Paediatric Anaesthesia
KEY POINTS
- The ductus arteriosus is a communication between the pulmonary artery and aorta in the foetus, and usually closes spontaneously after birth.
- Prematurity is a major risk factor for patent ductus In these premature infants, patent ductus arteriosus is associated with respiratory distress syndrome, necrotizing enterocolitis, and cerebral intraventricular haemorrhage.
- Clinical features are related to left-to-right shunting of blood and echocardiogram is essential for
- Treatment may be medical (pharmacological closure), transcatheter device closure, or surgical ligation (via thoracotomy or thoracoscopy), depending on ductal anatomy and patient’s weight and
- Anaesthetic management should consider the patient population, from frail preterm infants to older children; surgical approach; and location of the procedure, as well as ventilatory strategy and ensuring adequate analgesia.
INTRODUCTION
The ductus arteriosus (DA) is a foetal artery, connecting the pulmonary artery to the aorta. Failure of its closure after birth results in a patent ductus arteriosus (PDA) (Figure 1). This results in a left-to-right shunt, and depending on the magnitude of shunting, can lead to cardiovascular and respiratory complications.
Improvements in medical care and enhanced survival of extremely premature infants have led to a significant increase in the incidence of PDA. Consequently, it has become a more common indication for thoracic surgery in this fragile population.
PATHOPHYSIOLOGY
The DA is a key component of the foetal circulation, establishing a communication between the pulmonary artery and the descending thoracic aorta. It most commonly arises just distal to the origin of the left subclavian artery.
In the foetus, the high-resistance pulmonary circulation leads to shunting of blood from the pulmonary artery into the aorta through the DA. The hypoxic foetal environment and prostaglandins produced by the placenta maintain its patency.
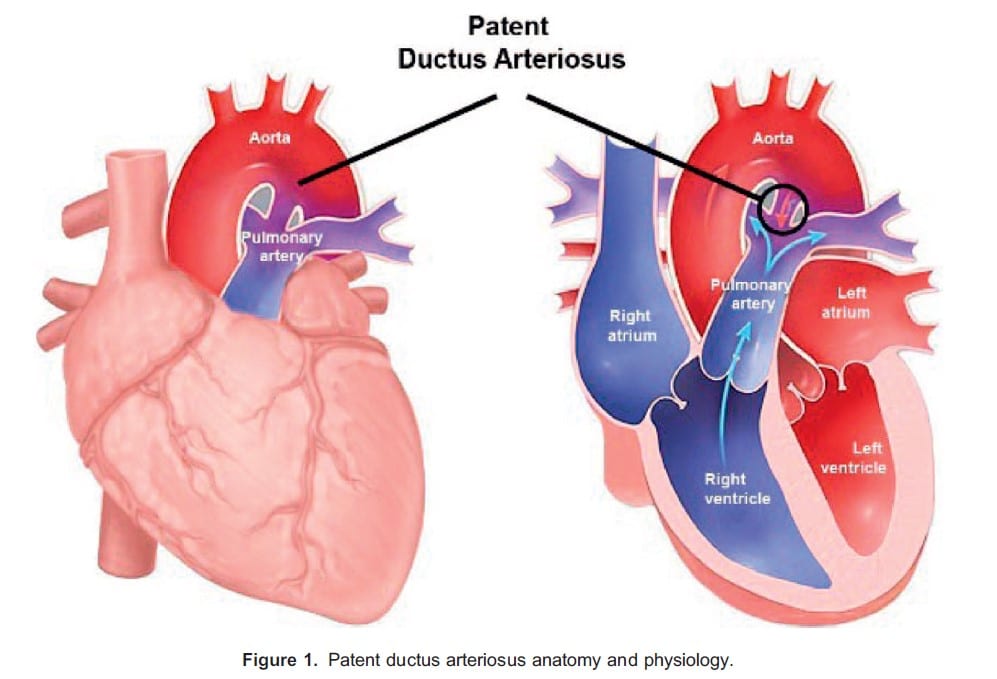
After birth, as the lungs expand, there is a decrease in pulmonary vascular resistance, together with an increase in systemic vascular resistance, due to umbilical cord clamping. The decrease in prostaglandin E concentration by the loss of the placental source and increase in oxygen content of arterial blood will induce DA smooth muscle contraction, leading to functional closure of the DA within 18 to 24 hours after birth, followed by anatomical closure, usually within 2 to 3 weeks of birth.1
Pathophysiology of PDA differs widely in preterm infants and term infants or children.
