Cardiothoracic Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding aortic stenosis:
a. It is one of the most common causes of valvular heart disease in developed countries.
b. It is presented in up to 30% of adults over the age of 65.
c. Rheumatic heart disease is the main cause of aortic stenosis in developing countries.
d. It is severe at a valve area of less than 1 cm2.
e. Aortic stenosis combined with congestive cardiac failure (CCF) has a 2-year mortality of 50%. - Regarding medical management of aortic stenosis:
a. Nitrates are generally contraindicated
b. Angina is managed with beta-blockers or calcium channel antagonists.
c. Statins reduce ischaemic cardiovascular events in the elderly patient.
d. Digoxin or diuretics are no longer used in the management of associated CCF.
e. CCF associated with stenosis is managed with angiotensin converting enzyme inhibitors. - The following statements on the peri-operative care of TAVI procedure are true:
a. Invasive arterial monitoring should be commenced prior to induction of anaesthesia.
b. Atrial fibrillation is usually well tolerated.
c. Patients are dependent on preload.
d. Hypotension should be treated aggressively.
e. In the event of a cardiac arrest cardiopulmonary resuscitation is likely to be effective.
Key Points
- Without valve replacement, symptomatic aortic stenosis has a poor prognosis.
- Elderly patients are often deemed unsuitable for surgical valve replacement due to co-morbidities and high mortality risk.
- Minimally invasive catheter techniques have rapidly developed and indications broaden to offer treatment to high-risk patients.
- Anaesthetic considerations and techniques for TAVI are similar to those required for other minimally invasive procedures performed in hybrid operating theatres.
INTRODUCTION
Aortic stenosis, a progressive narrowing of the aortic valve, is one of the most common valve problems in developed countries. In developing countries the main cause is attributable to rheumatic heart disease1. It can be asymptomatic over a prolonged period and present in up to 25% of adults over the age of 651. Major haemodynamic compromise usually occurs at a valve area of less than 1-1.5cm2 2.
Severity is classified according to valve area and mean pressure gradient. Mild above > 1.5cm2 and less than < 25mmHg. Moderate 1-1.5cm2 and 25-40mmHg. Severe <1 cm2 and >40mmHg. Very severe <0.6cm2 and gradient above 70mmHg3.
Disease progression causes the left ventricle to become pressure overloaded leading to concentric hypertrophy of the ventricular wall. High interventricular pressures are required to maintain cardiac output, with diastolic and systolic dysfunction eventually occurring on a background of increased afterload and reduced contractility2. Exertional dyspnoea, syncope and angina are the classic triad but syncope and heart failure herald a poor prognosis3.
Medical management aims to target co-morbid coronary artery disease and hyperlipidaemia to control angina, hypertension and heart failure, but this has not been shown to improve survival. Statins aim to prevent ischaemic cardiovascular events. Angina and hypertension are treated with beta or calcium channel blockade and it is usual to manage heart failure with digoxin and diuretics. Angiotensin converting enzyme inhibitors can be used cautiously once outflow obstruction is excluded, but nitrates are generally avoided due to the potential for excessive or unpredictable hypotension abilities. Without surgical or trans-catheter intervention, the survival rate of severe symptomatic aortic stenosis is 50-60% at 1 year3.
Surgical aortic valve replacement (SAVR) has been considered gold standard treatment, improving quality of life and survival. In-hospital and 30-day mortality for SAVR is low at 3.2% with life expectancy post procedure returning to that near a control population2, 4.
The European Heart Survey on valvular heart disease in 2003 reported 31.8% of patients did not undergo intervention due to co-morbidities 5. Left ventricular dysfunction, severe aortic stenosis and older age are predominant characteristics of these groups despite the relatively good survival figures above in patients 79 years or older 5.
Trans-thoracic aortic valve implantation (TAVI) and balloon aortic valvuloplasty (BAV) are less invasive techniques for treatment of severe aortic stenosis with long and short-term benefits respectively.
The first BAV was performed in 1983. Unfortunately, device registries showed high re-stenosis rates. In 2002, at the University of Rouen in France, Dr Alain Cribier performed the first case of percutaneous aortic valve replacement on a 57year old patient with inoperable aortic stenosis 6.
