Intensive Care Medicine
KEY POINTS
- Extracorporeal membrane oxygenation (ECMO) is an advanced life support technique that can temporarily replace the function of the lungs and/or heart while they recover from underlying reversible pathology.
- Patient selection is key to optimizing chances for good outcomes.
- There are 2 distinct configurations of the ECMO circuit: V-A ECMO and V-V ECMO.
- Indications for ECMO can be summarised in 2 categories: rescue therapy and bridging therapy.
- The only absolute contraindications to ECMO are comorbid conditions in which recovery, decannulation and organ transplantation are not feasible and patient refusal.
INTRODUCTION
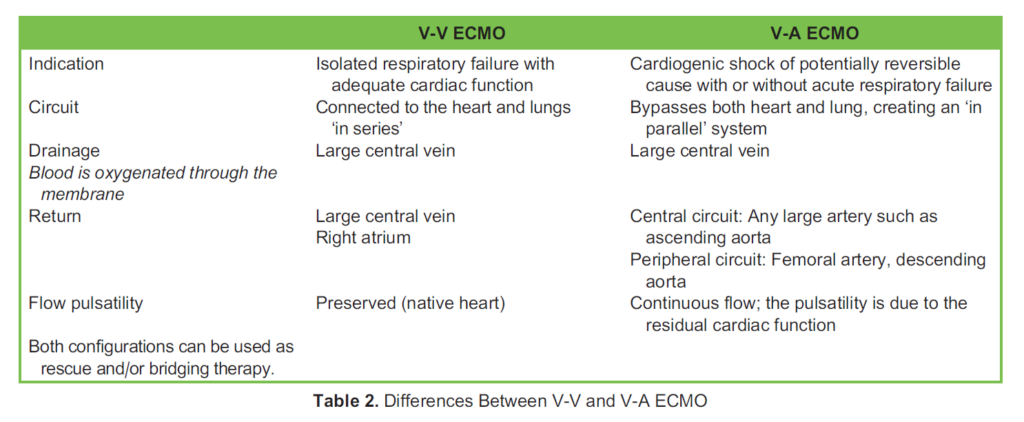
Extracorporeal membrane oxygenation (ECMO) is an external artificial circulation in which venous blood from the patient passes through a circuit and becomes enriched with oxygen while carbon dioxide is removed. Blood then reenters the patient’s circulation via the venous system (veno-venous, V-V ECMO) or the arterial system (veno-arterial, V-A ECMO). These are 2 distinct configurations of the ECMO circuit, each offering different physiologic support depending on the underlying condition.
V-V ECMO
For V-V ECMO, the main therapeutic strategy involves temporarily supporting patients with respiratory failure during recovery when, despite conventional maximal ventilatory management, they remain hypoxemic or hypercarbic. The three most common reasons for this include severe acute respiratory distress syndrome (ARDS), chronic obstructive pulmonary disease exacerbations and as a bridge to lung transplantation.1 In this context, V-V ECMO provides complete pulmonary support by oxygenating and removing carbon dioxide from the circulation while also limiting the development of ventilator-induced lung injury2 such as alveolar strain, atelectrauma, reabsorption atelectasis and overdistension.3
V-A ECMO
The general indication for V-A ECMO is cardiocirculatory failure (with or without respiratory involvement), with refractory cardiogenic shock. This includes acute myocardial infarction (more than 80% of these cases), fulminant myocarditis, acute cor pulmonale, massive pulmonary embolism, primary transplant graft failure, postcardiotomy cardiogenic shock, acute exacerbation of chronic heart failure, toxic ingestions and refractory arrhythmias.4 V-A ECMO provides both haemodynamic and respiratory support (full cardiopulmonary bypass) as the circuit bypasses both the heart and lungs, and oxygenated blood from the circuit mixes with arterial blood, directly perfusing distal organs.5
INDICATIONS FOR ECMO
Indications for ECMO can be summarised in 2 categories: rescue therapy (to buy time and allow the supported organ to recover) and bridging therapy.
