Paediatric Anaesthesia
KEY POINTS
- Effective perioperative management of a patient with a craniopharyngioma entails a multidisciplinary team including an endocrinologist, an oncologist, a neurosurgeon, an anaesthesiologist, and an intensivist.
- Hypothalamic damage and endocrine derangements are more common in the paediatric population due to slow tumour growth. A patient’s perioperative course may be complicated by intracranial hypertension, growth failure, hypothyroidism, diabetes insipidus, obesity, and seizures.
- Treatment options include medical management of clinical symptoms, surgical treatment with subtotal resection and postoperative radiation therapy, or complete resection. Other less-invasive procedures include endoscopic cyst fenestration or placement of an Ommaya reservoir into the tumour cyst for delivery of antineoplastic agents.
- Key anaesthetic considerations include surgical approach, patient positioning, perioperative steroid supplementation, surgical damage to the optic chiasm and the circle of Willis, and management of diabetes insipidus and other endocrine disorders.
INTRODUCTION
Craniopharyngiomas are rare, histologically benign brain tumours derived from pituitary gland embryonic tissue that are seen most commonly in children between the ages of 5 and 10 years. They represent about 2% to 6% of all paediatric primary intracranial tumours.1 Effective perioperative management entails a multidisciplinary team including an endocrinologist, an oncologist, a neurosurgeon, an anaesthesiologist, and an intensivist. Preoperative evaluation should include determination of tumour size and proximity to critical neural and vascular structures, presence of compressive symptoms, effect on intracranial pressure (ICP), presence of any endocrine abnormalities, and any effects of radiotherapy already given. The anaesthesiologist plays an important role in directing the perioperative care of these patients, which is critical to help improve overall outcome.
CLINICAL PRESENTATION
Clinical presentation will depend on the site and size of the tumour. Craniopharyngiomas are slow growing and symptoms often are present for a year or more before the diagnosis is established.5 Hypothalamic damage and endocrine disturbances are more common in children and typically ensue before the appearance of visual symptoms.
Clinical presentation includes the following:
- Intracranial hypertension: nausea, vomiting, headache, papilledema, altered level of consciousness
- Visual pathway disturbances: bitemporal hemianopsia, homonymous anopsia, optic nerve atrophy in severe cases
- Endocrine disturbances: hypothyroidism, growth failure, hypogonadism, syndrome of inappropriate antidiuretic hormone secretion, diabetes insipidus (DI), precocious puberty
- Thalamus, hypothalamus, and/or frontal lobe disturbances: aphagia, obesity, poor energy, somnolence, emotional lability, hallucinations, autonomic disturbances
- Brain parenchyma disturbances: seizures, cognitive dysfunction.
TREATMENT
Historically, optimal treatment has been controversial and included two basic approaches: aggressive surgery with an attempt to achieve complete resection versus a more conservative partial surgical resection followed by radiation therapy to eradicate residual tumour.5 Medical management will be aimed at hormonal replacement therapy for endocrine abnormalities that are present.
Craniopharyngiomas are divided into two categories: adamantinomatous and papillary. Surgical resection in children is more challenging due to the predominance of the adamantinomatous histological subtype. This subtype of tumour is more cystic and calcified, larger at presentation, and more adherent to adjacent neural tissue as a result of fibrosis and inflammation.2
Surgical approach depends on both the location of the tumour and the age of the patient. A transsphenoidal endoscopic approach is typically used for intrasellar tumours in children older than 10 years. Tumours with suprasellar extension require an extended approach.
For the surgeon experienced in endoscopy, an endonasal approach, as compared to an open approach, has been shown to provide higher rates for gross total resection, with lower recurrence, lower rates of permanent DI, and less visual deterioration.7 Nevertheless, the diminutive size of the sphenoid sinus in infants and young children necessitates a frontal craniotomy to access the tumour.
One of the least invasive treatment options is the insertion of an Ommaya reservoir into the cystic aspect of the tumour followed by drainage with or without subsequent instillation of antineoplastic agents.7 Radiation therapy is used to treat patients who have undergone a partial surgical resection or to treat recurrence.
PREOPERATIVE EVALUATION
Preoperatively, diagnosis is marked by the presence of a mass on magnetic resonance imaging and/or computed tomography. The tumour can vary from a small, solid, well-circumscribed mass to a huge multi-locular cyst that invades the sella turcica and displaces neighbouring cerebral structures.5 A thorough history and physical examination looking for systemic manifestations provides important information regarding clinical presentation and subsequent treatment. In addition to performing a standard system-based preoperative anaesthetic evaluation, specific considerations in the paediatric patient with craniopharyngioma include the following:
- Endocrine evaluation to identify abnormalities including thyroid function tests, and determination of growth hormone, cortisol, sex hormones, adrenocorticotropic hormone, and prolactin levels. Hypoadrenalism, DI, and hypothyroidism have shown to have significant morbidity and mortality and should be corrected preoperatively.