Preterm Infants
In preterm patients, the immature DA remains open due to immature smooth muscle with a high threshold for response to oxygen and increased sensitivity to the vasodilating effects of prostaglandin E2 and nitric oxide. In addition, underdeveloped lungs are unable to metabolise prostaglandin E2. Other factors, summarized in Table 1, may contribute to interrupting or delaying the process of ductal closure, or even reopen it. The incidence of PDA in premature infants is around 20% to 60%, reaching 70% to 80% in infants with a birth weight ,<1000 g.1

In this group of patients, the decrease in pulmonary vascular resistance after birth results in a left-to-right shunt through the PDA, leading to pulmonary vascular congestion and pulmonary oedema, as well as low systemic cardiac output, which may compromise myocardial, renal, splanchnic, and cerebral perfusion. PDA is associated with respiratory distress syndrome, necrotizing enterocolitis, and cerebral intraventricular hemorrhage.2
Term Infants and Children
In term infants and children, PDA is a result of a significant structural abnormality. It occurs with increased frequency in several genetic syndromes, such as Char Syndrome. Though most cases of PDA are seemingly sporadic, many are believed to result from the association of genetic predisposition (autosomal recessive inheritance with incomplete penetrance) with environmental triggers (for example, congenital rubella).1
The incidence of PDA is about 1 in 2000 births, accounting for 5% to 10% of all congenital heart disease, with a female-male ratio of around 2:1. The clinical effect of PDA depends on the magnitude of left-to-right shunting, largely determined by the size of the ductus. Increased pulmonary blood flow and resulting increase in blood returning to the left heart results in left ventricular overload and left heart failure may ensue. Over time, the excessive pulmonary blood flow resulting from left-to-right shunting may lead to pulmonary hypertension and to the development of Eisenmenger Syndrome.
Ductal-Dependent Cardiac Lesions
There is a group of patients that benefit from a PDA: those with congenital heart disease which limits blood flow to the pulmonary or systemic circulation (examples: hypoplastic left heart, critical pulmonary stenosis, transposition of the great arteries). In these infants and children, a PDA will allow shunting of oxygenated blood and is often crucial for patient survival until palliative surgery or definitive surgical correction is performed.
DIAGNOSIS
Signs and symptoms depend on the magnitude of the left-to-right shunt and clinical presentations range from asymptomatic patients to those in congestive heart failure due to chronic volume overload. Clinical signs include tachypnoea (often noted only during feeding), failure to thrive, and “bounding” pulses due to increased pulse pressure. A continuous murmur may be audible over the left sternal border, radiating to the back, and this is frequently the diagnostic feature in otherwise asymptomatic individuals. Chest x-ray may show increased pulmonary vascular markings.3
Echocardiography is the ‘gold standard’ to confirm the diagnosis.3 It provides information on the size and morphology of the PDA and its magnitude, and it rules out any associated lesions, but indications for treatment also depend on the clinical course.
TREATMENT
Medical or interventional treatment is indicated in symptomatic PDA in premature patients.3 In older children, PDA closure is recommended to prevent potential later complications, such as heart failure, endocarditis, or pulmonary hypertension. There are different methods for closure, and the appropriate method will depend on patient age and weight, DA size, available equipment, and centre expertise.
Medical Treatment
Conservative management may include fluid restriction and/or diuretics, as well as shunt-limiting strategies by avoiding further decrease in pulmonary vascular resistance. These include increasing positive end-expiratory pressure or allowing permissive hypercapnia in mechanically ventilated patients.
The mainstay of pharmacological PDA closure is prostaglandin inhibition by cyclooxygenase inhibitors, most commonly with ibuprofen or indomethacin. Contraindications to their use include renal dysfunction, necrotizing enterocolitis, coagulopathy, and active bleeding. Ibuprofen may be preferred to indomethacin, due to a lesser incidence of renal side effects.
The first dose is 10 mg/kg for ibuprofen and 0.2 mg/kg for indomethacin. If PDA does not close or significantly reduce in size, further doses may be administered at 24-hour intervals, up to a maximum of 3 doses. Second and third ibuprofen doses are 5 mg/kg, while for indomethacin it will depend on the age of the infant at the time of treatment, varying from 0.1 to 0.25 mg/kg. If PDA remains symptomatic after the second dose or additional dosage is contraindicated, transcatheter device closure or surgical treatment may be indicated.