Rescue therapy on the basis of organ support provided5
- Cardiocirculatory system (V-A ECMO)
- Respiratory system (V-V ECMO, V-A ECMO)
- Both (V-A ECMO)
As a bridge to
- Recovery, when the underlying pathology is reversible (such as severe ARDS)
- Transplant (heart, lungs) or while awaiting transplant decision
- Advanced therapy, for example, permanent left ventricular assist devices
The support provided by both V-V and V-A ECMO is temporary, as a bridge to recovery or to allow transition to other long-term therapies (surgical assist devices or organ transplantation), but if the patient presents with a severe, irreversible organ disfunction, then it should be avoided.
Indications for V-V ECMO
V-V ECMO should be considered in patients with preserved cardiac function who have 1 or more of the following:
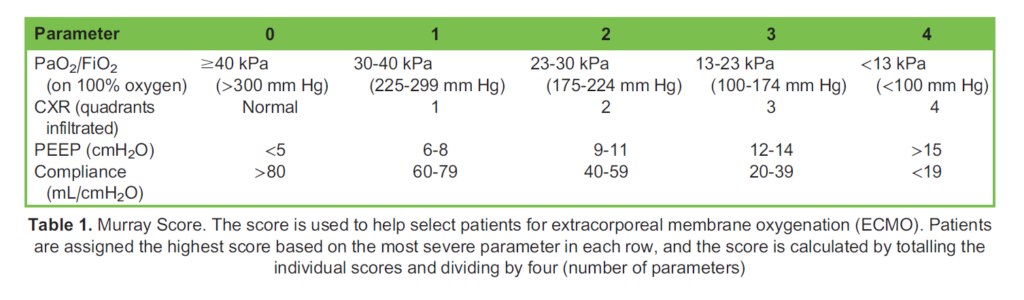
- Hypoxemic respiratory failure associated with a high mortality rate: the Murray score >3 (CESAR; Table 1) or PaO2/FiO2 <80 mm Hg (EOLIA) have been used, but to date, there are no strong data indicating the superiority of either of these6
- Hypercapnic respiratory failure: refractory hypercapnia causing acidosis despite optimal mechanical ventilation and medical management
- Persistently elevated plateau pressures despite optimal mechanical ventilation and medical management
Indications for VA-ECMO
In addition to the clinical conditions mentioned above, V-A ECMO is typically commenced in cardiogenic shock when cardiac output (CO) remains low (cardiac index [CI] <2 L/mi/m2) despite optimal therapeutics. For example, in a patient with low CI and poor left ventricle (LV) and/or right ventricle (RV) function despite adequate intravascular volume, high dose inotropic agents and intra-aortic balloon pump (IABP) (5).
Extracorporeal Cardiopulmonary Resuscitation (ECPR)
In the past few years, more attention has been drawn to ECPR, the emergent initiation of V-A ECMO in a patient who suffers a cardiac arrest and in which conventional CPR has not achieved return of spontaneous circulation. ECPR can be started in those patients in whom the underlying cardiac condition is believed to be reversible. It allows for adequate end-organ perfusion (including the brain) while other resuscitative efforts are continued.7 Evidence from the literature has shown a survival benefit, both short and long term, for patients receiving ECPR when compared with conventional CPR for witnessed in-hospital arrest of cardiac origin.8,9
CONTRAINDICATIONS TO ECMO
There are some contraindications to the initiation of ECMO support, according to the 2021 ELSO guidelines6 and the most recent literature.