- Evaluation of current medications including antiepileptic agents for seizure management and/or hormonal therapy.
- Ophthalmological assessment to provide information about visual acuity, field of vision, and possible papilledema.
- Assessment and correction of volume status and electrolyte disturbances. This will be particularly important in children with signs and symptoms of DI and increased intracranial pressure.
- A thorough airway examination, taking into consideration the differences between the adult and paediatric airways. Short stature and obesity in this patient population may pose a challenge in airway management. Obese children may have obstructive sleep apnoea and/or respiratory issues and thus should be identified preoperatively and considered at a higher risk for airway complications.
ANESTHETIC CONSIDERATIONS
Neurosurgery in the paediatric population is challenging to the anaesthesiologist due to the neurophysiological variations that exist between the adult and paediatric population. Cerebral blood flow and cerebral metabolic rate of oxygen are increased in infants and children as compared to adults and a tight coupling exists between the two. Although cerebral autoregulation exists in the paediatric brain, any extremes in blood pressure beyond the autoregulatory limits place the child at risk of developing ischemia or intracerebral hemorrhage.3
For nonemergent procedures, local fasting guidelines should be followed. Patients may present to surgery with hypovolemia due to DI, vomiting, fluid restriction, contrast agents, or osmotic diuresis. Adequate volume replacement is an important consideration prior to anaesthesia as well as when deciding upon an induction agent. Patients who are on hormonal therapy should receive their morning doses of medications as well as those on antiepileptic agents for seizure management.
Premedication will depend on the patient’s age, anxiety level, physical state, and comorbidities, including increased ICP or alterations in mental status. Premedication with opioids may pose a risk of hypoventilation with subsequent hypercarbia and hypoxemia in infants or children and should be avoided. Oral midazolam at a dose of 0.5 mg/kg may provide effective anxiolysis without significantly impacting oxygen saturation. (See ATOTW 367: ‘‘Paediatric anaesthesia: challenges with induction.’’)
MONITORING
Standard American Society of Anesthesiologists’ monitors including pulse oximetry, electrocardiography, blood pressure measurement, temperature monitoring, and end tidal CO2 measurement should be employed. Direct arterial blood pressure measurement should be obtained as it aids in anaesthetic management during surgical resection. Serial arterial blood gas sampling will allow for tight intraoperative control of the partial pressure of carbon dioxide (PaCO2), which may help improve surgical visualization, especially during endoscopic procedures. In addition, the proportionate increase in dead space, high flow rates, and small tidal volumes of infants and children may cause end tidal CO2 measurements to underestimate PaCO2. This may result in hypoventilation and alterations in ICP, making direct PaCO2 measurements crucial in helping to maintain cerebral perfusion pressure.4 Arterial line cannulation will also allow for direct blood pressure monitoring as well as the ability to closely monitor blood gases, serum electrolytes, and osmolality and haemoglobin levels. Placement of a urinary catheter is recommended as it will aid in monitoring urine output, which is crucial in the detection and management of DI.
VASCULAR ACCESS
Vascular access should include at least two large-bore peripheral intravenous lines for rapid resuscitation. There is a risk for significant bleeding due to the close proximity of the internal carotid artery and other vessels of the circle of Willis. Central venous cannulation is not necessarily required. Nevertheless, it should be considered depending on the size and extent of the tumour, presence of DI, possible prolonged use of inotropes, and if central venous pressure (CVP) measurement is deemed necessary.
INDUCTION AND MAINTENANCE OF ANAESTHESIA
Intravenous induction or inhalation induction with sevoflurane is considered appropriate for induction of anaesthesia. The intravenous induction drug of choice will depend on the child’s volume and haemodynamic status as well as on patient comorbidities. Irrespective of the agent used, the goal is to maintain cerebral perfusion pressure and avoid increases in ICP as well as major variations in mean arterial pressure. The sympathetic response to laryngoscopy should be blunted and preparations for a difficult airway undertaken if anticipated.
