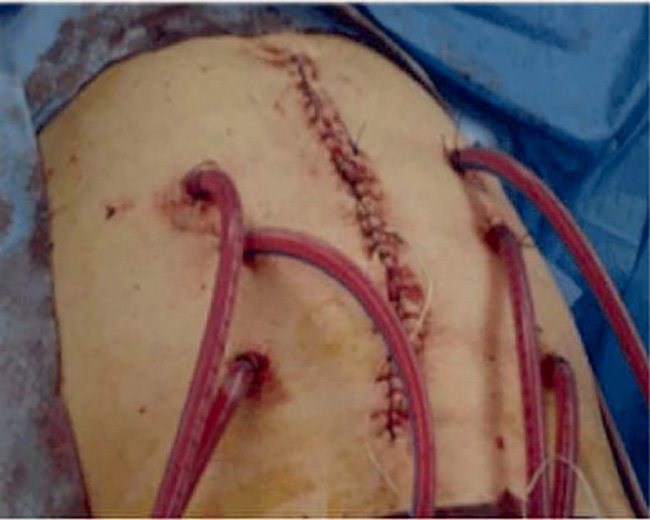
Figure 2. Closed technique. (From Halkia et al5; used with permission.)
element of the procedure varies depending on which chemotherapeutic regime is being used, but can vary from 30 minutes up to 2 hours.
PHYSIOLOGICAL CHANGES DURING HIPEC
Many multisystem physiological changes occur during the HIPEC component of the procedure which the anaesthetist must be aware of.7
- Cardiovascular: Increase in heart rate and central venous pressure. No significant changes in blood pressure.
- Respiratory: Increase in peak airway pressures. Decrease in PaO2/FiO2 ratio. Increase in end-tidal CO2 levels.
- Renal: Decreased perfusion to kidneys. Metabolic acidosis with increased lactate.
- Coagulation: Hyperthermia-associated coagulopathy can see a decrease in platelet count as well as an increase in Prothrombin Time (PT) and International Normalised Ratio (INR).
INTRAOPERATIVE MANAGEMENT
Fluid Management
Fluid management is important during both the CRS and the hyperthermic chemotherapy infiltration. There can be significant blood loss associated with the major debulking surgery as well as the evaporative losses related to the open abdomen. The HIPEC causes peritoneal inflammation that can cause perioperative third-space losses.3,8,9 Cardiac output monitoring, pulse pressure variation monitoring, urine output monitoring, and point-of-care testing can all be useful in monitoring fluid balance and haemodynamics. Fluid replacement is with crystalloid or colloid solutions as well as blood and plasma. The choices between crystalloid and colloid and restrictive or liberal regimes remain the subject of debate. Overall the anaesthetist should be aware of large fluid shift and should provide a fluid regime that ensures maintenance of systemic and regional perfusion.8,9
Haemodynamic Management
The maintenance of end-organ perfusion is of utmost importance. In response to the rising body temperature associated with the chemotherapeutic agent, the peripheral vasculature dilates. This results in an increase in heart rate in order to maintain cardiac output. During the closed technique a rise in airway pressures and central venous pressure occurs as a result of increased intra-abdominal pressure. The filling of the abdomen with saline and the chemotherapy has a similar effect to that of a pneumoperitoneum.3,9
Invasive cardiac output monitoring such as transoesophageal echo or a Swan-Ganz catheter is not recommended; however, more standard monitoring such as an arterial line, a central venous catheter, and a urinary catheter are useful for monitoring the haemodynamic status of the patient in real time.3 It should be remembered, however, that central venous pressure (CVP) will not give an accurate representation of the patient’s preload status due to the raised intra-abdominal pressure. The use of stroke volume variation may help guide fluid status intraoperatively.
Coagulation Management
An important factor to consider when anaesthetising someone for CRS and HIPEC is that there can be a significant coagulopathy associated with the procedure. Approximately one-third of patients will develop some sort of coagulopathy requiring transfusion of plasma products.3 There may be an association between disease severity as measured by the peritoneal carcinomatosis index and the risk of developing a coagulopathy.10
A coagulopathy can develop at either the CRS stage or the HIPEC stage. During the CRS stage, there may be major blood loss requiring transfusion, which can increase the risk of a coagulopathy due to intraoperative transfusion of red blood cells. During the HIPEC stage, coagulation may be impaired due to the hyperthermia, the protein loss, the tumour entity, or the chemotherapeutic toxicity.3,9
Coagulopathy can be monitored with laboratory tests including INR, APTT, and PT as well as with point-of-care testing using thromboelastography (TEG) or rotational thromboelastometry (ROTEM). There is some evidence for the use of tranexamic acid in an effort to reduce the requirement for transfusion of red cells during CRS.11 Bearing in mind that outcomes in surgical oncology where blood transfusions occur can be associated with increased morbidity, a restrictive transfusion policy may be adopted.