Absolute
- Critical comorbid conditions in which recovery, decannulation and organ transplantation are not feasible; these can range from advanced metastatic cancer to fatal intracerebral pathology (cerebral herniation, severe intracerebral haemorrhage or intractable intracranial hypertension) and irreversible destructive damage to the lung parenchyma, where lung healing does not seem feasible after decannulation10
- Patient refusal
Relative
- Mechanical ventilation at high settings (FiO2 >90%, Pplat >30 cmH2O) for 7 days or more
- Central nervous system pathology (haemorrhage, irreversible and incapacitating injury)
- Major pharmacologic immunosuppression (absolute neutrophil count <400/mm3)
- Any contraindication to anticoagulation or systemic bleeding
- Older age (increased mortality risk but no threshold is established; some authors recommend an individual case-by-case choice when patients are older than 70 years11)
- Aortic dissection or severe peripheral arterial disease
- When the cause of cardiogenic shock could be quickly corrected with no ECMO support (these apply to V-A ECMO4)
THE ECMO CIRCUIT
The ECMO circuit consists of an access cannula in the venous system (drainage cannula), a centrifugal pump, a blood gas exchange unit and finally a return cannula that can be placed in the venous system (V-V ECMO) or the arterial system (V-A ECMO) (Figure).
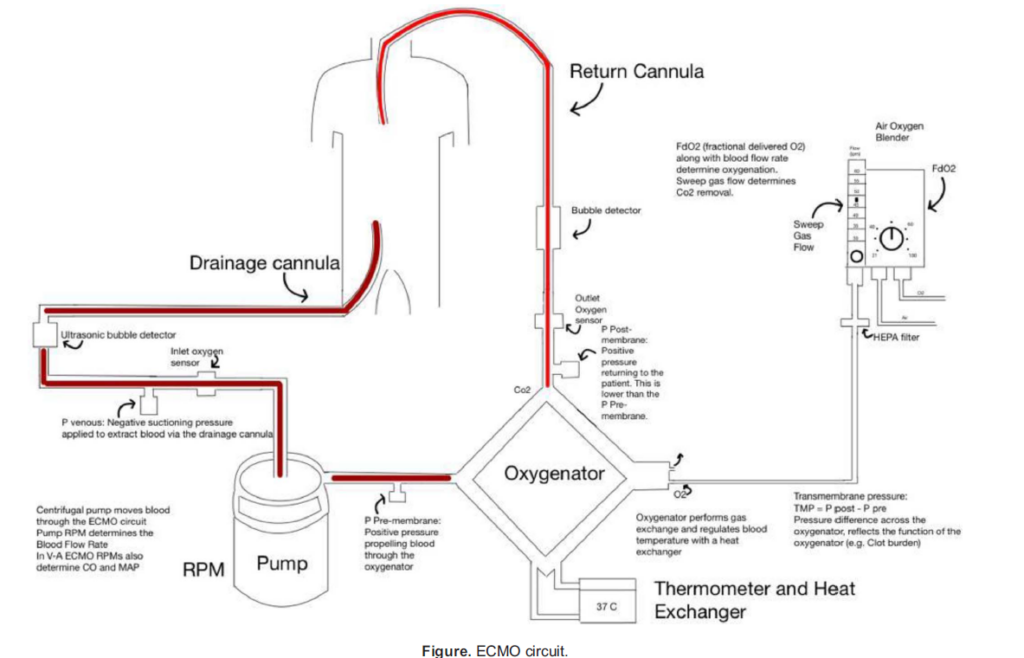
The negative suctioning pressure (inlet pressure, which should not exceed -300 mm Hg) in the drainage cannula allows the blood to flow from the patient’s venous system to the pump, where the centrifugal force then moves the blood to the oxygena- tor. From here, a consistent positive pressure (outflow pressure, which should not exceed 400 mm Hg) in the return cannulas allows blood from the oxygenator to reenter the patient’s circulation.
