Intensive Care Medicine
KEY POINTS
- Once the patient has been established on extracorporeal membrane oxygenation (ECMO), review of the circuit parameters, blood pressure, and laboratory values should be a part of the daily assessment.
- Whilst on ECMO, there may be problems with the circuit, pump, gas exchange, or membrane/oxygenator that might require urgent intervention.
- ECMO is not free from complications, and risks such as of bleeding, thrombosis, limb ischaemia, or cerebral hypoperfusion while patients are on the circuit must be constantly monitored and corrected.
- Liberation from ECMO must be tailored to the patient’s clinical condition, on a case-by-case basis, as standardised criteria do not exist to date.
INTRODUCTION
Extracorporeal membrane oxygenation (ECMO) is an advanced life support technique that can temporarily replace the function of the lungs and/or heart while they recover from underlying reversible pathology. Once the patient has been established on ECMO (please refer to ATOTW 514), review of the circuit parameters, blood pressure, and laboratory values should be a part of the daily assessment. As well, complications can arise from ECMO, and these must be recognized and intervened upon to prevent permanent patient harm.
DAILY ASSESSMENTS FOR ECMO PATIENTS
In addition to routine ICU care, patients on ECMO need specific attention. ECMO circuit parameters, blood pressure, and labo- ratory values should be reviewed with every assessment.
ECMO Circuit Parameters
These can be easily monitored and adjusted on the external ECMO console screen, except sweep gas flow rate and FIO2, which have their own separate flowmeters on the gas blender (Table 1).
Flow
Blood flow is determined by the rotations per minute: the higher the rotations per minute, the higher the flow, and the more oxygen delivered.

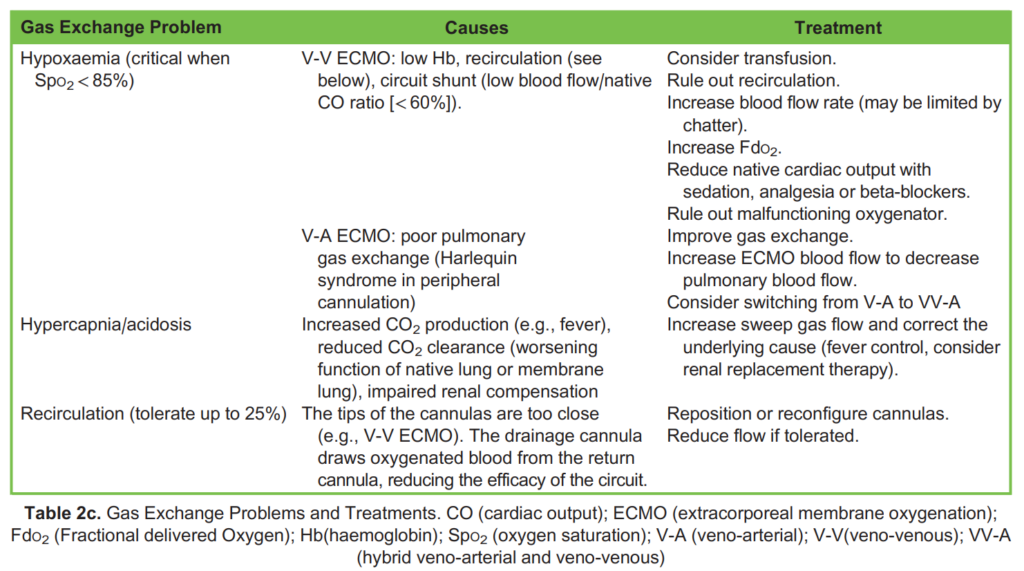
For veno-venous (V-V) ECMO patients, flow rate is set to ensure adequate oxygen supply to tissues, based on delivery to con- sumption ratios (DO2:VO2) rather than to peripheral oxygen saturation:
DO2 ¼ CaO2 3 CO;
where CaO2 is arterial oxygen content and CO is cardiac output. CaO2 is obtained by the following formula:
CaO2 ¼ 1:34 3 Hb 3 SaO2 þ ½PaO2 3 0:003];
where Hb is haemoglobin, SaO2 is the oxygen saturation of arterial blood, and PaO2 is partial pressure of arterial oxygen multi- plied by 0.003 which is the solubility coefficient of oxygen in plasma, therefore further obtaining:
CaO2 ¼ 1:34 3 15 g=dL 3 100 þ ½100 3 0:003] ¼ 20:4 mL O2=dL
Where 1.34 is a constant that expresses the millilitres of oxygen carried by each gram of haemoglobin, 15 g/dL is the standard concentration of haemoglobin in the adult male, and the value 100 corresponds to the expected value of O2 saturation (SaO2).
