General Topics
Key Points
- Understanding the complex pathophysiology, comorbidities, and multiorgan complications of end-stage liver disease patients, is essential for success.
- There are unique anaesthetic challenges across the different phases of liver transplantation.
- Vigilance and anticipation are essential to prevent critical events during the operation.
- Coagulopathies are complex, and thromboelastography is a valuable tool.
- Multidisciplinary pre-operative planning, preparation, and optimisation is essential for success.
INTRODUCTION
Orthotopic liver transplantation is the treatment of choice for eligible patients with end-stage liver disease. In the United States, alcoholic cirrhosis is the most common aetiology for end-stage liver disease, while viral hepatitis (Hepatitis C, followed by B) is the most common indication for transplantation. Other indications for transplant include: hepatic tumours, primary biliary cirrhosis, primary sclerosing cholangitis, some metabolic and vascular conditions, and acute liver failure (which is most commonly caused by acetaminophen/paracetamol toxicity). Predominant aetiologies for cirrhosis will obviously vary with geographic location.
With a finite and limited number of organs available for transplantation, cadaveric donor grafts are allocated to patients based on their risk of death within 3 months (Figure 1). This risk has been quantified using The Model for End-Stage Liver Disease (MELD) score, which ranges from 6 – 40, and is calculated from the serum bilirubin, INR, and serum creatinine. The MELD score has been further modified to take into consideration a patient’s level of hyponatremia, which has been shown to provide a more prognostic value than the traditional MELD score alone. Exception points are given to patients with hepatocellular carcinoma and portopulmonary hypertension. For a successful outcome in patients with these conditions, they require transplantation before their liver function deteriorates. The modification in the criteria for this subgroup of patients increases their chances of receiving a transplant in a timely fashion despite having lower standard MELD score.
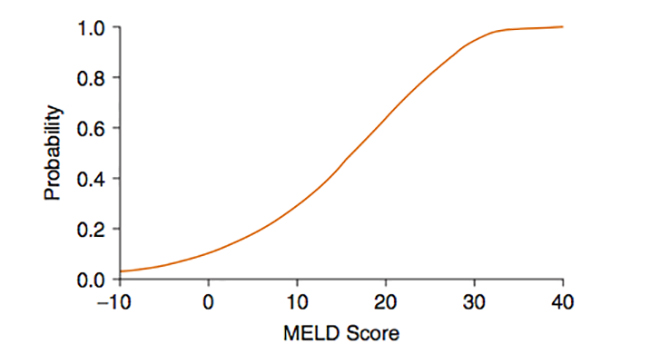
Figure 1. Relation between MELD Score and 3-month mortality. (Wiesner RH, McDiarmid SV, Kamath PS et al. MELD and PELD: Application of survival models to liver allocation. Liver Transplant. 2001)
PREOPERATIVE CONSIDERATIONS
Patients with end-stage liver disease have complex multi-organ pathophysiology which has many implications for the anaesthetist. The primary goal of pre-transplant evaluation is to establish the patient’s ability to withstand the stress of surgery, which requires a multidisciplinary team approach (anaesthesia, surgeons, intensivists, hepatologists, etc.). The extent and importance of multidisciplinary planning, coordination, and cooperation (both pre, intra, and post-operatively) is one of the factors which makes managing liver transplantation so unique and complex. As such, a system-based approach is essential when evaluating these patients preoperatively.
Neurologic
A significant number of patients with cirrhosis may present with hepatic encephalopathy. Although the exact mechanism is unknown, it is believed to be related to elevated levels of ammonia shunted into the systemic circulation via the dysfunctional and decompensated liver. Infection, gastrointestinal bleeding, and/or hyponatremia can further exacerbate the degree of encephalopathy. Monitoring and evaluating a patient’s fluctuation in consciousness and cognition can be graded using the West Haven Criteria (Figure 2). In severe case of acute liver failure, patients may have significant cerebral oedema, which needs to be included as part of their perioperative management.
