QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- True or False: Whole blood may be separated into the following components:a. Albumin
b. Cryoprecipitate
c. Dextran
d. Fresh frozen plasma
e. Erythropoietin - True or False: What does fresh frozen plasma contain?a. Red blood cells
b. White blood cells
c. Platelets
d. Albumin
e. Coagulation factors - How are platelets stored? What is their shelf life?
- Will a Rhesus positive foetus carried by a Rhesus negative mother be affected during the mother’s first pregnancy? Explain why.
INTRODUCTION
Blood products are any therapeutic substances that are prepared from human blood. These can be classified into blood components (red cell concentrates, platelet concentrates, fresh plasma and cryoprecipitate) and plasma derivatives (albumin, coagulation factors and immunoglobulins).
Blood component therapy in the UK developed from the use of plasma to produce factor VIII for the treatment of haemophilia in the 1980s. Blood services obtained plasma for fractionation by separating it from whole blood donations. In the UK, whole blood is no longer used routinely for transfusion. Instead, it is processed into its various components and only those that are specifically needed by the patient are used.
In this article, we aim to discuss the blood components and plasma derivatives that we encounter as anaesthetists. There are numerous published guidelines for blood transfusion and blood component therapy. Where relevant, we have included recommendations published by the Association of Anaesthetists of Great Britain and Ireland in 2005, the American Society of Anesthesiologists Task Force on Perioperative Blood Transfusion and Adjuvant Therapies in 2006 and in the fourth edition of the UK Handbook of Transfusion Medicine published in January 2007.
COMPONENT PRODUCTION
Figure 1 illustrates the process by which whole blood is used to produce blood components and plasma derivatives and the steps that are taken to make them safe and effective.
Donors are chosen to exclude anyone whose blood may harm the recipient, for example by transmitting infection, or anyone who may be harmed by donating blood. Donors can give 450-500 ml of whole blood up to three times a year. Blood components may be prepared from whole blood donations or collected by apheresis. Apheresis is the process by which only the component that is to be used is separated out and collected and the rest is returned to the donor.
Whole blood is centrifuged to separate plasma from red cells. Once this process is complete, the concentrated red cells become too viscous to infuse safely and are depleted in the nutrients glucose and adenine. After collection, red blood cells are re-suspended and stored in a preservative solution to keep them in good condition. The most commonly used solution in the UK is SAGM, which contains sodium chloride, adenine, glucose and mannitol. The glucose provides an energy source for the metabolic requirements of the cells, and adenine helps maintain cellular levels of ATP. Saline and mannitol provide an aqueous medium of appropriate osmolality and pH.
Blood screening varies across the globe, but in the UK, all donations are tested for hepatitis B (surface antigen), human immunodeficiency virus (HIV) (antibody), human T-lymphotrophic virus (HTLV) (antibody), hepatitis C (antibody and RNA), and syphilis (antibody). Tests for malaria antibodies, Trypanosoma cruzi (Chaga’s disease) antibodies or West Nile virus RNA are performed when the donor may have been exposed to these infections through travel. Some donations are tested for cytomegalovirus antibody to meet the needs of specific patient groups. No test yet exists for the reliable detection of variant Creutzfeldt-Jakob disease (vCJD). All blood components used in the UK and Ireland since October 1999 have been leucodepleted (removal of white blood cells by filtration) in an attempt to reduce the potential risk from transfusion-transmitted vCJD. Each donation is tested to establish the ABO and RhD group of the donor’s red cells.
Plasma derivatives are partially purified preparations of human plasma proteins that are produced in a pharmaceutical process from large volumes of plasma. Normally at least 20, 000 individual donations are used. Thawing, the addition of ethanol and exposure to varying temperatures, pH and ionic strengths are combined with filtration, chromatography and centrifugation to separate the different groups of proteins. Further purification and virus inactivation steps are also performed. The final products are supplied as solutions or freeze-dried powders. This pooling of huge numbers of donations potentially increases the risk of infective disease transmission.
Since 1999, the UK has imported plasma for fractionation from areas reporting a low incidence of Bovine Spongiform Encephalopathy (BSE), the animal disease associated with vCJD. The USA supplier only provides plasma from male donors to reduce the risk of Transfusion-Related Acute Lung Injury (TRALI). Women are excluded, as they may be a source of antibodies that increase the risk of TRALI. These antibodies are thought to occur during pregnancy.