Cardiac output can be calculated using a Fick estimate. Assuming a normal CO of 5 L/min,
DO2 ¼ 20:4 mL O2=dLð310Þ 3 5L=min ¼ 1020 mL
To calculate VO2:
VO2 ¼ CO 3 ðCaO2 – CvO2Þ
VO2 ¼ 5L=min 3 ð3–5 mL O2=dLÞ 3 10 ¼ 250 mL=min
Based on the above formulas, a normal DO2/VO2 ratio would be around 4:1 (1020:250 ¼ 4,08).
In a similar way, for veno-arterial (V-A) ECMO patients, flow may be increased if there are ongoing signs of cardiogenic shock, and decreased as the patient shows signs of recovery. Flows typically should not be lowered below 1 L/min in an anticoagulated patient or below 2 L/min in a patient without anticoagulation for more than a few minutes, at the risk of circuit clot developing.
Sweep and FiO2
Sweep gas rate should be increased if patients develop hypercarbia or if they show increased work of breathing. As well, FIO2 can be increased in the event of hypoxemia.
Chatter
Chatter is erratic bouncing movement in the ECMO cannula. It usually happens when the pump is trying to suck more blood out of the patient than available. It is either a sign of relative volume deficit or of malposition of the drainage cannula, causing intermittent obstruction to flow.
Anticoagulation
Every patient on ECMO should be anticoagulated unless there is uncontrolled life-threatening bleeding. The consequences of inadequate anticoagulation in ECMO include oxygenator failure and ischaemic stroke.1 An activated partial thromboplastin time (aPTT) ratio of 1.7 to 2.3 or an activated partial thromboplastin time (aPTT) of 4560 seconds is usually appropriate. The oxygenator needs to be visually inspected on a daily basis, to assess the presence of any clots (dark spots) or fibrin deposits (white), and therefore monitor the adequacy of anticoagulation and adjust accordingly.2
Blood Pressure
The arterial line tracing of a patient on V-V ECMO should look like a normal arterial line tracing and the patient should have a pulse pressure. On the other hand, in V-A ECMO the flow from the pump is nonpulsatile, therefore the arterial tracing will lose a considerable amount of pulsatility.
In addition, as the jet from the return cannula is retrograde and competes with the patient’s native CO, it causes a significant increase in afterload. If the patient’s heart cannot generate the force required to overcome this afterload, then the aortic valve will not open, which may lead to the total absence of pulsatility. This is common in severe cardiogenic shock, where only mean blood pressure can be measured.3 Complete absence of pulsatility should prompt investigation of whether blood is being ejected from the left ventricle, as stagnant blood in the pulmonary circulation and heart will clot, with potentially devastating consequences. The left ventricle must in some circumstances be drained with a separate vent limb, or another limb of ECMO return be supplied to the pulmonary artery to restore flow through that circulation.
Laboratory Values
Management of the ECMO patient requires frequent laboratory investigations, a minimum of which would include full blood count with clotting values, arterial blood gases (gas exchange, lactate monitoring), electrolytes, serum urea, and creatinine. According to the most recent guidelines by the Extracorporeal Life Support Organisation, the target for haemoglobin levels should range between 70 and 80 g/L and transfusions should be considered below this threshold.4 pH should be kept at a physiologic range between 7.35 and 7.45, and sweep should be adjusted to correct this.
TROUBLESHOOTING
ECMO troubleshooting can be classified as one of the following:
1. Circuit problems (Table 2a)
2. Pump problems (Table 2b)
3. Gas exchange problems (Table 2c)
4. Membrane/oxygenator problems (Table 2d)
COMPLICATIONS OF ECMO
In addition to the complications already mentioned in the text (bleeding, thrombosis, ischaemic stroke), some additional scenarios are worth mentioning.