Therefore, the circuit itself could be functionally divided in three limbs12:
- The drainage limb (from the drainage cannula to the pump)
- The limb between pump outflow and the ‘venous’ side of the oxygenator
- The return limb, extending from the ‘arterial’ side of the oxygenator to the return cannula
Although the two configurations differ slightly, the main principles remain the same (Table 2). In V-V ECMO, the deoxygenated blood is drained from a large central vein, and the oxygenated blood is returned to either the right atrium or another large central vein. In V-A ECMO, venous blood is drained from a large central vein and is returned to the arterial circulation either centrally (via the ascending aorta) or peripherally (via the femoral artery and eventually into the descending aorta).
Circuit Components
Cannulas
Modern cannulas are strong and thin walled and made of biocompatible silicone polyurethane (which is malleable at body temperature, aiding insertion). They are sometimes coated with additional polymers to reduce platelet activation and triggering of the inflammatory cascade. However, no coating to date has been proved to eliminate the well-known risk of bleeding and coagulopathy associated with circuit exposure. The cannulas are also reinforced with stainless steel to reduce the risk of kinking and collapsing (with subsequent ECMO flow interruption) sometimes caused by high suction pressures.13
The drainage cannula allows blood to flow through the pump to the oxygenator. They are typically 23 to 29 F (about 7.5- 10 mm in diameter) and are configured with multiple holes on the sides to optimise suctioning of blood from the circulation. They are normally sited in large central veins such as the inferior vena cava via the femoral vein or the superior vena cava (SVC). The tip should be sited at the border between the right atrium and SVC to minimise the risk of recirculation in V-V ECMO.
The return cannula returns blood from the oxygenator to the patient. They are typically 15 to 19 F for V-A ECMO or 21 to 23 F for V-V ECMO. They are sited in either the right atrium (for V-V ECMO) or the femoral artery with the tip either in the iliac artery or in the distal abdominal aorta (for V-A ECMO).
Pump
Roller pumps, which generate direct suction on the inflow catheter, have been used for decades but have now been replaced by novel centrifugal pumps.14 The latter seem less destructive of blood components, with reduced platelet activation and reduced inflammatory response.15 Centrifugal pumps use a rotating system of blades (called impellers) that are magnetically coupled to a motor. The blades spin to create a vortex, resulting in a pressure difference to drive the blood flow. Typical pump speeds are between 2000 and 6000 rotations per minute (RPM), and the rate at which the blood travels through the pump is the flow (L/min). An external interface displays the speed, flow, inflow and outflow pressures and their difference (Dp). The pump settings can also be adjusted using this interface.
An excessive increase in Dp (Pin–Pout) when all the other variables are stable might indicate deterioration of the membrane (see below), which could negatively affect gas exchange, therefore requiring replacement of the component.
Requirements of extracorporeal blood flow vary on the basis of the underlying pathology:
- 3 L/m2/min for cardiac support (V-A ECMO)
- 60-80 mL/kg/min for respiratory failure (V-V or V-A ECMO)16
In general, about 3 to 4 L/min is indicated for an 80-kg adult; therefore, pump speed should be adjusted to allow this.6 This flow rate would support the normal metabolism of the patient, and this is valid for both V-A and V-V ECMO.
Flow is dependent on various factors in the circuit, such as pump speed (RPM), blood volume, preload, downward resistance and afterload. A higher flow will be determined by higher pump speed, high blood volume or high preload. In contrast, high down- stream resistance or afterload will lead to low flows. Blood flow (L/min) is fundamental in V-V ECMO, and haemodynamic stability is necessary to run V-V ECMO, as it supports only pulmonary and not cardiac function. V-V ECMO will contribute to systemic oxygenation depending on the flow rate relative to the patient’s CO (this is the blood flow/CO ratio).