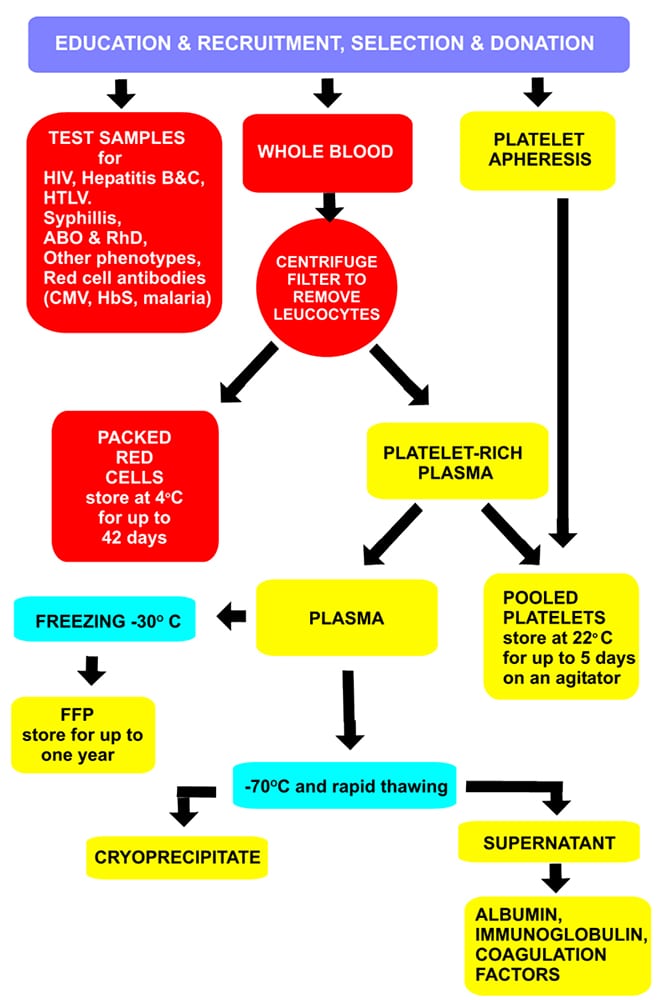
Figure 1. Production of blood components and plasma derivatives
BLOOD COMPONENTS
Blood component therapy is used to correct coagulopathy and thrombocytopenia once exsanguination has been stopped by physical means. The availability of near-patient testing equipment, such as thromboelastography (TEG), can provide useful information to help guide component therapy.
Whole blood
Although in many countries it still accounts for most transfusions, whole blood is rarely used in many developed countries. Whole blood has a shelf life of 35 days and typically, 70ml of citrate preservative solution is added to 420 ml blood.
Red cell components
The objective of a red blood cell transfusion is to improve oxygen delivery to the tissues. The different preparations of red cell components are described below:
Packed red cells
Packed red cells are produced by removing between 150-200ml of citrated plasma from a unit of whole blood. A unit of packed red cells has a haematocrit of between 60-70%. Storing red cells just above freezing allows survival for up to 42 days, however during this time, the cells slowly deteriorate. There is a gradual loss of 2,3-diphosphoglycerate (2,3-DPG), leading to increased haemoglobin affinity for oxygen (left shift of the oxygen dissociation curve) and reduced oxygen delivery to the tissues. There is also gradual loss of membrane integrity as the cells begin to age. Potassium leaks out of the cells into the suspension fluid and there is a gradual build up of lactate. Some cells may eventually die and become haemolysed. Despite these storage problems, giving packed red cells is the fastest way to increase the oxygen delivering capacity of the blood. As a guide, a unit of packed red cells will roughly increase a patient’s haematocrit by 3% and haemoglobin by 1 g/dl.
Microaggregate-free blood
Microaggregate-free blood is used to prevent reactions to leucocyte and platelet antigens.
Specially designed machines are used to wash the red blood cells (RBCs), which are then suspended in sterile saline. Washed red cells usually have haematocrits of 70-80% and a volume of about 180ml.
Saline washing removes residual plasma (98%), and reduces the concentration of leucocytes, platelets and cellular debris. It may be carried out at any time during the shelf life of a unit of blood but because washing is performed in an open system, the resultant red cell component can only be stored for 24 hr at 1-6°C.