Analogous to other percutaneous techniques like endovascular aortic aneurysm repair, TAVI is often preferred in patients deemed to be of high operative risk or where open surgery is contraindicated. A review in 2011 estimated the mortality in a higher risk population for SAVR as 16.4% 7. These patients scored 10 or greater using the Society of Thoracic Surgeons Predicted Risk of Mortality (STS-PROM) score. In comparable risk patients selected for TAVI, the overall in-hospital mortality has been reported as 7.4% 8.
The European PARTNER trial in 2011-12 showed TAVI was at least as good as SAVR for the high risk patient, possibly better and should be the standard of care for the inoperable patient. Significant mortality differences are quoted according to the approach used for TAVI (trans-femoral 5.9%, trans-apical 12.9% and trans-subclavian, 9.7%) 8.
Over the last few years, trans-thoracic catheter techniques have emerged as primary treatment options in aortic stenosis for the inoperable and high-risk patient. There have been over 200,000 aortic implants performed worldwide with good results in comparison to SAVR9. Procedural outcomes report success rates at 98%, with 30-day mortality less than 5%. Symptom improvement and reduction in hospitalisation also occur in the immediate term10,11.
Device technology continues to improve and initial challenges in technique overcome. TAVI is well established with many valves, but the Edwards SAPIEN valve (figure 1) and the Medtronic CORE valve (Figure 2) are strong in the market. The valves are now in their second generation, hoping to reduce associated complications. Applications for the procedure broaden, with small-scale studies indicating a future role in patients with pure aortic regurgitation or failing bio prosthesis unsuitable for redo cardiac surgery 6.
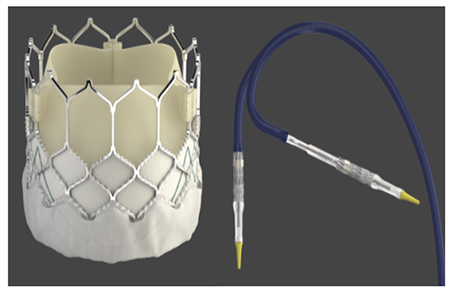
Figure 1. Edwards SAPIEN 3 Valve with command delivery system. Reproduced with permission from Edwards Life Sciences.
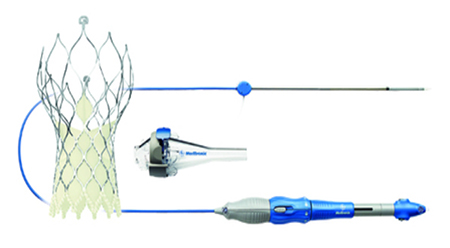
Figure 2. Medtronic Core Valve Evolut R with delivery system. Reproduced with permission from Medtronic.
Recently there is a trend toward offering TAVI to lower risk or younger patients. The Nordic Aortic Valve Intervention Trial (NOTION) compared all SAVR to TAVI in patients including those of low risk12. Results were numerically better with TAVI than SAVR even though the study was underpowered for a mortality benefit12. As with any new procedure, long- term results are unknown and valve-durability remains a question.
The following tutorial aims to focus on the anaesthetic concerns and management of patients for TAVI procedure.
INDICATION AND RISK ASSESSMENT
Patients are selected for TAVI based on clinical judgement and risk assessed by a multidisciplinary team of cardiologist, cardiothoracic surgeon and anaesthetist. The European Association for Cardiothoracic Surgery (EACTS) and European Society of Cardiology (ESC) position statements currently restrict TAVI to high-risk patients or those with contraindications to surgery13.
TAVI or BAV may be used as a bridging therapy to stabilize patients for future operative repair. TAVI may be used to facilitate coronary artery bypass grafts or as a treatment for a failing surgical bio-prosthesis. The European Society of Anaesthesiology (ESA) and ESC guidelines state TAVI may be considered in patients with severe aortic stenosis unsuitable for open repair that require urgent non-cardiac surgery13. This may be appropriate for those patients who require curative cancer surgery if a minimum of 1-year survival is predicted. Previous sternotomy is not a contraindication and in fact, TAVI may be preferable in those with patent bypass grafts. Patients with significant renal, respiratory or other medical conditions denied surgery might also benefit.
There are a number of contraindications to the procedure. Some of are relative and subject to change based on procedural advances, experience and improvements in valve technology.