Transcatheter Device Closure
Transcatheter PDA device closure procedures in the cardiac catheterization laboratory are increasingly popular since they are much less invasive than surgery. Low weight can be a limiting factor in the choice or success of this method but advances in medical bioengineering and recent availability of novel devices, which can be delivered though smaller sheaths, are gradually overcoming this limitation. Advantages include faster recovery, less postoperative pain and other potential longer-term consequences of surgery, as well as the possibility of being performed as outpatient procedures. Adverse events are mostly related to device placement, such as embolization and malposition.
For centres performing transcatheter PDA device closures in premature neonates, current contraindications are weight less than 700 g, age less than 3 days, size or shape of PDA not amenable, coarctation, pulmonary artery stenosis, intracardiac thrombus and active infection.
Surgical PDA Closure
Surgical ligation of a PDA is routinely performed through a left thoracotomy, via the fourth intercostal space, using a small incision, usually without the need for cardiopulmonary bypass. Video-assisted thoracoscopic clipping of PDA is an alternative, including in preterm infants.
ANAESTHETIC MANAGEMENT
Whilst in children a PDA is often an isolated lesion and its ligation or occlusion are usually straightforward procedures, in preterm infants anaesthetic concerns related to prematurity are paramount. The potential complications related to patient transport may favour the performance of the surgical procedure in the neonatal intensive care unit (NICU), obviating the need for transfer to an operating theatre. Ideally, all the requirements for non–operating room locations should be met by adhering to specific local protocols. Each hospital will determine what is best for their local environment. Great Ormond Street Hospital’s protocol for NICU PDA ligation is presented in Table 2 for reference.

Preoperative Assessment and Optimization
Anaemia should be corrected as it may predispose to heart failure (target haematocrit >30% and/or haemoglobin >10 g/dL). Although blood loss is typically minimal, there is the potential for catastrophic bleeding. As such, cross-matched blood should be prepared and readily available. Measures to prevent hypothermia are essential, such as adequate room temperature (>26°C) and appropriate patient- and fluid-warming devices. Due to high risk of endocarditis, antibiotic prophylaxis should be administered.
Most premature patients will already be intubated. If this is the case, ensure optimal size, position, patency, and fixation of the endotracheal tube. A large air leak may prevent adequate ventilation during lung retraction, therefore exchanging for a larger tube should be strongly considered in case the leak is more than 30% the tidal volume or is audible during inspiration at a 15 cm H2O pressure. Ventilator settings should also be reviewed: confirming the patient is not on unconventional ventilation modes (such as high-frequency oscillatory ventilation), fraction of inspired oxygen concentration and ventilation pressures (as peak inspiratory pressure will likely increase during lung retraction).
Monitoring
In addition to standard monitoring, including continuous end-tidal carbon dioxide measurement, it is strongly advised to monitor preductal and postductal oxygen saturations, using 2 pulse oximeters: 1 on the right upper limb and 1 on a lower limb. Blood pressure should be monitored in the right arm, accounting for the rare instances in which the left subclavian artery may need to be clamped. Invasive blood pressure monitoring is helpful if already established but it is not mandatory.
Anaesthetic Technique
Establishing at least 2 large-bore intravenous access lines for fluids or blood product administration is essential.
Premature patients are vulnerable to metabolic and respiratory derangements as a result of the stress triggered by major surgery and adequate anaesthesia and analgesia play a critical role in perioperative outcomes. High-dose opioids and muscle relaxation have long been the mainstays of the anaesthetic technique for surgical PDA ligation in this population. Analgesia in an important component of patient stability, both intra- and postoperatively, and evidence suggests the use of a minimum total dose of at least 10.5 μg/kg body weight of fentanyl or an equipotent alternative opioid dose, in order to promote respiratory stability and blunting of the stress response.4
Despite concerns about anaesthesia medications and their risk for neurotoxicity on the developing brain, the use of anaesthetic agents such as ketamine and halogenated agents is not consensual; they are still commonly used for this procedure to supplement high-dose opioid-based anaesthetic regimens in many specialist centres. More recently, there is an interest in the use of dexmedetomidine for congenital heart surgery, as it has been shown to have effect in some of the outcomes, possibly by contributing to the blunting of the surgical stress response.5
The ventilatory strategy should aim for the lowest fraction of inspired oxygen tolerated, in order to prevent pulmonary vasodilation and reduce left-to-right shunting, as well as maintain normocarbia to slight hypercarbia, since hyperventilation and hypocarbia may lead to cerebral ischemia.6
DA size and morphology are very variable, and it frequently is as large as or larger than the aorta and pulmonary artery. As such, to ensure the correct vessel will be ligated, a test clamp is usually performed. A decrease in saturation or distal perfusion should immediately be communicated to the surgeon during test clamping. After ductus ligation, an increase in diastolic blood pressure is expected.