Irradiated red cells
Gamma radiation is used to destroy the lymphocytes in a unit of packed red cells that are responsible for transfusion related graft versus host disease. This is used for severely immunocompromised patients, lymphoma patients, stem cell and marrow transplants and unborn children undergoing intrauterine transfusion.
Leucocyte depleted red cells
Leucocyte depleted red cells have had 99.9% of the white cells removed by freezing or microfiltration. This reduces, but does not eliminate the risk of cytomegalovirus (CMV), Epstein-Barr, HTLV infections and febrile reactions.
Since October 1999, all blood components in the UK and Ireland have been leucodepleted in an attempt to decrease the potential risk from transfusion-transmitted vCJD.
Monitoring for blood loss and indications for perioperative red blood cell transfusion 3
A visual inspection of the surgical field should be made regularly during surgery to look for excessive microvascular bleeding (coagulopathy). The amount of blood in the surgical drapes, suction canister, surgical swabs, on the operating theatre floor and in surgical drains should also be monitored carefully.
Usual monitoring systems (e.g., blood pressure, heart rate, oxygen saturation, urine output, electrocardiography) should be used to assess the adequacy of perfusion and oxygenation of vital organs. Special monitoring systems should be used when appropriate (e.g., echocardiography, oesophageal doppler monitoring, mixed venous oxygen saturation and blood gasses).
Haemoglobin or haematocrit should be measured when considerable blood loss or any indication of organ ischaemia occurs. Red blood cells should usually be administered when the haemoglobin concentration is low (e.g., less than 6g/dl in a young, healthy patient). Red blood cells are usually unnecessary when the haemoglobin concentration is more than 10g/dl. These are not absolute limits. For example, the threshold for giving blood may decrease if further blood loss is anticipated.
The decision to administer blood to patients with intermediate haemoglobin concentrations (i.e., 6- 10g/dl) should be based on any on-going indication of organ ischaemia, potential or actual on-going bleeding (rate and magnitude), the patient’s intravascular volume status, and the patient’s risk factors for complications of inadequate oxygenation. These risk factors include a low cardiopulmonary reserve and high oxygen consumption.
Granulocytes
Granulocytes are indicated for life-threatening infections in neutropenic cancer patients who are unresponsive to antibiotics.
They are prepared by separating white blood cells from blood donated by volunteers whose leucocyte count has been increased by pre-treatment with corticosteroids, or those with chronic granulocytic leukaemia. The donations must be cross-matched because they contain large numbers of red blood cells. They should also be irradiated to remove the lymphocytes. They are collected either by apheresis or derived from whole blood and can only be stored for 24 hours at 20-24°C.
Platelets
Platelets are non-nucleated, smooth, disc-shaped cellular fragments that are produced by ‘budding off’ from megakaryocytes in the bone marrow. They measure 2-4 micrometres in diameter and have a circulatory life span of 8-14 days. The normal adult count is 150-400 x 109 /l blood; 10-20% of the total platelet population lies within the spleen.
Platelets are required for primary haemostasis. They release vasoconstrictors (e.g., thromboxane A2 and serotonin), are involved in the initiation of coagulation and are essential for retraction of the final clot.
Collection and storage of platelets
Platelets for transfusion are collected in two ways:
- Pooled platelets. This is a two-step procedure. Firstly, one unit of platelets is produced from a unit of whole blood. Then, 4-6 of these units (from different donors) are ‘pooled’ together in a single pack to be given to a thrombocytopenic patient.
- Apheresis platelets. These have the advantage of being collected from a single donor (to reduce the risk of disease transmission). As blood cycles through the apheresis machine, platelets are removed and all other blood constituents are returned to the donor. The amount of platelets collected with this procedure represents the equivalent of 4-6 units of random donor platelets.
Platelets last for 3-5 days if stored on an agitator at 22°C and at a pH of between 6.2 and 7.8. Each bag has a volume of 250-350ml. Platelets should be inspected prior to infusion and packs should be rejected, or referred for further opinion, if there is any unexpected appearance such as discolouration. Platelets are not usually cross-matched with the recipient, but where possible ABO specific platelets should be used.