There are several options for maintenance of anaesthesia. The technique chosen should be tailored to ensure a rapid awakening at the completion of surgery to allow for a complete neurological examination. Agents such as propofol, remifentanil, sufentanil, and dexmedetomidine have all been used with techniques ranging from total intravenous anaesthesia to combination techniques with inhalation agents. (See ATOTW 392: ‘‘A practical approach to propofol-based TIVA in children.’’) Regardless of the agents chosen, their effects on cerebral nervous system physiology and haemodynamics should be considered and closely monitored. Intermittent or continuous infusion of a neuromuscular blocking agent should be employed to ensure a motionless surgical field. Mannitol and other ICP-lowering interventions may be needed for children presenting with increased ICP. Normal saline is the most commonly used fluid for neurosurgical procedures as its hyperosmolarity protects against cerebral oedema. Nevertheless, it is important to remember that large quantities may produce hyperchloremic metabolic acidosis and hypernatraemia. Topical nasal vasoconstrictors such a cocaine or phenylephrine are routinely applied to the nasal passages to minimize bleeding. However, these drugs can cause hypertension and tachycardia and these side effects should be closely monitored for and treated appropriately. In the rare setting of acute presentation of tumours obstructing the fourth ventricle and aqueduct of Sylvius, an emergent ventriculostomy may be required with all the anaesthetic implications of an emergent surgery.1
POSITIONING
Patient positioning is of paramount importance to the anaesthesiologist. Surgical approach will ultimately dictate the position of the patient. Supine position with head tilt is commonly employed for resection of a craniopharyngioma. The Mayfield frame is often used to fix the head with paediatric pins or the head may be supported in a horseshoe frame. Pin application in the paediatric patient carries the risk of intracranial haematoma, dural tear, and/or skull fracture, and should be done cautiously.1 During and after positioning it is of utmost importance that the endotracheal tube position be verified. It is not uncommon for endobronchial tube migration or cuff herniation at the glottic opening with head and neck manipulation to occur. Adequate venous drainage from the head and neck must be ensured as venous congestion may lead to increased ICP. Intravenous access lines, monitoring cables, and arterial line tubing should be free of kinking or risk of dislodgement. Padding of pressure points cannot be underestimated as pressure ulcers after prolonged hours of surgery may occur, especially in the paediatric patient.
INTRAOPERATIVE CONCERNS AND RISKS
The following table summarizes some of the intraoperative considerations and risks associated with craniopharyngioma resection.
DIABETES INSIPIDUS
Diabetes insipidus results form a deficiency of antidiuretic hormone, or arginine vasopressin. This hormone, produced in the hypothalamus, stored and released from the posterior pituitary, acts on the distal tubules and collecting ducts of the kidneys to promote reabsorption of water. A lack of antidiuretic hormone due to surgical resection therefore results in diuresis and electrolyte imbalance. Approximately 75% of patients develop DI following extended surgical resection of a pituitary tumour and DI occurs in 10% to 44% of patients after transsphenoidal pituitary surgery.6 Managing DI in children can be challenging, and often physicians from different specialties have different protocols, which may further complicate treatment. It is for this reason that a multidisciplinary discussion should be held during the preoperative evaluation to determine what management algorithm will be followed.
Signs suggestive of DI include the following:
- Urine output ≥ 4 mL/kg/h
- Serum Na ≥ 145 mEq/L
- Serum osmolality >300 mOsm/kg
- Urine osmolality < 300 mOsm/kg
- Polyuria ≥ 30 minutes
- Other causes of polyuria ruled out (eg, mannitol, saline, osmotic contrast agents, glucose, diuretics).
In the authors’ institution, the standard vasopressin concentration is 30 milliunits/mL. When there is intraoperative evidence of DI, a vasopressin infusion is started at 1 milliunit/kg/h and the rate is slowly (every 5 to 10 minutes) increased (maximum 10 milliunits /kg/h) to avoid hypertension. The goal is to decrease the urine output to less than 2 mL/kg/h.
For fluid management, the authors’ institution algorithm includes the following:
- Replace fluid deficits with normal saline or lactated Ringer solution as needed to support blood pressure until antidiuresis is established.
- After a reduction in the rate of diuresis, the intravenous fluids rate should be at two-thirds maintenance (plus fluids necessary for blood replacement and blood pressure support).
- Replace blood loss with normal saline, lactated Ringer solution, 5% albumin, or blood products as appropriate.
- Check serum sodium every hour.
Vasopressin infusion is continued postoperatively for management in the intensive care unit.