Debate surrounds mitral regurgitation (MR) and chronic haemodialysis as absolute contraindications. Analysis of 316 patients undergoing TAVI in a single UK centre between 2008 and 2013 found significant MR was independently associated with an all-cause increased mortality post TAVI14. However, the same analysis noted significant improvement in the degree of a patient’s MR severity post TAVI with mortality rates appearing lower in comparison to similar patients treated for MR medically14. The analysis found patients with functional MR tended to do better. Adverse outcomesappear to be associated with organic MR, concomitant AF and pulmonary hypertension15. Current focus surrounds the assessment of MR severity and its cause pre-procedure in relation to other co-morbidities to help effective patient management planning.
A paper in 2012 found no reason not to offer TAVI to haemodialysis patients15. Overall hospital stay tended to be longer in haemodialysis patients. Aims are to preserve pre-existing renal function in those with grade 3 to 4 chronic kidney disease. Adequate renal replacement therapy with access to renal physician support during the peri-operative period is needed in patients who are dialysis dependent15.

Preoperative diagnostics on the severity of stenosis and adequacy of vascular access can be studied with trans-thoracic, trans-oesophageal echo and exercise testing16. Angiography with aortogram, CT and MRI are appropriate to assess anatomy to correctly size a prosthesis and disease severity16. All efforts should be made to identify and optimise intercurrent diseases along with congestive cardiac failure associated with the condition.
PROCEDURAL PLAN
TAVI is commonly performed by 4 routes: trans-femoral, trans-subclavian, trans-aortic, or trans-apical. The trans-carotid route has recently been described in the literature as an alternative access route where vessel access is difficult but is uncommonly performed17. Each route presents slightly different challenges to the team. There is no evidence one approach is superior to another and no recommendations on access site selection17. An individual patient approach is important with careful CT assessment of calcium distribution in access vessels, especially the ascending aorta17.
- Trans-femoral is least invasive and most common approach. Adequate iliofemoral vessel sizes above or equal to 7 mm with minimal calcification and tortuosity are required16.
- Trans-subclavian access to the aortic valve is less remote. Left is preferable to right. Valve delivery distance, including the catheter, is shorter – avoiding bending in the iliofemoral axis and thorac-oabdominal aorta. This improves control at deployment with more accurate positioning. This may help to reduce incidence of paravalvular leak and complete heart block18.
- Trans-aortic or direct aortic approaches are more invasive, using an upper J shaped mini sternotomy19. Indications are diseased iliofemoral access or unsuitability for trans-apical route due to poor left ventricular function.
- Trans-apical is most invasive, bypassing the arterial vessels. Risks such as atrial rupture, ventricular pseudoaneursym, deteriorating LV function, peri-operative bleeding and respiratory complications from thoracotomy increase significantly19. Tran-apical is a useful route if porcelain aorta is present which is contraindicated via the trans-aortic approach. Advantages include no access limitation, with shorter distance and antegrade implantation route facilitating exact positioning17.
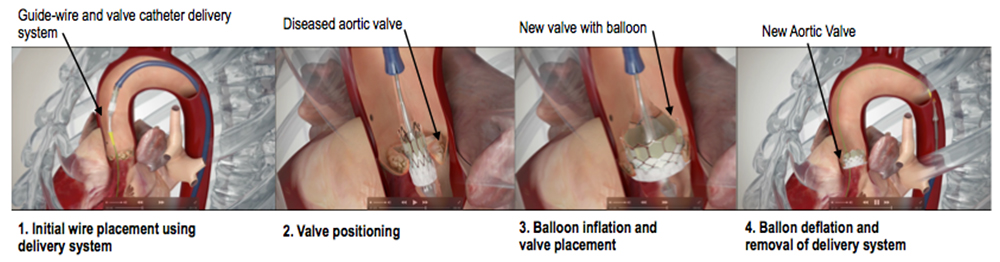
Figure 3. TAVI procedure illustration. Femoral approach using the Edwards SAPIEN 3 Valve Reproduced with permission from Edwards Life sciences
Outcomes post trans-apical route are often inferior to the trans-femoral approach with an increased risk of adverse events. This may reflect the severity of co-morbid disease necessitating this route and the more invasive nature of the procedure2.
It is vital to communicate a plan of management for unexpected events and any limitation in resuscitation must be well discussed and documented in advance. This can be reinforced at the time of the World Health Organization surgical safety checklist. TAVI takes place in a cardiac catheterisation lab or ideally a hybrid operating theatre16,19. The environment can be hostile with a sparse and crowded workspace requiring vigilance and organisation to maintain access. Patients may sustain serious complications, for example major haemorrhage, vascular or apical rupture and cardiac arrest. There should be provision for urgent femoral cardiopulmonary bypass, defibrillation, and pacing with surgical intervention as required 16,19.