The risk of transmission of bacterial infection is higher with platelet transfusions than red cells, particularly if they have been stored for 3 days or more. Bacterial contamination may occur at the time of collection and the storage bags are made of special plastic, which allows gas exchange (oxygen and carbon dioxide) to occur across its walls at 22°C. This helps preserve platelet function but promotes bacterial growth. The longer the platelets are kept prior to transfusion, the higher the risk of bacteraemia.
Recommendations and indications for platelet transfusion 2, 3
Appropriate use of platelet transfusion can reduce the volume of red blood cells transfused.
When a platelet transfusion is considered, the quality of endogenous platelets should be taken into account as well as the patient’s platelet count. Platelet transfusion may be indicated despite an apparently normal platelet count if there is known or suspected platelet dysfunction e.g. in a patient taking a platelet inhibitor such as clopidogrel.
In stable patients, a platelet count of >10 ×109 / L, in the absence of active bleeding is satisfactory.
In surgical or obstetric patients with normal platelet function, platelet transfusion is rarely indicated if the platelet count is known to be greater than 100×109 / L and is usually indicated when the count is below 50x 109 / L in the presence of excessive bleeding.
Invasive intervention in a patient with a platelet count of <50×109 / L, e.g. surgery, insertion of a chest drain, percutaneous tracheostomy or central venous line, will require a platelet transfusion to increase the platelet count to >50×109 /L.
The decision on whether to give platelets to patients who have platelet counts between 50 and 100×109, should be based on the potential for platelet dysfunction, expected or ongoing bleeding and the risk of bleeding into a confined space (e.g., the brain or eye). Thromboelastography may be useful in guiding platelet therapy.
When thrombocytopenia is due to increased platelet destruction (such as heparin induced thrombocytopenia, idiopathic thrombocytopenia purpura and thrombotic thrombocytopenic purpura), prophylactic platelet transfusion is ineffective and rarely indicated.
Platelets are administered in adult dose equivalents. Each adult therapeutic dose can be expected to raise the platelet count by approximately 20×109 / L in most adult patients.
Fresh frozen plasma
The use of fresh frozen plasma (FFP) has increased significantly in the past few years. FFP is mainly used to provide replacement coagulation factors in cases of excessive bleeding or to prevent bleeding in those patients with abnormal coagulation. It contains all the coagulation factors and albumin. It is also a source of plasma cholinesterase.
Collection and storage of FFP
FFP is collected as the supernatant after centrifuging a donation of whole blood. It is frozen within 8 hours and may be stored for up to 1 year at -30°C. Under these conditions, the loss of Factors V and VIII is kept to a minimum. Frozen packs are brittle and should be handled with care.
The frozen plasma can be thawed using a dry oven (10 minutes), microwave (2-3 minutes) or a water bath (20 minutes). Thawed FFP is best used immediately but may be stored at 4°C and infused within 24 hours, provided it is kept at this temperature or returned to the blood bank for storage within 30 minutes of being removed from a 4 ̊C fridge or transport box.
Recommendations and indications for FFP transfusion 3
FFP transfusion is indicated for:
- Correction of excessive microvascular bleeding in the presence of a PT greater than 1.5 times normal, an INR greater than 2.0, or an aPTT greater than twice normal.
- Correction of excessive microvascular bleeding that is secondary to coagulation factor deficiency in patients transfused with more than one blood volume (approximately 70ml/Kg) and when PT or INR and aPTT cannot be obtained in an appropriate amount of time.
- Urgent reversal of warfarin therapy when human prothrombin complex is unavailable
- Correction of known coagulation factor deficiencies for which specific coagulation factor concentrates are unavailable.
- Heparin resistance (antithrombin III deficiency) in a patient requiring heparin.
FFP is not indicated purely to increase plasma volume or albumin concentration.
Fresh frozen plasma should be given in doses calculated to achieve a minimum of 30% of plasma factor concentration (usually achieved with administration of 10-15ml/kg of FFP), except for urgent reversal of warfarin anticoagulation, for which 5-8ml/kg of FFP is usually enough. 4-5 platelet concentrates, 1 unit single-donor apheresis platelets, or 1 unit fresh whole blood provide a quantity of coagulation factors similar to that contained in 1 unit FFP.
In the UK, all children born after 1st January 1996 (following which the risk from contaminated foods was vastly reduced) who require plasma, receive FFP sourced from the USA to minimise vCJD transmission. This FFP is methylene blue treated to minimise the risk of viral infection.