Blood Gas-Exchange Unit
The blood gas-exchange unit is made of a membrane oxygenator (also known as a membrane lung, or oxygenator). Modern oxygenators are composed of hollow fibres that separate the blood and gas phase and allow gas to diffuse across it. The fibres are usually made of silicone rubber, polypropylene (microporous) or polymethylpentene.16
Blood and gases flow on different sides of the membrane in opposite directions. The counter current gas flow, known as the sweep speed, can be adjusted and can range between 0 and 15 L/min. Carbon dioxide clearance is mainly achieved by adjusting sweep speed (sweep gas flow rate L/min).17 CO2 removal is easily and efficiently achieved by ECMO through counter current diffusion (blood flow rate, L/min). Other factors determining CO2 removal on ECMO are diffusion properties, design and area of the membrane, inlet PCO2 (therefore the gas exchange of the native lung) and presence of water vapour (which can impede CO2 removal).
The air-oxygen blender is a device that determines the desired fraction of oxygen being delivered to the oxygenator. This is called FdO2 (fraction of delivered oxygen percentage) and varies between 21%, O2/air mix, and 100% O2. Therefore, oxygenation is determined by the surface area, integrity and design of the membrane; blood flow rate (L/min) and the time of contact between the blood and membrane; blood flow/CO ratio; the FdO2 of sweep gas; and the oxygen saturation of preoxygenator blood. The membrane lung should provide full O2 and CO2 exchange in those cases in which full support is needed.16
Finally, blood is warmed to 378C by the heat exchanger before being returned to the patient via the reinfusion cannula.
ANAESTHESIA AND VENTILATION FOR ECMO
Sedation, analgesia and paralysis are major issues for critical care patients receiving ECMO, as they present with altered physiology and pharmacokinetics. In addition, the membrane traps some of the most commonly used analgesics and sedatives, requiring higher doses.18 Patients are typically deeply sedated during the cannulation period but may be allowed to wake up after stable support is achieved.
Indications for sedation include the following:
- To relieve pain and anxiety
- To decrease oxygen consumption and CO2 production
- In a restless and agitated patient to prevent dislocation of lines
- To allow a normal sleep pattern at night
- Before any procedure
Indications for muscle relaxants include
- Patient-ventilator asynchrony
- When patient movements interfere with venous return
- When excessive patient movement threatens accidental decannulation
A level of sedation of about 0 and -1 on the Richmond Agitation-Sedation Scale has proved to be enough to manage patients on ECMO with the aid of continuous infusion opioids and analgesics, often without the use of neuromuscular blockage agents.19 Although not all patients on ECMO require endotracheal intubation or mechanical ventilation, most patients on ECMO will require mechanical ventilation at some point. The main difference between a ‘standard’ intensive care unit patient and a patient on ECMO is that the latter does not rely on their native lungs for oxygenation or ventilation, and this allows adjustment of ventilation settings without worrying about the development of hypoxia and/or hypercarbia. In the past few years, it has been observed that patients are extubated sooner than in the past, and more commonly, if they remain awake during ECMO.18
There is insufficient evidence to support one specific ventilation strategy in ECMO. Protective ventilation (tidal volume <6 mL/kg and Pplat <29 cmH2O) may contribute to lung recovery by reducing the release of pulmonary cytokines1 as well as limiting the risk of volutrauma and barotrauma. Other goals to bear in mind are limiting the FiO2 (as hyperoxia can cause reabsorption atelectasis and further damage lung tissue) and maintaining positive end-expiratory pressure (PEEP) to avoid atelectrauma and lung consolidation. V-V ECMO patients can be started on ‘ECMO settings’ or ‘rest settings’, which is a pressure control mode, keeping the driving pressure at 10 cmH2O with a respiratory rate of 10 breaths/min, a PEEP of 10 cmH2O and an FiO2 of 40%. The aim is to keep alveolar units recruited as much as possible while avoiding further injury to the lungs. The tidal volume may be minimal and is expected to improve as lung compliance improves.
SUMMARY
ECMO therapy is indicated in patients with reversible respiratory and/or cardiac pathology when recovery and decannulation are feasible. The ECMO circuit is made of several components that can provide oxygenation and replace heart function, depending on the modality employed (V-V or V-A ECMO). Patients on ECMO have special clinical and pharmacologic requirements to optimise tolerance and safety while receiving treatment.