Cardiothoracic surgeons and a perfusionist team must be available along with the following management considerations:
- Haemodynamic: This may involve use of vasopressors, inotropic agents, pacing and defibrillation4, 16,19.
- Anticoagulation: A heparin ACT of greater than 250 seconds is usually required. Patients continue aspirin and clopidogrel pre-procedure depending on clinical requirements4, 16,19.
- Monitoring: Invasive monitoring using trans-oesophageal echo and pulmonary artery flotation catheters as appropriate4, 16,19. Awake patients will have transthoracic echo.
- Cardiopulmonary bypass: Provision of femoral-femoral cardiopulmonary bypass especially in those patients whose ejection fraction is less than 20%19.
- Radiation exposure: High quality fluoroscopic equipment is used. All staff should be protected and measures taken to avoid contrast induced kidney injury4, 16,19.
PERI-OPERATIVE MANAGEMENT
Before induction, basic anaesthetic monitoring should be established with ECG and pulse oximetry. Urometer and temperature monitoring are routine for GA. Insertion of the following invasive lines prior to induction are performed as appropriate. Points 1 and 2 are advisable; points 3-5 can be decided on a case-by-case basis. Pulmonary artery catheters and ventricular assist devices may only be needed in those patients at high risk.
- Large bore peripheral access – with a rapid infuser as blood loss may become excessive.
- Arterial line to respond rapidly to changes in haemodynamics.
- Central venous line to administer vasopressors and inotropic support.
- Introducer sheath allowing for the insertion of a temporary pacing wire or pulmonary artery flotation catheter. The sheath is also useful for rapid infusion16, 19.
- Percutaneous ventricular assist device to facilitate the procedure if left ventricular dysfunction is significant16.
Initial haemodynamic goals should aim to optimise preload and maintain systolic pressure and an adequate diastolic time. Sinus rhythm is ideal, aiming for 80 beats per minute with adequate contractility. New onset atrial fibrillation is poorly tolerated so adequate rate control is required. Judicious use of fluids, vasopressors and inotropic agents may be needed to achieve this. Elevated filling pressures are required to fill the non-compliant left ventricle. The aim is to treat hypotension aggressively as cardiopulmonary resuscitation is unlikely to be effective through a stenosed aortic valve.
Anaesthetic technique is unlikely to be associated with significant differences in outcome. Local anaesthesia (LA) and sedation work well for trans-femoral approaches. Pain is minimal except when vascular structures are stretched. Patients requiring a trans-femoral approach can be sedated with fentanyl and small increments of midazolam as required with local anaesthetic wound infiltration. Low dose target controlled infusions of propofol or remifentanil are also suitable alternatives described in case reports14. Advantages can include rapid assessment of any neurological complications, avoidance of respiratory complications and rapid recovery with earlier hospital discharge20.
Regional techniques aim to reduce opioid requirement by avoiding excessive sedation and associated cognitive impairment. Continuous intercostal nerve block, thoracic paravertebral block, and thoracic epidural block have been reported for trans-apical approaches4. Local wound infiltration and lumbar epidural block are described for the trans-femoral approach but require caution as patients are heparinised to an ACT of 250 seconds. The need for emergency cardiopulmonary bypass in this situation should also be considered19.
Immobility is an advantage of general anaesthesia (GA) in patients who cannot lie supine for long periods and allows for better control of complications should they occur. Trans-oesophageal echo imaging under GA is superior to intermittent trans-thoracic echo and may facilitate delivery and deployment of the valve. This may help reduce malposition and incidence of paravalvular leak post op4, 21. The incidence of paravalvular leak depends on a number of factors including the type of valve used, irregularity of annular calcification and any under-sizing of the device. Improvements in second-generation valves may further reduce these complications6.
Induction and maintenance agent choice depend on the procedural approach and the anaesthetist. Anaesthetic requirements for TAVI are usually reduced due to the minimally invasive nature of the procedure, advanced age, renal impairment and reduction in cardiac output. Analgesic requirements are minimal for trans-femoral and moderate for trans-aortic or trans-apical routes with the large doses of opioid needed in SAVR not usually required4.
Maintenance is usually via inhalational agents or TIVA, both resulting in rapid recovery.