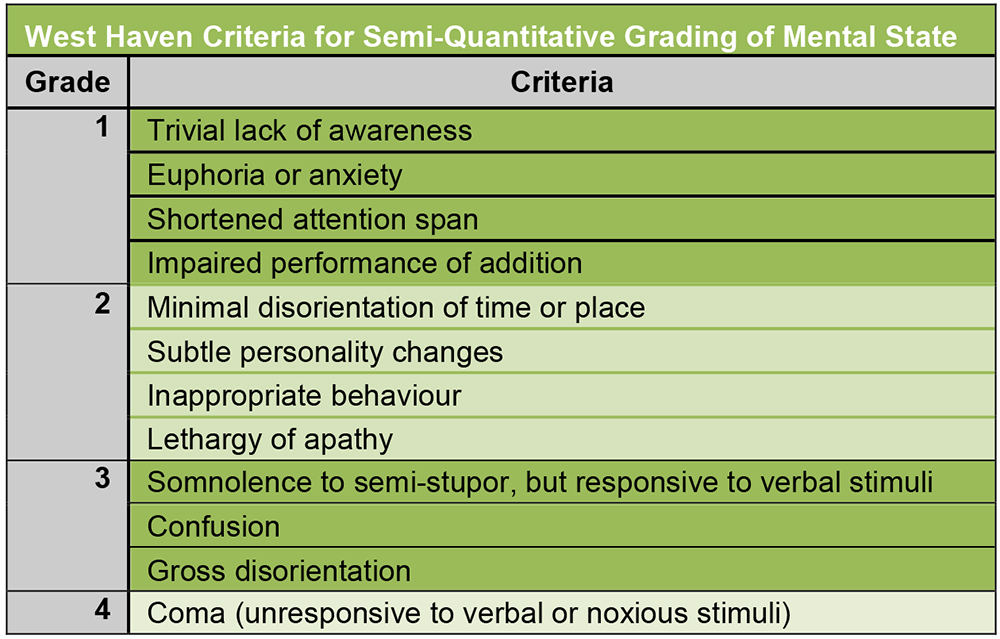
Figure 2. West Haven Criteria for Grading of Hepatic Encephalopathy. (Hepatic Encephalopathy-Definition, nomenclature, diagnosis, and quantification: Final report of the Working Party at the 11th World Congresses of Gastroenterology, Vienna, 1998)
Hematologic
Cirrhotic patients should be expected to have deranged coagulation. Increased bleeding comes from a quantitative deficiency of coagulation factors as well as a thrombocytopenia and dysfibrinogenemia. Concomitant elevations in Von Willebrand’s factor, Factor VIII, and decreases in Protein C & S also contribute to a hypercoagulable state. Because of the complex derangements in coagulation, thromboelastography (TEG) or thromboleastometry (ROTEM) are invaluable point of care tools in managing the haemostatic resuscitation and stabilization of liver transplant patients. In centres utilizing these technologies, a working knowledge of their use and interpretation is essential.
The thrombocytopenia common in patients with end-stage liver disease is thought to be secondary to hypersplenism and the consumption of platelets via intravascular coagulation. The platelets that remain in circulation are often dysfunctional due to defective signal transduction and intrinsic defects in thrombin and adenosine diphosphate (ADP).
While seemingly counterintuitive, pre-operative platelet transfusion to correct thrombocytopenia is not recommended, as transfused platelets will also become sequestered in the recipient’s spleen, thus yielding limited efficacy and increasing the risk of thrombotic events. Transfusion after the transplanted liver is in place will be discussed later.
It is important to remember that the combined derangements of coagulopathies and thrombocytopenia contribute to an increased risk of both bleeding and thrombosis.
Cardiac
The cardiovascular changes in cirrhosis resemble those seen in sepsis: tachycardia, increased cardiac output, low mean arterial pressures, and low systemic vascular resistance. These physiologic changes result from an enhanced production (or diminished hepatic clearance) of vasodilatory substances such as nitric oxide and endogenous cannabinoids. The portal hypertension seen in cirrhosis eventually leads to the development of portosystemic shunts and a hyperdynamic circulation, which creates an increased venous capacitance. As cardiac complications form a large percentage of the perioperative morbidity in liver transplantation, most centres routinely conduct cardiac stress testing if the patient has >2 cardiac risk factors.