Complications
- Inadvertent ligation of aorta or pulmonary artery can
- Rarely, massive and sudden bleeding might occur if a major blood vessel is injured (the DA is thin and friable in premature infants).
- Dissection near the vagus nerve may cause
- Thoracic duct damage may lead to chylothorax and require
- Recurrent laryngeal nerve injury may result in vocal cord
Postoperative Care
Adequate pain control is particularly important in very premature infants, since reducing pain minimises endocrine and metabolic responses to surgery, thus improving clinical outcomes. Pain after thoracotomy is expected to be severe and may lead to ineffective chest expansion, predisposing to atelectasis and hypoxaemia. In fact, the use of a high-dose opioid-based regime reduces postoperative inotropic or vasopressor requirements in neonates after cardiac surgery,7 and mitigates the respiratory compromise associated with a thoracotomy incision.5 Regional analgesia, such as intercostal nerve blocks, is beneficial as an analgesia adjunct and useful as part of a multimodal analgesia regime.
Postligation cardiac syndrome occurs in almost 50% of preterm patients after surgical PDA closure, and it is defined as systolic arterial pressure less than the third percentile requiring vasopressor use, with associated ventilation and oxygenation failure. Younger patients (less than 28 days old) and those weighting less than 1000 g are at an increased risk of postligation cardiac syndrome. PDA ligation triggers an acute increase in afterload, due to raised systemic vascular resistance and reduced preload from decreased pulmonary blood flow, which result in myocardial dysfunction. Symptoms usually begin 8 to 12 hours following intervention. Treatment is based on decreasing afterload while maintaining preload and cardiac output with drugs or combinations of drugs such as milrinone and/or dobutamine. Hydrocortisone ought to be considered in the setting of severe and/or refractory hypotension, as there is an increased risk of adrenal insufficiency in this group of patients.8
SUMMARY
- Failure of DA closure after birth results in a PDA and a left-to-right
- Improved survival of extremely premature patients has increased the incidence of PDA in this population. PDA may also be diagnosed in term infants or children, in whom it is the result of a structural abnormality.
- Anaesthetic management of surgical closure of PDA will depend on the surgical technique (transcatheter device closure or surgical ligation through thoracotomy or thoracoscopy) and patient population (preterm infants versus older children).
- Aspects related to anaemia correction, availability of cross-matched blood, hypothermia prevention, and monitoring with 2 pulse oximeters should be considered, as well as a carefully planned ventilatory strategy.
- Ensuring appropriate pain control is crucial, both intra- and
REFERENCES
1. Gournay V. The ductus arteriosus: physiology, regulation, and functional and congenital anomalies. Arch Cardiovasc Dis. 2011;104(11):578-585.
2. Cote´ CJ, Lerman J, Anderson BJ. A Practice of Anesthesia for Infants and Children. 6th ed. Philadelphia, PA: Elsevier; 2018.
3. Rhee CJ, Justino H, Yacouby SM, et al. Patent ductus arteriosus. Accessed September 25, 2021. https://www.texaschildrens.org/sites/default/files/uploads/documents/heart/Patent%20Ductus%20Arteriosus.pdf
4. Janvier A, Martinez JL, Barrington K, et al. Anesthetic technique and postoperative outcome in preterm infants undergoing PDA closure. J Perinatol. 2010;30(10):677-682.
5. Yang L, Weishuai B, Ping L, et al. Dexmedetomidine improves the outcomes in paediatric cardiac surgery: a meta-analysis of randomized controlled trials. Interact CardioVasc Thorac Surg. 2018;26(5):852-858.
6. Wolf, A. Ductal ligation in the very low-birth weight infant: simple anesthesia or extreme art? Pediatr Anesth. 2012;22(6):558-563.
7. Anand KJ, Hickey PR. Halothane-morphine compared with high-dose sufentanil for anesthesia and postoperative analgesia in neonatal cardiac surgery. N Engl J Med. 1992; 326(1):1-9.
8. Giesinger RE, Bischoff AR, McNamara PJ. Anticipatory perioperative management for patent ductus arteriosus surgery: understanding postligation cardiac syndrome. Congenit Heart Dis. 2019;14(2):311-316.