Recovery is usually done in a coronary care unit but intensive care unit placement may be required, especially in trans-apical and trans-aortic cases. The emphasis is on maintaining cardiac function, organ perfusion with the help of inotropes or vasopressors as indicated and early extubation. The post procedure recovery is expected to be shorter in an uncomplicated case with shorter intensive care stays and time to discharge from hospital compared to SAVR.
PROCEDURAL STAGES
There are 4 stages and each has its own set of problems for the team.
1. Pre-deployment
Access is gained via the femoral artery and right radial artery with a pigtail catheter. A right trans-venous pacing wire is inserted through the femoral vein. Serious problems at this point are usually related to the femoral arterial site for the sheathed valve. Haemorrhage, dissection and vascular rupture are all possible4, 16,19.
2. Balloon valvuloplasty
This may be required for certain valves but not others. It will depend on whether BAV was part of the patients work up in the preceding 4-6 weeks. The aortic valve should be dilated to ensure the position of the prosthesis in the root. Valvuloplasty allows for easier passage of the prosthesis. It is performed under rapid ventricular pacing at a rate of 180-220 beats per minute19. This must be kept to 5 to 10 seconds as it may induce subsequent arrhythmia and ischaemia. There is one valve (Lotus), which does not need this pacing after initial positioning but then takes 5 minutes to unfurl like an umbrella and this valve can be re-loaded if the initial position is less than satisfactory. The Edwards valve needs pacing and the whole event is finished in 6-8 seconds. At this point, the accepted practice is to administer a small bolus of vasopressor between sequences of rapid pacing to ensure a systolic above 75mmhg. Infusions of vasopressor or inotropes can also be used4, 16,19.
3. Positioning and deployment
Balloon deployment devices require rapid ventricular pacing to reduce cardiac output to ensure stable positioning and prevention of myocardial injury. Self-expanding devices do not require rapid pacing, as mentioned above. During positioning, large diameter catheters can obstruct blood flow in vessels perfusing the brain – urgent deployment should take place to avoid this4, 6,19.
Problems during this phase include16, 19:
- Device embolisation into the aorta or left ventricle
- Paravalvular regurgitation
- Incorrect placement
- Coronary ostial obstruction
- Arrhythmia
- Left ventricular asystole, pulseless electrical activity, especially with poor ventricular function
- Atrio-ventricular block
- Impingement of the mitral valve leaflet
Trans-oesophageal echo is especially useful at this time with trans-thoracic imaging reasonable for awake patients. It can provide important information on left ventricular function, the need for further dilation, to ensure correct position and to assess paravalvular leak.
4. Post-deployment
Device position, function and vessels are assessed with angiography. Vessels are repaired and the delivery system removed. As the afterload on the left ventricle has been reduced, the cardiac output is increased and discontinuation of vasopressors, the use of other anti-hypertensives and sometimes an infusion of GTN may be required to control the resultant hypertension16, 19.
If a temporary pacemaker is used, it is important to ensure stability and function. Patients with certain prostheses are more likely to need this and 3-7% need a permanent pacemaker2, 4. All access sites should be monitored for bleeding. Blood loss can be difficult to assess especially if retroperitoneal. Haemothorax and pericardial tamponade may be easier to detect in an intensive care environment and a low index of suspicion is required to recognise these complications.
Poor outcome post TAVI is related to existing patient co-morbidity as well as the procedure2, 4,6. Aortic regurgitation may result from paravalvular leak related to uneven calcification of the annulus. Vascular events may require immediate surgical correction with sternotomy and cardiopulmonary bypass but carry a very high mortality risk.
Complications tend to involve cardiac, neurological and renal systems. Non-revascularised coronary artery disease is common and so percutaneous revascularisation may often need to be achieved beforehand as part of a staged revascularisation. As described previously, coronary obstruction may occur due to a misplaced device or valve leaflet occlusion of coronaries arising low in the sinus of Valsalva.
Stroke is a known complication. The European SAPIEN Aortic Bio-prosthesis Multi-Region Outcome Registry showed a stroke rate of 2.4%22. The risk is falling – possibly due to smaller catheters being used, causing less trauma or obstruction of vessels leaving the aortic arch. Embolic filters placed in the brachiocephalic and left common carotid artery have led to reductions in cerebral lesions6.
Renal function from registry data suggests a rise in creatinine post TAVI in 5% to 28% of cases23. This usually improves due to an increase in cardiac output, but factors associated with deterioration include use of contrast, hypoperfusion and the need for blood transfusion2.