Cirrhotic patients may also develop portopulmonary hypertension, characterized by a mean pulmonary artery pressure of greater than 25mmHg, and a pulmonary vascular resistance > 240dyn*s/cm5 at rest. Preoperative echocardiography is essential in assessing myocardial dysfunction and screening for pulmonary hypertension. In patients with confirmed (or a high suspicion of) pulmonary hypertension, any abnormalities should be evaluated with a nuclear stress test or a right heart catheterization. A right-heart catheterization is the primary means of distinguishing pulmonary venous hypertension (which is common and of no prognostic significance) from portopulmonary hypertension.
Due to the significant morbidity associated with liver transplantation and patients with portopulmonary hypertension, anyone with confirmed pulmonary hypertension should be further evaluated and every attempt at optimisation considered, then reevaluated for suitability of orthotopic liver transplantation (OLT) (Figure 3).
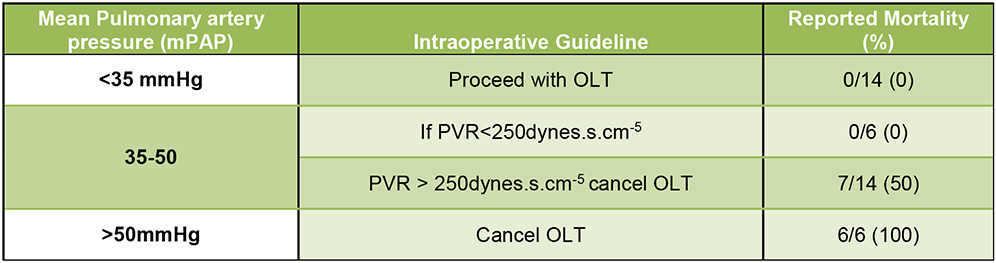
Figure 3. Current Mayo Clinic Intraoperative Guidelines Concerning Pulmonary Hemodynamics in Patients with Portopulmonary Hypertension (Runo, J. “Liver Transplantation for Portopulmonary Hypertension” Clinical Liver Disease, September 2014)
Pulmonary
In cirrhotic patients, the accumulation of ascites may be significant enough to cause increased diaphragmatic pressure and restrictive lung physiology. Additionally, patients with excessive ascites may also develop hepatic hydrothorax (more common on the right side).
A diagnosis of hepatopulmonary syndrome should be suspected in patients with hypoxemia (PaO2 < 60mmHg) who have no evidence of cardiac disease. Patients with hepatopulmonary syndrome tend to have significant intrapulmonary vascular dilation with delayed shunting (diagnosed on echocardiography), resulting in a wide alveolar-arterial oxygen gradient (>15-20mmHg) and platypnea (dyspnoea in the upright position, relieved by lying down). The only definitive therapy for hepatopulmonary syndrome is liver transplantation. However, the presence of hepatopulmonary syndrome reduces pulmonary reserve and further increases the perioperative morbidity.
Vascular
Most cirrhotic patients will have at least some component of portal hypertension, defined as a hepatic venous portal gradient of > 5mmHg. This is caused by an increase in intrahepatic vascular resistance and portal venous blood flow, which increases the pressure gradient between the portal vein and the hepatic vein. Collateralization via varices will subsequently form to relieve this pressure. However, as portal hypertension worsens, the liver produces excessive endogenous vasodilators, causing splanchnic vasodilation, increased portal flow, and further exacerbation of the portal hypertension.
Renal
Patients with advanced cirrhosis often develop significant hypovolemic hyponatremia. The hyponatremia is secondary to increased anti-diuretic hormone (ADH) secretion, resulting in worsening ascites and oedema. Alternatively, patients may also develop hypovolemic hyponatremia secondary to overdiuresis. As cirrhosis progresses, a reduction in systemic vascular resistance may cause a compensatory activation of the renin angiotensin system, leading to renal hypoperfusion. This renal hypoperfusion can lead to hepatorenal syndrome, defined in Figure 4.

Figure 4. Diagnostic Components of Hepatorenal Syndrome
As opposed to patients with prerenal azotaemia, patients with hepatorenal syndrome do not respond to a 1.5 L fluid challenge. The only definitive therapy for hepatorenal syndrome is liver transplantation.
INTRAOPERATIVE CONSIDERATIONS
The intraoperative management of patients undergoing liver transplantation is one of the most complex found in the field of anaesthesiology. The grossly deranged physiology, combined with the rapid hemodynamic disturbances throughout the surgery, necessitate a carefully planned and collaborative intraoperative approach.