Temperature Management
CRS combined with HIPEC can be associated with both hypothermia and hyperthermia. During the cytoreductive period of the procedure, there is risk of hypothermia due to the extensive debulking procedure and large area of surgical exposure. Hypothermia increases the risk of blood loss and surgical wound infections, and alters the pharmacokinetics of commonly used anaesthetic drugs. Efforts must be made to prevent this hypothermia by using warmed fluids, forced-air warmers, and warming blankets and by monitoring the patient temperature and the room temperature, ensuring the patient remains normothermic.3 Prior to commencement of the intraperitoneal chemotherapy, controlled hypothermia can commence by changing to cool intravenous fluids and decreasing room temperature in anticipation of the chemotherapy, which can lead to a hyperthermia.8 The carrier solution for HIPEC is heated to 42°C-43°C. Body temperature may rise to up to 40.5°C. This hyperthermia increases the systemic oxygen demand, and may lead to an increased metabolic demand with rising end tidal CO2 levels and a concomitant metabolic acidosis. Hyperthermia also puts the patient at risk of coagulopathies, renal and liver dysfunction, neuropathies, and seizures. During the HIPEC phase, cooling blankets and cold head-wraps or cooling blocks can be used to help to regulate the temperature.
THE ROLE OF REGIONAL ANAESTHESIA
Intra- and postoperative pain management can be achieved via epidural anaesthesia. A well-functioning epidural reduces the need for both intra- and postoperative opioids. HIPEC is a painful procedure with pain scores similar to, if not greater than, to those associated with thoracotomy. As many of these patients will already be established on complex analgesia regimes, an epidural, ideally a patient-controlled epidural analgesia is a useful adjunct.12 Along with a decreased requirement for postoperative ventilation, the opioid-sparing effects of an epidural result in decreased incidence of bowel dysfunction and atony.
CHEMOTHERAPEUTIC CONSIDERATIONS
Depositing the chemotherapy in the peritoneal cavity allows higher doses to be used than would be tolerated systemically. By heating the agent it increases cell permeability and metabolic activity, thus increasing its tumouricidal effects.
The anaesthetist should be aware of the side effects associated with the use of chemotherapeutic agents. In addition to the more common allergic reactions, such as flushing, nausea, and vomiting, consideration should also be given to the direct cardiotoxic effects of the chemotherapy agents. In particular, cisplatin has been associated with prolongation of the QT interval and there have been case reports describing pulseless ventricular tachycardia. It is also important to consider the nephrotoxicity of cisplatin and the need to prevent renal injury. Other agents that are in use, such as mitomycin C and doxorubicin, are associated with myelotoxicity and neurotoxicity.3,9
SAFETY IN THE THEATRE
The increased use of chemotherapeutic agents in the operating theatre has the potential to pose a risk to theatre staff. Personnel exposure can arise from different routes, such as air contamination and pulmonary absorption, contact contamination, manipulation of perfusates, or manipulation of objects or tissues exposed to chemotherapeutic agents. It is also worth noting that bodily fluids are considered to be chemotherapy contaminated for 48 hours. With the open technique, the cavity is covered by a plastic sheet to try to minimise exposure and there is a smoke evacuator under the sheets to minimize vapour contamination. The closed technique, in theory, provides the highest level of protection for operators.
Education and training of personnel who are preparing and using chemotherapy in the operating theatre is important in helping to minimize risks. Routine medical surveillance of personnel who work with chemotherapeutic agents in theatre may be undertaken with checking of routine bloods and monitoring for any adverse side effects.13
Ambient air and biological monitoring has demonstrated that the use of intraperitoneal chemotherapy is safe for both patients and staff provided that the standard safety and chemotherapeutic handling protocols are adhered to. These protocols require specific preparation of the operating theatre and selection of appropriate personnel, use of personal protective equipment (PPE), careful handling of chemotherapy by trained personnel and avoidance of any spills.
Appropriate PPE has to be used by all participating personnel at all times. This consists of face masks, gloves, protective gown, and overshoes. Simple surgical masks do not provide adequate protection; filtering masks should be used and changed every 2 hours. Double-gloving and wearing gloves no longer than 30 minutes are advised. Double-gloving confers a high protection, using an air layer as an effective barrier. Goggles should also be worn in order to protect eyes from splashes.11
Adequate ventilation and the use of a smoke evacuator are essential for the control of air contamination. Air conditioning should be continuous throughout surgical operations. The air conditioner filters should be of high-efficiency particulate air (HEPA) type, while doors should be closed with air-tight closures. Notices should be placed outside the doors to alert people to the use of chemotherapeutic substances.