Clinical and echocardiographic follow-up of valves over 5 years is well documented and late leaflet failure is rare. Only time will tell if valve durability matches that of surgical prostheses24. This is especially important if TAVI is offered to younger patient groups in the future12, 25. There are indications that a reoperation on these patients 10 years later would not be straightforward. Surgery would initially be routine to access the aorta but then the degree of implant fibrosis into the root could result in more major surgery with aortic root replacement.
CONCLUSION
Trans-thoracic aortic valve implantation is now a well-established technique for the treatment of aortic stenosis in high-risk patients. Experiences in TAVI and valve technology are rapidly progressing6. The patient population in particular is challenging and will continue to benefit most from a multidisciplinary approach throughout their peri-operative course.
Knowledge surrounding indications for the procedure is valuable and relevant to anaesthetists involved in peri-operative medicine clinics. Patients with severe aortic stenosis may present for non-cardiac surgery and knowledge of referral pathways outside tertiary cardiac centres is useful.
Procedural concerns and anaesthetic management are applicable to a number of similar procedures performed in hybrid operating or radiology suites. There are similarities with neuroradiological procedures, thoracic and endovascular abdominal aneurysm repair. As the evidence and indication for these procedures grow, it is likely that anaesthetists will increasingly be required to facilitate safe peri-operative care in these environments.
ANSWERS TO QUESTIONS
- Regarding aortic stenosis:
a. True
b. False: It is present in 25% of adults over the age of 65 years.
c. True
d. True
e. True - Regarding medical management of aortic stenosis:
a. True: Nitrates will reduce systemic vascular resistance which will in turn reduce coronary perfusion..
b. True: Either agent can be used but not together.
c. True
d. False: Both agents help to improve symptoms of aortic stenosis. Digoxin improves contractility; diuretics reduce pulmonary oedema.
e. True: ACE-i can be used with caution. - The following statements on the peri-operative care of TAVI procedure are true:
a. True: Haemodynamic instability can be detected and managed promptly.
b. False: Rhythms other than sinus reduce cardiac output. Appropriately timed atrial contractions contribute up to 40% of left ventricular preload.
c. True: Elevated filling pressures are required to fill the non-compliant left ventricle.
d. True: Reduction in systemic vascular resistance reduces coronary perfusion
e. False: Cardiopulmonary resuscitation is unlikely to be effective through a stenosed valve.
REFERENCES AND FURTHER READING
- Chambers, J.B. (2009) ‘Aortic stenosis’, European Journal of Echocardiography, 10(1), pp. i11–i19 doi:10.1093/ejechocard/jen240.
- Chacko, Matthew, and Laurence Weinberg. “Aortic Valve Stenosis: Perioperative Anaesthetic Implications of Surgical Replacement and Minimally Invasive Interventions.” Continuing Education in Anaesthesia, Critical Care & Pain 12.6 (2012): 295–301.
- Wikipedia. [Place unknown]: Wikimedia Foundation; 2016 Jul 5. Aortic stenosis; Available from: https://en.wikipedia.org/wiki/Aortic_stenosis. (Accessed 10 July 2016)
- Klein, A. A., et al. “Transcatheter Aortic Valve Insertion: Anaesthetic Implications of Emerging New Technology.” British Journal of Anaesthesia (2009) 103.6: 792–799.
- Lung, B. “A Prospective Survey of Patients with Valvular Heart Disease in Europe: The Euro Heart Survey on Valvular Heart Disease.” European Heart Journal (2003) 24.13: 1231–1243.
- Bourantas, C. V., and P. W. Serruys. “Evolution of Transcatheter Aortic Valve Replacement.” Circulation Research (2014) 114.6: 1037–1051.
- Thourani VH, Ailawadi G, Szeto WY, Dewey TM, Guyton RA, Mack MJ, Kron IL, Kilgo P, Bavaria JE. Outcomes of surgical Aortic valve replacement in high-risk patients: A Multiinstitutional study. The Annals of Thoracic Surgery. (2011) Jan; 91(1): 49–56.
- Leon, Martin B., et al. “Transcatheter Aortic-Valve Implantation for Aortic Stenosis in Patients Who Cannot Undergo Surgery.” New England Journal of Medicine (2010) 363.17: 1597–1607.