Equipment & Monitoring
- Monitoring involves standard ASA monitors; five lead ECG, pulse oximetry, non-invasive blood pressure, and temperature monitoring.
- Invasive arterial blood pressure: Many institutions commonly utilize arterial lines placed at 2 sites (two radial, or a radial and femoral), due to the need for frequent blood draws and constant hemodynamic monitoring.
- Central venous catheter: This access is essential for monitoring right atrial pressures, as well as for the administration of vasopressors and inotropes.
- Pulmonary artery catheter: Their use varies by local practice, but many centres find them invaluable in patients with porto-pulmonary hypertension.
- Venous access: Large and rapid volume losses necessitate ensuring adequate venous access. The preferred method varies between individual centres (introducer sheaths, large bore peripheral access, rapid infusion catheters, etc.). Special attention should be made to ensure there is more than one point of access, and to avoid using access below the diaphragm (femoral/lower extremity access), as pathways via the Inferior Vena Cava (IVC) may be clamped or be where a source of bleeding is.
- Transoesophageal echocardiography: This is being used with more frequency to help assess myocardial function, adequacy of inferior vena cava reconstructions, and volume status.
- Warming devices: Multiple modalities for temperature management are essential, with several warming devices normally utilized to prevent hypothermia.
- Thromboelastography (TEG): Because of the widely deranged coagulation status with broad fluctuations throughout the operation, TEG is an invaluable tool for guiding effective blood product and factor replacement.
- Rapid transfusion: Due to the extensive blood loss in these surgeries, rapid transfusion devices are invaluable and should be utilized as a standard of care.
- Cell salvage: The large volume of blood lost creates an ideal environment for the use of intraoperative cell salvage. Use of cell salvage may also help address some of the hyperkalaemia associated with blood transfusion. Even in cases of hepatocellular carcinoma, cell salvage and transfusion has a growing consensus for its utility.
- Processed EEG: This modality might have a role during liver transplantation but given generalised slowing of brain activity (EEG) in encephalopathic patients, more evidence is required prior to advocating routine use.

Induction
Patients with ascites, recent food intake, or suspicion for gastroparesis must be considered for a rapid sequence induction. Due to the restrictive lung physiology commonly found in cirrhotics, expect significant reductions in functional reserve capacity (FRC), as well as time to desaturation. Pharmacologically, patients with end-stage liver disease have larger volumes of distribution, but decreased serum protein levels, decreased hepatic extraction, and decreased renal excretion. Combined, these physiologic changes make the pharmacokinetics of most anaesthetic drugs unpredictable.
Stages of Surgery
Liver transplantation is divided into 3 intraoperative stages: the dissection phase (where the surgeon makes the incision, drains ascites and prepares to remove the dysfunctional liver), the anhepatic phase (after the vessels to the liver are clamped and liver is explanted), and the reperfusion stage (with the completion of portal vein anastomosis and the perfusion of the transplanted liver). Each of these phases is described in detail, below.
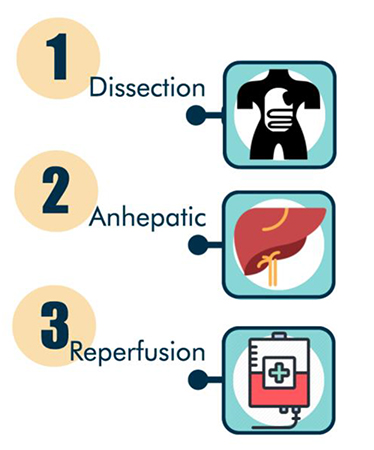
Dissection Stage
This stage of the surgery starts with incision and ends with clamping of the hepatic vessels (the hepatic artery, portal veins, and the suprahepatic and infrahepatic inferior vena cavas). In patients with a large amount of ascites, removal of the ascites can result in significant hypotension, necessitating aggressive fluid resuscitation. Volume expansion can be achieved with crystalloids, albumin, and blood products. Crystalloids that require hepatic metabolism, such as lactated ringers, should be avoided. Potassium-containing solutions are also not a good option in hyperkalaemic patients.