Cryoprecipitate
If FFP is thawed to refrigerator temperature (4°C) a precipitate forms that is very rich in high molecular weight proteins, including factor VIII, von Willebrand factor and fibrinogen; this is known as cryoprecipitate. Viral infection risk is as for whole blood, and it is administered through a standard blood filter.
Collection and storage of cryoprecipitate
UK and Irish Blood Centres prepare cryoprecipitate in volumes of 20-40ml. 1 unit of whole blood provides about 15ml of cryoprecipitate. One pack contains at least 150-300mg of fibrinogen and 70iu of factor VIII.
Recommendations and indications for cryoprecipitate 3
Transfusion of cryoprecipitate is rarely indicated if fibrinogen concentration is greater than 150mg/dl. The indications for its use are as follows:
- When the fibrinogen concentration is less than 100mg/dl in the presence of excessive microvascular bleeding.
- To correct excessive microvascular bleeding in massively transfused patients when fibrinogen concentrations cannot be measured in a timely fashion.
- For patients with von Willebrand’s disease and congenital fibrinogen deficiencies.
The decision to transfuse patients with fibrinogen concentrations between 100-150mg/dl should be based on the potential for anticipated or ongoing bleeding and the risk of bleeding into a confined space (e.g., brain or eye). Bleeding patients with von Willebrand disease should be treated with specific concentrates if available. If concentrates are not available, cryoprecipitate may be indicated.
Cryoprecipitate is usually issued by the hospital blood bank as 10 prepared units of cryoprecipitate (300 ml volumes). It may be pooled in one bag.
PLASMA DERIVATIVES
Human albumin solution (HAS; previously called plasma protein fraction)
When all the cellular material and coagulation factors have been removed from plasma, it contains mostly albumin. This is available as 4.5% and 20% (salt poor albumin) solution. Both contain 140-150 mmol/L sodium, but the latter contains less sodium per gram of albumin. Neither contains clotting factors.
Albumin is heat-treated to kill viruses. It has a shelf life of 2 years and should be stored at room temperature.
It has been used to restore circulating blood volume where volume deficiency has been demonstrated, for example, after removing ascitic fluid, but it has largely been superseded by non-human colloidal solutions.
Immunoglobulin
Immunoglobulins (antibodies) are proteins secreted by plasma cells involved in immunological defence systems. They may be administered to humans and are obtained from:
Pooled human plasma
- Human normal immunoglobulins – These are used as long-term replacement therapy for missing or poorly functioning immunoglobulins in patients with antibody deficiency disorders and also, in short, high dose courses, as immunomodulatory agents in a range of autoimmune or inflammatory diseases. It may be given intramuscularly for the prophylaxis of certain infections, e.g. measles, hepatitis and rubella in pregnant women. Some forms are given intravenously as replacement therapy in immunodeficiency and in various immune related disorders.
- Hyperimmune immunoglobulins – These are obtained from donors with high titres of specific antibodies and are used for passive immunization against particular infections, or to avoid potentially harmful immune reactions. They are given intramuscularly and are available against hepatitis B, tetanus, rabies, Rhesus D antigen and herpes viruses. They are screened for hepatitis and HIV infection.
Immunomodulatory therapy with high-dose intravenous immunoglobulin (IVIG)
IVIG is sourced from a plasma pool of 1000-10 000 donors and provides polyclonal immunoglobulins to a wide variety of pathogens. Originally used to treat idiopathic thrombocytopenia and congenital agammaglobulinaemia, the indications for its use have increased over the past 10 years. The mechanisms of action include blocking IgG- binding to Fc receptors on macrophages, anti- inflammatory effects and effects on T and B lymphocytes. Because the product is expensive and often in short supply, it should only be prescribed for conditions where there is good evidence of its effectiveness.
Indications for IVIG are summarised in Table 1.