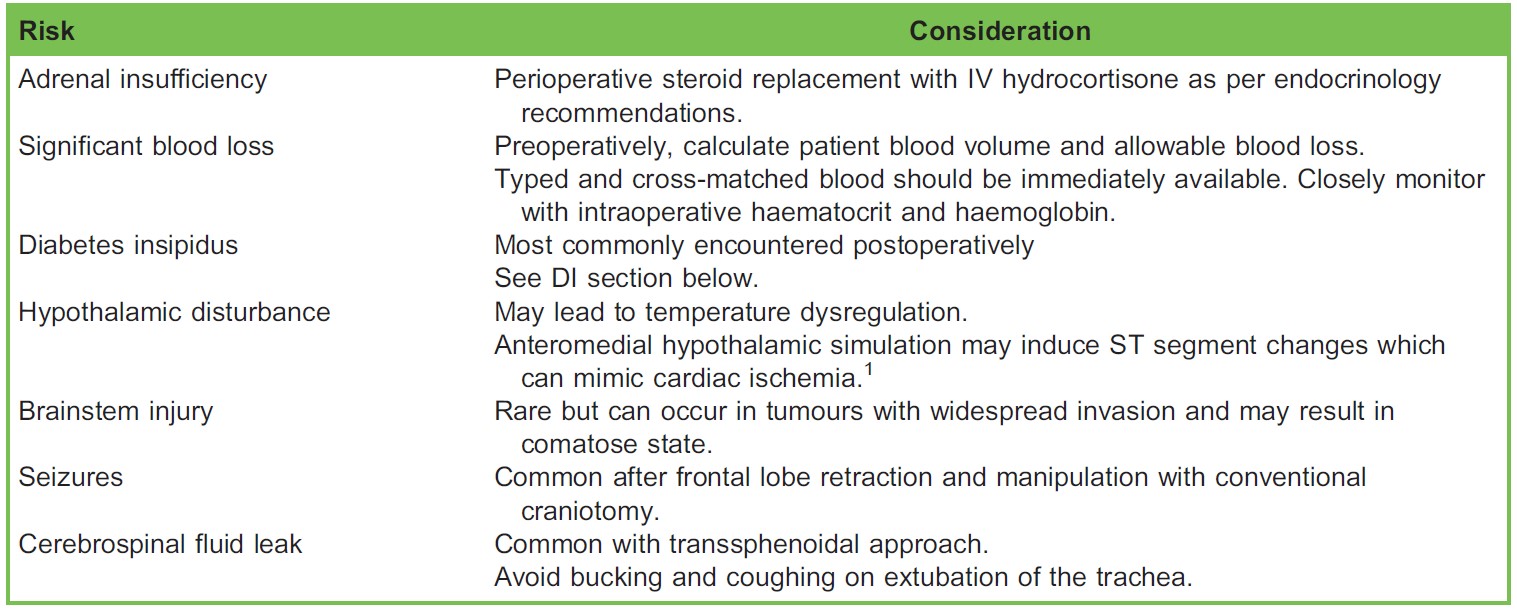
Table. Intraoperative Considerations and Risks Associated with Craniopharyngioma Resection
POSTOPERATIVE CARE
Patients undergoing an uneventful intraoperative course can be extubated at the conclusion of surgery. This allows for rapid neurological evaluation and avoids complications associated with postoperative mechanical ventilation. Postoperative care should continue in the intensive care unit with input from a multidisciplinary team including a neurosurgeon, an anaesthesiologist, an endocrinologist, and an intensivist. This team should be informed of any intraoperative concerns encountered both from a surgical and anaesthetic standpoint; management and potential complications should also be addressed. Plans for ongoing or potential endocrine derangements should be reviewed and most recent laboratory data shared. Fluid balance and potential seizure control should be discussed and managed accordingly. A plan for analgesia should be discussed by the multidisciplinary team, involving the pain management team if needed. Acute paediatric pain management is increasingly characterized by a multimodal approach leading to smaller doses of both opioid and nonopioid analgesics. Medications such as nonsteroidal anti-inflammatory drugs, ∝-2 adrenergic agonists, acetaminophen, and opioids can be used in combination to maximize pain control while decreasing untoward drug-induced side effects. Haemodynamic parameters and patient age and level of awareness as well as other comorbidities should be taken into consideration at the moment of drug election and dosing.
REFERENCES
- Moningi S. Anaesthetic management of children with craniopharyngioma. J Neuroanaesth Crit Care. 2017;4:30-37.
- Alli S, Isik S, Rutka J. Microsurgical removal of craniopharyngioma: endoscopic and transcranial techniques for complication avoidance. J Neurooncol. 2016;130:299-307.
- Krass IS. Lippincott Williams & Wilkins. Physiology and metabolism of brain and spinal cord. In: Handbook of Neuroanaesthesia. Philadelphia, PA: 2007.
- Johnson J, Jimenez D, Tobias J. Anaesthetic care during minimally invasive neurosurgical procedures in infants and children. Paediatr Anaesth. 2002;12:478-488.
- Harsh G, Recht L, Marcus K. Craniopharyngioma. https://www.uptodate.com/contents/craniopharyngioma?search=craniopharyngioma%20children&source=search_result&selectedTitle=1~47&usage_type=default&display_rank=1#H19. Accessed April 23, 2018.
- Wise L, Sulpicio S, Ferrari L, et al. Perioperative management of diabetes insipidus in children. J Nerusosurg Anesthesiol. 2004;16:220-225.
- Reddy G, Hansen D, Patel A, et al. Treatment options for pediatric craniopharyngioma. Surg Neurol Int. 2016;7(Suppl 6):S174-S178.