During the dissection stage, the anaesthetic focus is on maintaining hemodynamic stability, addressing coagulopathy, and managing electrolytes in preparation for the anhepatic stage. Prior to removing the native liver, the patient must be hemodynamically optimized to tolerate the major vascular clamping required for the total hepatectomy (this varies with the technique used to re-anastomose the inferior vena cava).
The standard technique of clamping the inferior vena cava (IVC) results in an extreme loss of preload, with subsequent hypotension. Optimization for this clamping technique requires involves volume loading, administration of vasoactive medications, and correction of hypocalcaemia. If the patient cannot tolerate IVC clamping, either a piggyback technique (Figure 5a and 5b) or veno-venous bypass (Figure 6) can be employed, both of which allow better preservation of preload during the hepatectomy.
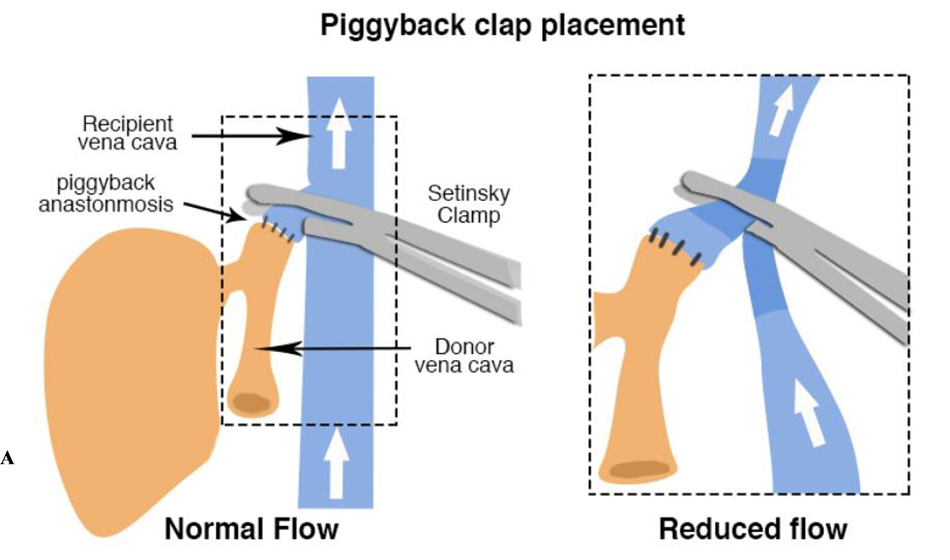
Figure 5a. Location and effect of “piggy-back” clamp placement in liver transplantation surgery
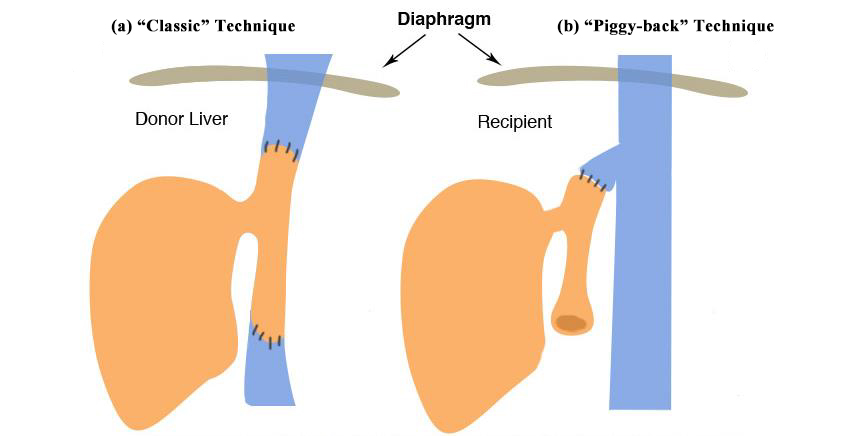
Figure 5b. Common anastamotic variations in liver transplant. (a): Two-caval anastomosis in the conventional technique. (b): Piggy-back technique with preservation of the retrohepatic cava and anastomosis to the hepatic vein stump.)
Anhepatic Stage
Surgically, the anhepatic stage involves removal of the native liver and re-anastomosis of the transplanted liver graft, but prior to vascular unclamping and reperfusion.