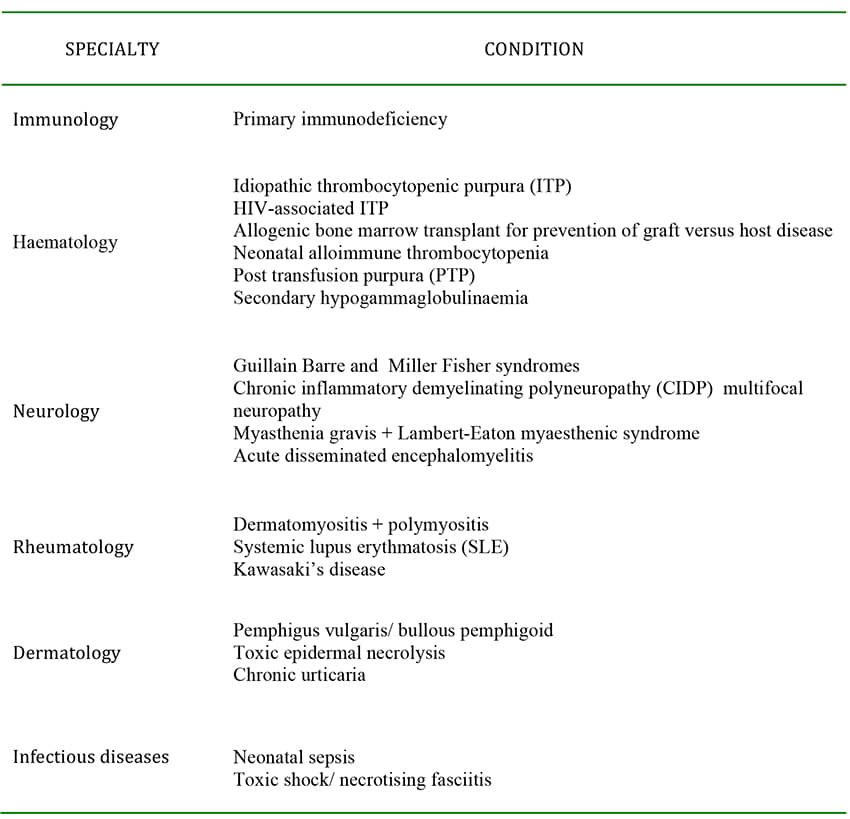
Table 1. Conditions where IVIgG may have benefit 1
Haemolytic disease of the newborn and Anti RhD immunoglobulin (anti-D) 1
Haemolytic disease of the newborn (HDN) occurs when the mother has anti-red-cell IgG antibodies in her plasma. These can cross the placenta and bind to fetal red cells that have the matching antigen. The three most common red cell alloantibodies that cause severe HDN are anti-D, anti-C and anti-Kell (anti-K).
Fetal red cells binding enough maternal antibodies are destroyed in the fetal reticuloendothelial system. This leads to extravascular haemolysis and fetal anaemia. If severe, the fetus may die in utero of heart failure (hydrops fetalis). If the fetus survives to birth, the neonate rapidly develops jaundice and is at risk of neurological damage due to hyperbilirubinaemia. The fetus is only at risk if its red blood cells bear the antigens against which the antibody is targeted.
Development of red cell antibodies in the mother may occur as a result of previous pregnancies or as a result of a previous blood transfusion.
The most important cause of HDN is antibody to the RhD antigen (anti-D). There are no naturally occurring rhesus D (RhD) antibodies. This develops in RhD negative women who have carried a RhD positive fetus. The first pregnancy is usually unaffected although it can sensitise the mother so that future pregnancies with RhD positive babies enhance antibody production progressively, putting later pregnancies at increasing risk. In the UK, the severity and incidence of this condition have been decreased by smaller family size and the introduction of RhD immunoglobulin.
Anti-D immunoglobulin is prepared from the plasma of donors who have high levels of anti-D either following exposure to RhD positive cells during pregnancy or intentional immunization. Anti-D products contain specified levels of anti-D and can be given intramuscularly or intravenously. It is administered to Rh D negative women who may have been exposed to RhD positive fetal red cells that have entered the maternal circulation, for example, following trauma. The anti-D destroys the RhD positive red cells and prevents active immunization, therefore preventing the production of maternal RhD antibodies.
The National Institute for Clinical Excellence (NICE) has recommended that routine antenatal anti-D prophylaxis should be offered to all non-sensitized pregnant women who are RhD negative at 28 and 34 weeks of pregnancy. This is to decrease the number of mothers at risk who still develop anti-D during pregnancy. In addition, anti-D is indicated following potentially sensitizing events that may cause feto-maternal bleeding. Beyond 20 weeks , patients with any of these events should receive anti- D followed by a test that helps to establish the volume of fetal red cells in the maternal circulation (a Kleihauer test or equivalent), as a higher dose of anti-D may be needed. If there is repeated ante partum haemorrhage (APH) during the pregnancy, further doses of anti-D should be given at six-weekly intervals.