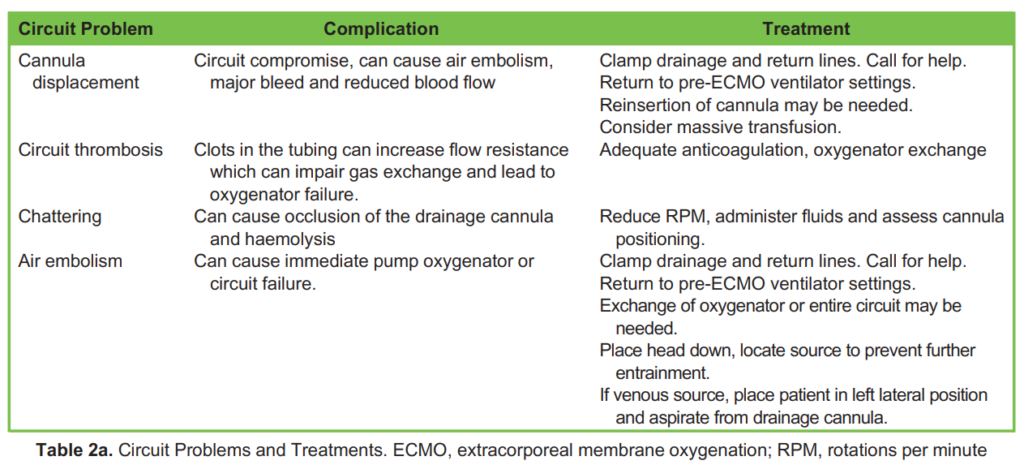

Vascular Injury
This is common at the time of initial cannulation or when patients are decannulated. The incidence is around 7% to 14% and injuries include dissection, pseudoaneurysm, and sometimes bleeding in the retroperitoneal cavity. Large-volume haemato- mas are more common in these patients due to the requirement for systemic anticoagulation during ECMO.5
Limb Ischemia
The risk of limb ischemia increases in peripheral V-A ECMO, when large femoral artery cannulas (> 20 Fr) are inserted (espe- cially if with difficulty) in the context of haemodynamic instability, and in young women who already present with peripheral arterial disease.6 Clinical presentation can range from pain and pallor to gangrene.5 As an additional option, a smaller perfu- sion cannula can be connected to the outflow cannula to prevent limb ischemia.
Circuit Air Embolism
The most common causes of circuit air embolism are through peripheral venous access or via the circuit itself (for example, if the membrane gets ruptured or when connectors between tubing are loose). In the event of air embolism within the circuit, measures are to be taken emergently. Options include clamping the circuit, providing maximal haemodynamic and ventilatory support (FIO2 100% to allow reabsorption of air bubbles) and placing the patient in Trendelenburg position.2
Harlequin Appearance
This condition, also known as north-south syndrome, represents an extreme complication of V-A ECMO, when cannulation occurs peripherally (femoral) and the cardiac function starts to recover.3 The name of the syndrome derives from its specific appearance, with the patient’s head and upper extremities becoming cyanotic, while the lower half of the body looks normally perfused.

Harlequin syndrome occurs because two parallel circulations coexist: one from the venous system, through the ECMO and back to the patient’s femoral artery (bypassing the patient’s heart and lungs), the second one generated by the patient’s recovering cardiac function. The two circulations (blood oxygenated by ECMO and blood whose oxygenation depends on the patient’s lung function) will mix in the aorta. The interface between the anterograde (native) and retrograde (ECMO) flows will vary between the aortic arch and the abdominal aorta, depending on the degree of recovery of native cardiac function.5 If, despite improvement in cardiac func- tion, native gas exchange in the lungs remains poor, blood from the native circulation will be deoxygenated. This will cause hypoxia of the upper extremities (above the mixing point), while the rest of the body (below the mixing point) will receive normal oxygenation (depending on ECMO function). Harlequin syndrome can be dealt with by relocating the arterial cannula, converting the circuit to central V-A ECMO or even veno-arterio-venous (VA-V) ECMO. In the latter modality, a third venous cannula is sited, for example in the internal jugular vein, and connected in a “Y” fashion to partially offload the femoral arterial return cannula.5
WEANING FROM ECMO
Decisions around weaning are made on a case-by-case basis and depend on the clinician’s judgement. Every patient should be eval- uated daily for the readiness to wean. A variety of ECMO liberation protocols have been published, but there are no agreed-upon cri- teria. In general, for V-V ECMO, weaning can be commenced when the pathologic pulmonary process has resolved to the point that the native lungs can provide adequate oxygenation and ventilation. The lungs’ performance can be tested with the cannulae still in place to predict how the patient will do after decannulation.7 A trial off V-V ECMO is reasonable when the native lung starts providing 70% to 80% of the systemic oxygenation, when pulmonary compliance and airway resistance allow for ventilation at reasonable pressures, the FIO2 provided by the ventilator is < 50% to 60% and the partial pressure of arterial CO2 can be maintained at a near to normal level.
When the patient is thought to be ready for weaning, sweep gas flow is reduced to zero and the patient is observed for signs of respiratory distress. Typically, patients are kept “off sweep” for at least 24 hours before a decision is made to decannulate.
Liberation from V-A ECMO is a bit more complex than V-V ECMO and requires the cause of cardiogenic shock to have resolved, along with several criteria. Two of the most widely used ones are below:
1. Mean arterial pressure (MAP) > 70 mm Hg with or without the support of mechanical devices (Intra-aortic balloon pump (IABP), Left ventricular assist device (LVAD)) and with ongoing low doses of vasopressors and inotropes; left ventricular ejection fractions (LVEF) > 25% to 30%; normal right ventricle function.