At this point in the operation, patients (especially with a piggy-back or conventional anastomosis technique) are essentially hypovolemic, as venous return via the IVC is partially or completely non existent. Some volume resuscitation may be necessary, but management will commonly involve maintaining haemodynamics with vasopressors and inotropes (such as norepinephrine). Addressing hypotension via excessive fluid resuscitation during this stage will generally result in gross hypervolemia once the new liver is perfused and venous return is re-established. This hypervolemia is not only detrimental to cardiopulmonary function, but can also result in congestion of the transplant liver.
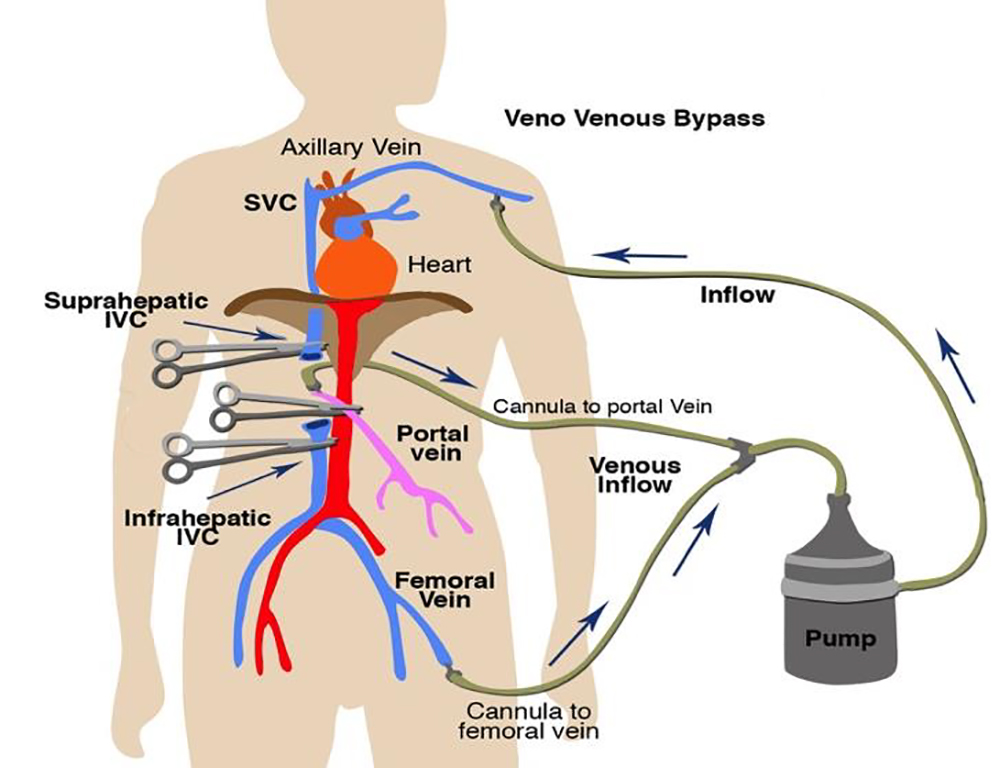
Figure 6. Standard Veno-venous bypass circuit for liver transplantation surgery
Once all the vessels are clamped, all systemic hepatic function ceases. While functionally anhepatic, the patient may rapidly develop worsening acidosis and hypocalcaemia (due to the lack of lactate and citrate metabolism). Acidaemia and anuria leads to increasing hyperkalaemia, which must be aggressively treated in preparation of hepatic reperfusion.
Treating hyperkalaemia and acidosis can start early in the case, with diuretics, continuous renal replacement therapy, and minimizing blood transfusions, as well as potentially washing blood products in cell saver devices to reduce the potassium load. As reperfusion of the transplanted liver draws closer, transient hyperkalaemia can be further countered with insulin, glucose, and bicarbonate infusions. Acute acidosis can be further stabilised with hyperventilation.
Existing coagulopathy will progressively worsen in proportion to the length of the anhepatic period, especially in the presence of the physiologic derangements of acidosis, hypothermia, and electrolyte imbalances.
Vascular anastomoses of the transplanted liver are the last steps of the anhepatic stage. The vessels are usually connected in the following temporal sequence: suprahepatic inferior vena cava, infrahepatic inferior vena cava, portal vein, hepatic artery, and then bile duct.