Coagulation factor concentrates
Coagulation factors can be transfused individually to treat specific factor deficiencies. These include:
Recombinant factor VIII and factor IX
In the UK, most patients with severe haemophilia now receive coagulation factors obtained by recombinant gene engineering, so the risk of viral infection is removed.
Factor VIII concentrate and factor IX concentrate (plasma derived)
These concentrates are produced by fractionation of large pools of plasma. Current licensed products are all treated to inactivate viral infections and are not reported to transmit hepatitis or HIV. In many parts of the world, cryoprecipitate or plasma are used to treat haemophilia because plasma derivatives are not available or are, like recombinant products, very expensive.
Factor II, VII, IX and X concentrate (prothrombin complex concentrate, PCC: plasma derived)
The main indication for this is warfarin overdose where there is life-threatening bleeding. It has been used in patients with haemorrhage, particularly where there is a contraindication to the use of FFP, but it has not been tested in clinical trials in this situation. It does not contain factor V or VIII.
Recombinant factor VIIa (Novo Seven®)
This was originally developed for use in haemophilia patients with inhibitors to factors VIII or IX of the clotting cascade. Other indications are still being established. It works by activating coagulation and platelet adhesion, but only if tissue factor is exposed. It appears to have only a local effect with no action on systemic coagulation. It requires the presence of platelets and other coagulation factors.
Its use in major haemorrhage secondary to trauma, surgery or obstetric causes is unlicensed. There may be risks of thrombotic complications, and as the drug is currently extremely expensive, UK hospitals have special procedures for making it available.
IMPORTANT POINTS
- Whole blood is a precious resource from which blood components and plasma derivatives are derived.
- Blood components include red cell concentrates, platelet concentrates, fresh plasma and cryoprecipitate. Plasma derivatives include albumin, coagulation factors and immunoglobulins.
- The collection and storage of blood components and plasma derivatives has many phases that are specific to type. These are in place to ensure that the blood product is safe and effective.
- Transfusion of blood components and plasma derivatives is not without risk and requires careful consideration. See References below for blood component transfusion guidelines.
ANSWERS TO QUESTIONS
- True: a, b, d
Dextran is a synthetic macromolecule composed of polymerized glucose subunits. Erythropoietin is a hormone naturally present in human blood. However, it is not practical to purify erythropoietin from blood; instead it is manufactured using recombinant DNA technology. - True: d, e
FFP contains no red blood cells, no white blood cells and no platelets. It is a mixture of coagulation factors and albumin. - Platelets should be stored on an agitator at 22°C and at a pH of between 6.2 and 7.8. (Platelet function is rapidly lost with storage at 4°C. At this temperature, most of the platelets are non-functional by 24 to 36 hours) Platelets have a shelf life of 3-5 days.
- Generally no. The mother will not have any Rh antibodies (e.g. anti-D antibodies) unless she has been previously exposed to Rh-positive blood. This could occur in a previous pregnancy with an Rh- positive foetus. With the appropriate use of anti-D passive immunisation and the avoidance of Rh- positive transfusions in women of childbearing age, Rh sensitisation during delivery in this situation should be a rare event.
WEBLINKS
- McClelland, DBL. Handbook of Transfusion Medicine, United Kingdom Blood Services, 4th Edition, January 2007. Available from UK Blood Transfusion and Tissue Transplantation Services Website: www.transfusionguidelines.org.uk/index.aspx?publication=htm (accessed 26 May 2012)
REFERENCES AND FURTHER READING
- Working Party of The Association of Anaesthetists of Great Britain and Ireland (2005). Blood Transfusion and the Anaesthetist. Blood Component Therapy. The Association of Anaesthetists of Great Britain and Ireland, London, 2005.
- American society of Anaesthesiologists Task Force on Perioperative Blood Transfusion and Adjuvant Therapies. Practice Guidelines for Perioperative Blood Transfusion and Adjuvant Therapies: An Updated Report. Anesthesiology; 2006; 105; 198-208.