POSTOPERATIVE CARE
Postoperatively, patients should be cared for in the critical care setting, depending on the facilities, either an intensive care unit or a surgical high-dependency unit. This is to monitor for postoperative physiological changes and allow for management of complications of the surgery or physiological derangement and correction of any coagulopathy.14 Careful fluid balance assessment and monitoring is essential as vasodilation may occur after HIPEC. In this instance, vasopressors may need to be instituted and avoidance of large-volume fluid resuscitation where patients are not fluid responsive.
Early enteral feeding is recommended for these patients, as nutrition is crucial to promote wound healing and improve intestinal transit.9
COMPLICATIONS
As with all major abdominal surgery, there is a risk of postoperative complications such as bowel perforation, anastomotic leakage, bile leakage, fistula formation, postoperative bleeding or infections, and venous-thromboembolic complications. A postoperative ileus is also common after abdominal surgery with HIPEC.
There may also be additional complications associated with the chemotherapy such as a transient leukopenia and deranged transaminases, which may have an impact on surgical complications.
SUMMARY
In patients undergoing HIPEC, the anesthetist is challenged by fluid shifts, temperature changes, increased intraabdominal pressure, and an increased metabolic rate. It is of utmost importance to be vigilant with fluid management and haemodynamic monitoring perioperatively. The use of regional anaesthesia in the form of an epidural is a useful adjunct to pain management, bearing in mind the risk of developing a coagulopathy, which can be monitored by pointof- care testing. Knowledge of both chemotherapeutic safety and handling as well as the adverse effects of the particular agents is also important for the anaesthetist. Following the procedure, it is recommended that the patient is monitored in an acute care setting.
REFERENCES
- Neuwirth MG, Alexander HR, Karakousis GC. Then and now: cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (HIPEC), a historical perspective. J Gastrointest Oncol. 2016;7(1):18-28.
- Elias D, Goe´ re´ D, Dumont F, et al. Role of hyperthermic intraoperative peritoneal chemotherapy in the management of peritoneal metastases. Eur J Cancer. 2013;50(2):332-340.
- Raspe´ C, Piso P, Wiesenack C, et al. Anesthetic management in patients undergoing hyperthermic chemotherapy. Curr Opin Anaesthesiol. 2012;25(3):348-355.
- Arjona-Sa´ nchez A, Medina-Ferna´ ndez FJ, Mun˜ oz-Casares FC, et al. Peritoneal metastases of colorectal origin treated by cytoreduction and HIPEC: an overview. World J Gastrointest Oncol. 2014;6(10):407-412.
- Halkia E, Tsochrinis A, Vassiliadou DT, et al. Peritoneal carcinomatosis: intraoperative parameters in open (coliseum) versus closed abdomen HIPEC. Int J Surg Oncol. 2015;Article ID 610597, 6 p. DOI: 10.1155/2015/610597
- Rodriugez Silva CR, Moreno Ruiz FJ, Estevez IB, et al. Are there intra-operative hemodynamic differences between the Coliseum and closed HIPEC techniques in the treatment of peritoneal metastasis? A retrospective cohort study. World J Surg Oncol. 2017;15 (1):51.
- Schmidt C, Creutzenberg M, Piso P, Hobbhahn J, Bucher M. Peri-operative anaesthetic management of cytoreductive surgery with hyperthermic intraperitoneal chemotherapy. Anaesthesia. 2008;63(4):389-395.
- Webb CA, Weyker PD, Moitra VK, et al. An overview of cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion for the anesthesiologist. Anesth Analg. 2013;116(4)924–931.
- Raspe´ C, Flother L, Schneider R, et al. Best practice for perioperative management of patients with cytoreductive surgery and HIPEC. Eur J Surg Oncol. 2017;43(6):1013-1027.
- Hurdle H, Bishop G, Walker A, et al. Coagulation after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy: a retrospective cohort analysis. Can J Anaesth. 2017;64(11):1144-1152.
- Sargant N, Roy A, Simpson S, et al. A protocol for management of blood loss in surgical treatment of peritoneal malignancy by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Transfusion Med. 2016; 26(2):118- 122.
- Osseis M, Weyrech J, Gayat E, et al. Epidural analgesia combined with a comprehensive physiotherapy program after cytoreductive surgery and HIPEC is associated with enhanced post-operative recovery and reduces intensive care unit stay: a retrospective study of 124 patients. Eur J Surg Oncol. 2016;42(12):1938-1943.
- Kyriazanos I, Kalles V, Stehpanopolous A, et al. Operating personnel safety during the administration of hyperthermic intraperitoneal chemotherapy (HIPEC). Surg Oncol. 2016;25(3):308-314.
- Mogel HD, Levine EA, Fino NS, et al. Routine Admission to intensive care unit after cytoreductive surgery and heated intraperitoneal chemotherapy: not always a requirement. Ann Surg Oncol. 2016;23(5):1486-1495.