Reperfusion Stage
Prior to graft reperfusion, the IVC is unclamped, restoring preload to the heart. Reperfusion of the transplanted liver begins when the portal vein is unclamped. On unclamping, desaturated blood from the obstructed portal circulation, inflammatory mediators (such as Il-6 and TNF alpha, potassium, protons, and intrahepatic cold fluids/components), and a varying amount of embolic material are rapidly flushed into the patient’s circulation. These ischemic components commonly result in hemodynamic instability, with a sudden decrease in blood pressure, heart rate, systemic vascular resistance, and cardiac output. These acute physiologic changes can quickly result in a worsening of pulmonary hypertension and right heart failure. A sustained 30% decrease in mean arterial blood pressure for more than 1 minute during the first 5 minutes after reperfusion is defined as Post-Reperfusion Syndrome (PRS). The risk of PRS increases proportionally with prolonged cold ischemic time. Special mention needs to be made for regarding hyperkalaemia. Rapid increases in potassium are common at this stage, and can easily lead to cardiac arrest when severe. Rapid treatment for hyperkalaemia should be available, and close monitoring for ECG changes maintained during this stage of the transplantation.
To pre-emptively minimise the risk of post-reperfusion syndrome, consider the following:
- Pre-emptive administration of calcium to correct hypocalcaemia
- Sodium bicarbonate or tris-hydroxymethyl aminomethane (THAM) infusion to adjust pH
- Inotropes or vasopressors for circulatory support
Vigilance and close communication with the surgeons must be maintained during this stage. Attention should be devoted to rapidly diagnosing and treating dysrhythmias (which can easily progress to cardiac arrest) and hypotension.
Neohepatic Phase
After reperfusion, the remainder of the vascular anastomoses must be completed, including the hepatic artery and the bile duct. Immunosuppression, usually in the form of a steroid bolus, is important to verify. While varying between institutions, this is usually administered at or just before reperfusion of the liver.
During the neohepatic phase, the function of the new liver can be variable, so the anaesthetist must carefully monitor and appropriately treat any acidosis, coagulopathy, and temperature disturbances while hepatic function returns. The final step of implanting the transplanted liver is anastomosing the bile duct, after which, the abdomen is closed and the patient is taken to the Intensive Care Unit (ICU). Extubation usually occurs early in the ICU admission, but this practice varies between centres, and is predominantly dependent on the patient’s stability.
POSTOPERATIVE CONSIDERATIONS
Establishing that the transplanted liver is functional is important for guiding the patient’s recovery and ensuring physiologic acceptance of the transplant. Early signs of graft function include a rise in body temperature due to increased metabolism, hyperglycaemia from enhanced gluconeogenesis, normalization of coagulopathy, bile production, and lactate clearance. Ultrasound scans are often conducted intraoperatively and immediately postoperatively to confirm patent hepatic and portal vasculatures.
The clinician must be vigilant to monitor for vascular complications (both venous and arterial) as they can easily compromise the graft survival. If function of the new liver is inadequate or absent, the patient will likely require continuing transfusion of blood products and coagulation factors, correction of acidosis via renal replacement therapy, as well as vasopressor support.
Understanding the complex physiologic changes and anatomic considerations of liver transplantation are a valuable tool for all anaesthetic practitioners, regardless of their actual involvement in transplantation surgery. The complexities and nuances that occur within the short and intense period of hepatic transplantation serve as an excellent template for studying and understanding hepatic physiology and derangement, which can occur in a variety of settings.
SUMMARY
- Liver transplantation patients with end-stage liver diseases have extensive physiologic and metabolic derangements that need to be quantified and optimized preoperatively.
- Preoperative cardiac assessment is essential for risk stratification and physiological optimization.
- Anaesthetic management of these patients should focus on the potential for rapidly fluctuating haemodynamics, metabolic state, coagulation status, and physiology during the perioperative period.
- Close communication with all operative staff is key to anticipating and stabilising physiologic changes throughout the operation.
REFERENCES AND FURTHER READING
- Moitra V. Chapter 16: Liver Transplantation. Yao and Artusio’s Anesthesiology: Problem-Oriented Management Eighth Edition; 369-388
- Wagener G. Liver Anesthesiology and Critical Care Medicine. New York, NY: Springer New York; 2012.