REFERENCES
1. Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378(21):1965-1975.
2. Kozinn J, Wrisinger WC. ECMO for adults with severe respiratory failure. Mo Med. 2019;116(1):58-62.
3. Schmidt M, Pellegrino V, Combes A, et al. Mechanical ventilation during extracorporeal membrane oxygenation. Crit Care. 2014;18(1):203.
4. Eckman PM, Katz JN, El Banayosy A, et al. Veno-arterial extracorporeal membrane oxygenation for cardiogenic shock: an introduction for the busy clinician. Circulation. 2019;140(24):2019-2037.
5. Makdisi G, Wang IW. Extra corporeal membrane oxygenation (ECMO): review of a lifesaving technology. J Thorac Dis. 2015;7(7):E166-E176.
6. Tonna JE, Abrams D, Brodie D, et al. Management of adult patients supported with venovenous extracorporeal membrane oxygenation (VV ECMO): guideline from the Extracorporeal Life Support Organization (ELSO). ASAIO J. 2021;67(6):601-610.
7. Kumar KM. ECPR-extracorporeal cardiopulmonary resuscitation. Ind J Thorac Cardiovasc Surg. 2021;37(suppl 2):294-302.
8. Chen YS, Lin JW, Yu HY, et al. Cardiopulmonary resuscitation with assisted extracorporeal life-support versus conventional cardiopulmonary resuscitation in adults with in-hospital cardiac arrest: an observational study and propensity analysis. Lancet. 2008;372(9638):554-561.
9. Shin TG, Choi JH, Jo IJ, et al. Extracorporeal cardiopulmonary resuscitation in patients with inhospital cardiac arrest: a comparison with conventional cardiopulmonary resuscitation. Crit Care Med. 2011;39(1):1-7.
10. Harnisch L-O, Moerer O. Contraindications to the initiation of veno-venous ECMO for severe acute respiratory failure in adults: a systematic review and practical approach based on the current literature. Membranes. 2021;11:584.
11. Supady A, Taccone FS, Lepper PM, et al. Survival after extracorporeal membrane oxygenation in severe COVID-19 ARDS: results from an international multicenter registry. Crit Care. 2021;25:90.
12. Poonam Malhotra Kapoor. Manual of Extracorporeal Membrane Oxygenation in the ICU. 1st ed. New Delhi, India: Jaypee Brothers Medical Publishers; 2013.
13. Strunina S, Hozman J, Ostadal P. The peripheral cannulas in extracorporeal life support. Biomed Tech (Berl). 2019;64(2):127-133.
14. Lequier L, Horton SB, McMullan DM, et al. Extracorporeal membrane oxygenation circuitry. Pediatr Crit Care Med. 2013;14(5 suppl 1):S7-S12.
15. Morgan IS, Codispoti M, Sanger K, et al. Superiority of centrifugal pump over roller pump in paediatric cardiac surgery: prospective randomised trial. Eur J Cardiothorac Surg. 1998;13(5):526-532.
16. Extracorporeal Life Support Organization. ELSO Guidelines for Cardiopulmonary Extracorporeal Life Support. Version 1.4. Ann Arbor, MI: Extracorporeal Life Support Organization; 2017.
17. Guglin M, Zucker MJ, Bazan VM, et al. Venoarterial ECMO for adults: JACC scientific expert panel. J Am Coll Cardiol. 2019;73(6):698-716.
18. Mossadegh C. Monitoring the ECMO. Nursing Care and ECMO. 2016 Nov 10:45-70.
19. DeGrado JR, Hohlfelder B, Ritchie BM, et al. Evaluation of sedatives, analgesics, and neuromuscular blocking agents in adults receiving extracorporeal membrane oxygenation. J Crit Care. 2017;37:1-6.