2. Oxygen saturation > 95% and venous oxygen saturation > 70%; improvement of chest x-ray, normal ventilation, no pulmonary oedema.7
Once the criteria for V-A ECMO weaning are met, the pump support is slowly reduced, which will allow the patient to develop more preload. Blood flow is decreased by 0.5 to 1.0 L/min, but never below 2.0 L/min as this might cause clotting in the circuit at a very delicate stage. Every decrease should be maintained for at least 60 minutes. During attempted weaning, the LVEF needs to be monitored, as well as the ventilator support, since the pulmonary blood flow will also increase. A good indicator of readiness to wean completely is a normal cardiac index (CI) > 2.5 L/min with a LVEF > 25%. Signs of decompensation at this stage might be inadequate CO, haemodynamic instability (MAP less than 60 mm Hg on increased vasopressor support) inadequate tissue perfusion with increase in blood lactates or deterioration of ventricular function on either transthoracic or transoesophageal echocardiography.8 These signs should prompt the clinicians to restart the patient back on full support and monitor recovery.
DECANNULATION
Prior to decannulation, the heparin infusion must be stopped for at least 30 to 60 minutes (or any other systemic anticoagulation held for an appropriate decrement time). In addition, some authors suggest administering an additional dose of inotropes after clamping the cannulas to aid maintenance of CO. The peripheral, percutaneously inserted cannulas can then be removed at the bedside, and pressure applied on the entry sites. Surgically inserted and central cannulas will need removal in theatre, with additional ligation or repair of arterial blood vessels when required. Special attention should be given to venous decannulation, as air can enter the patient’s venous system through the cannulae side holes. This can be easily prevented by performing a Valsalva manoeuvre on the ventilator, or by administering short-term neuromuscular blocking agents. After decannulation, organ perfusion should be closely monitored for any signs of decompensation. Finally, a severe inflammatory response syndrome (SIRS)-like response can be observed after decannulation (in addition to shortly after cannulas insertion). This is referred to as post-ECMO SIRS, and it is not uncommon (60%), lasting up until a week after decannulation.9 Post- ECMO SIRS is challenging to distinguish from systemic infection and should therefore be treated as the latter, until proven oth– erwise, to optimise outcomes.9
SUMMARY
ECMO is an advanced life support technique that can temporarily replace the function of the lungs and/or heart while they
recover from underlying reversible pathology. In addition to routine intensive care unit care, patients on ECMO need
specific attention to parameters such as circuit parameters, blood pressure, and laboratory values. The ECMO circuit itself
may also require troubleshooting or maintenance when certain scenarios arise. ECMO is associated with complications
such as bleeding, thrombosis, ischaemic stroke, and some specific scenarios such as the Harlequin appearance. Every
patient should be evaluated daily for the readiness to wean and once ready, decannulation should be attempted.
REFERENCES
1. Mossadegh C. Monitoring the ECMO. Nurs Care ECMO. 2016;Nov 10:45-70.
2. Eckman PM, Katz JN, El Banayosy A, Bohula EA, Sun B, van Diepen S. Veno-arterial extracorporeal membrane oxygena- tion for cardiogenic shock: an introduction for the busy clinician. Circulation. 2019;140(24):2019-2037.
3. Rupprecht L, Lunz D, Philipp A, Lubnow M, Schmid C. Pitfalls in percutaneous ECMO cannulation. Heart Lung Vessel. 2015;7(4): 320-326.
4. Singh G, Nahirniak S, Arora R, et al. Transfusion thresholds for adult respiratory extracorporeal life support: an expert con- sensus document. Can J Cardiol. 2020;36(9):1550-1553.
5. Al Hanshi SAM, Al Othmani F. A case study of Harlequin syndrome in VA-ECMO. Qatar Med J. 2017;2017(1):39.
6. Pillai AK, Bhatti Z, Bosserman AJ, et al. Management of vascular complications of extra-corporeal membrane oxygenation. Cardiovasc Diagn Ther. 2018;8(3):372-377.
7. Bishop MA, Moore A, Extracorporeal Membrane Oxygenation Weaning. Treasure Island, FL: StatPearls Publishing; 2023.
8. Thangappan K, Cavarocchi NC, Baram M, Thoma B, Hirose H. Systemic inflammatory response syndrome (SIRS) after extracorporeal membrane oxygenation (ECMO): incidence, risks and survivals. Heart Lung. 2016;45(5):449-453.
9. Kirupaharan P, Blazoski C, Hilton R, Feduska E, Leong R, Baram M. Systemic inflammatory response syndrome after extracorporeal membrane oxygenation decannulation in COVID-19 patients. Cureus. 2023;15(3):e36436.