- Grubb KJ, Fields T, Cheng A, Settles DM, Stoddard M, Flaherty MP. Transaxillary Transcatheter Aortic Valve Replacement with a Self-Expanding Valve under Conscious Sedation: Case Discussion and Review of the Literature. Clin Surg. 2016; 1:1019. http://www.clinicsinsurgery.com/pdfs_folder/cis-v1-id1019.pdf
- Smith, Craig R., et al. “Transcatheter Versus Surgical Aortic-Valve Replacement in High-Risk Patients.” New England Journal of Medicine (2011) 364.23: 2187–2198.
- TAVI: A Solution for All Patients: A Pro-Con Debate at ESC 2015. Medscape. Aug 30, 2015. http://www.medscape.com/viewarticle/850256 (Accessed 10 July 2016)
- Vahanian, A., et al. “Guidelines on the Management of Valvular Heart Disease (version 2012): The Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS).” European Heart Journal 33.19 (2012): 2451–2496.
- Khawaja, M.Z., Williams, R., Hung, J., Arri, S., Asrress, K.N., Bolter, K., Wilson, K., Young, C.P., Bapat, V., Hancock, J., Thomas, M. and Redwood, S. ‘Impact of preprocedural mitral regurgitation upon mortality after transcatheter aortic valve implantation (TAVI) for severe aortic stenosis’, Heart, (2014) 100(22), pp. 1799–1803.
- Rau, S., Wessely, M., Lange, P., Kupatt, C., Steinbeck, G., Fischereder, M. and Schönermarck, U. ‘Transcatheter Aortic valve implantation in dialysis patients’, Nephron Clinical Practice, (2012) 120(2), pp. c86–c90.
- Jobeir, Med Asaad. Anesthesia for Transcatheter Aortic Valve Implantation.
http://www.sha-education.com/sites/all/sha23_presentations/30-1-2012/066002.pdf. (Accessed 10 July 2016) - Bleiziffer, S., et al. “Which Way in? The Necessity of Multiple Approaches to Transcatheter Valve Therapy”. Current Cardiology Reviews 9.4 (2014): 268–273.
- Petronio A, Carlo M, Giannini C, Carlo F, Bortolotti U. Digital, E and publishing (2013) Subclavian TAVI: More than an alternative access route. Available at: https://www.pcronline.com/eurointervention/S_issue/volume-9/supplement-s/7/subclavian-tavi-more-than-an-alternative-access-route.html (Accessed: 31 December 2016)
- Zhu, Haibei, et al. “Direct Aortic Transcatheter Aortic Valve Implantation: Anaesthesia Attentions.” Proceedings of Singapore Healthcare 24.1 (2015): 59–64. http://psh.sagepub.com/content/24/1/59.full.pdf+html (Accessed 10 July 2016)
- Wiegerinck, E. M. A., et al. “Towards Minimally Invasiveness: Transcatheter Aortic Valve Implantation Under Local Analgesia Exclusively.” International Journal of Cardiology (2014) 176.3: 1050–1052.
- Developed by the American Society of Anesthesiologists Task Force on Perioperative Transesophageal Echocardiography, et al. “Practice Guidelines for Perioperative Transesophageal Echocardiography.” The Journal of the American Society of Anesthesiologists (2010) 112.5: 1084–1096.
- Schymik, Gerhard, et al. “European Experience with the Second-Generation Edwards SAPIEN XT Transcatheter Heart Valve in Patients with Severe Aortic Stenosis.” JACC: Cardiovascular Interventions (2015) 8.5: 657–669
- Bagur, R., et al. “Acute Kidney Injury Following Transcatheter Aortic Valve Implantation: Predictive Factors, Prognostic Value, and Comparison with Surgical Aortic Valve Replacement.” European Heart Journal (2009) 31.7: 865–874.
- Early Transcatheter Aortic-Valve Device Durability Comes Under Scrutiny. Medscape. May 31, 2016. http://www.medscape.com/viewarticle/864039 (Accessed 10 July 2016)
- Sapien 3 TAVR Superior to Surgery in Intermediate-Risk Aortic-Stenosis Patients, Says Study. Medscape. Apr 05, 2016. http://www.medscape.com/viewarticle/861469 (Accessed 10 July 2016)
Figure 1 and 3 reproduced with permission from Edwards Life sciences http://www.edwards.com/eu/Products/TranscatheterValves/Pages/sapien3.aspx?WT.ac=S3campaignprod.
Figure 2 reproduced with permission from Medtronic http://www.corevalve.com/evolut-r/index.htm?loc=corevalve_com_Intl_EvolutR_ContinueToEvoluteRSiteBtn



