General Topics
KEY POINTS
- Blood conservation techniques reduce the risk associated with blood transfusion and improve patient outcomes.
- Strategies should be patient specific and include preoperative optimisation of haemoglobin, management of comorbidities and medications, intraoperative blood preservation techniques, and careful postoperative management.
- A multidisciplinary and multimodal approach is key to effective blood conservation.
INTRODUCTION
Perioperative anaemia is a common finding in surgical patients. It occurs in 20% to 40% of cases and is a specific indicator for increased postoperative morbidity and mortality.1,2 Anaemia is defined by the World Health Organisation (WHO) as haemoglobin (Hb) concentration <130 g/L for men, <120 g/L for women, and <110 g/L for pregnant woman. Historically, red blood cell (RBC) transfusion has been generously used to treat perioperative anaemia and blood loss; however, it has since been shown that a restrictive strategy for red cell transfusion is as effective, if not superior, to liberal transfusion in many critically ill patients.3 It is also important to remember some of the risks associated with transfusion (see Table 1). In 2016, the Serious Hazards of Transfusion (SHOT) report found that 87% of serious complications were related to human error. Pulmonary complications and transfusion-associated circulatory overload were the most common causes of death and morbidity.4
In 2010 the WHO targeted avoidable transfusion by improving patient blood management. It aimed to provide guidance, training, and support for the safe and rational use of blood products and therefore minimise unnecessary exposure to blood products. It targeted 3 pillars of care (see Figure 1).2
These pillars of care can be best addressed by considering the patients’ surgical journey, from preoperative, to intraoperative, through postoperative recovery. We must take a combined approach depending on individual patient requirements and the nature of the intended surgery.
In 2012, a study in Yorkshire looked at the implementation of a local blood management programme for more than 700 hip and knee replacements. They introduced a preoperative algorithm for identifying and treating anaemia and intraoperative guidelines on blood conservation strategies. This was found to reduce the prevalence of anaemia at the time of surgery from 26% to 10%. Perioperative blood loss also fell by 20%; the transfusion rate for total hip arthroplasty went from 23% to 8% and for total knee arthroplasty from 8% to 0%.5
It is important to remember that for some patients, blood conservation may also be the only option in managing perioperative anaemia. For example, the Jehovah’s Witnesses blood doctrine specifically prohibits procedures involving blood ingestion or transfusion. This includes the use of RBCs, white cells, platelets, or plasma.
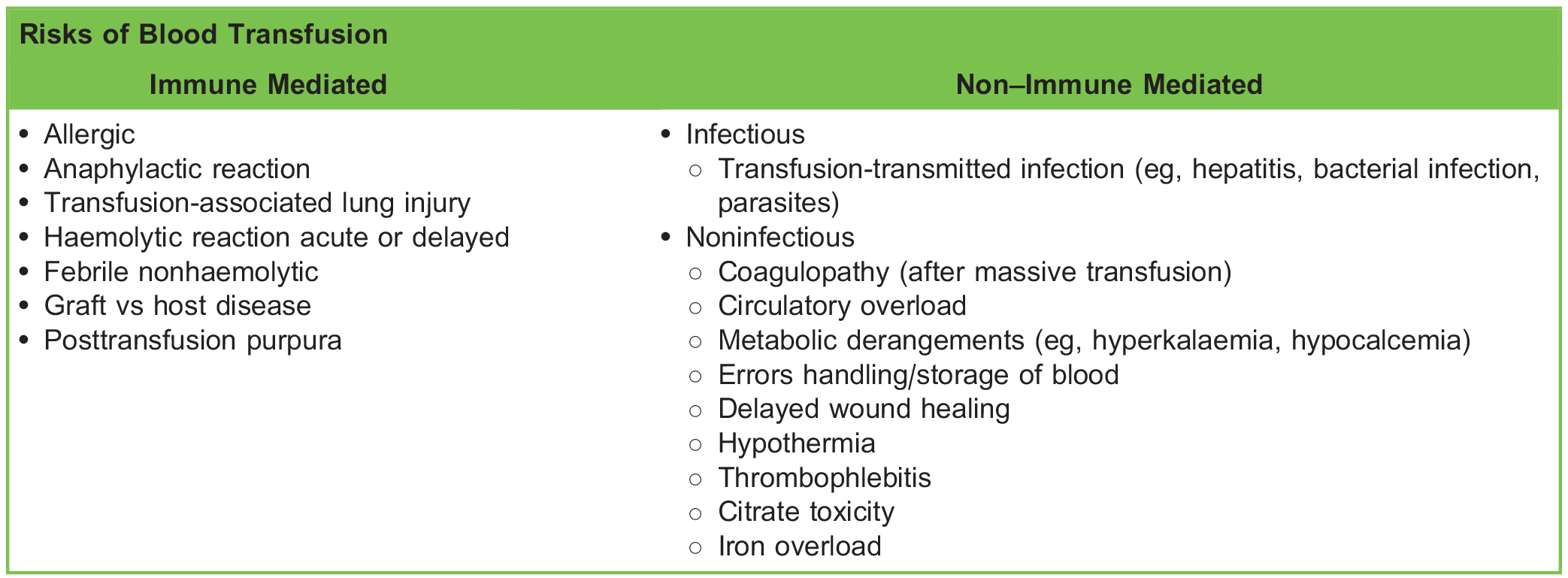
Table 1. Risks Associated With Blood Transfusion
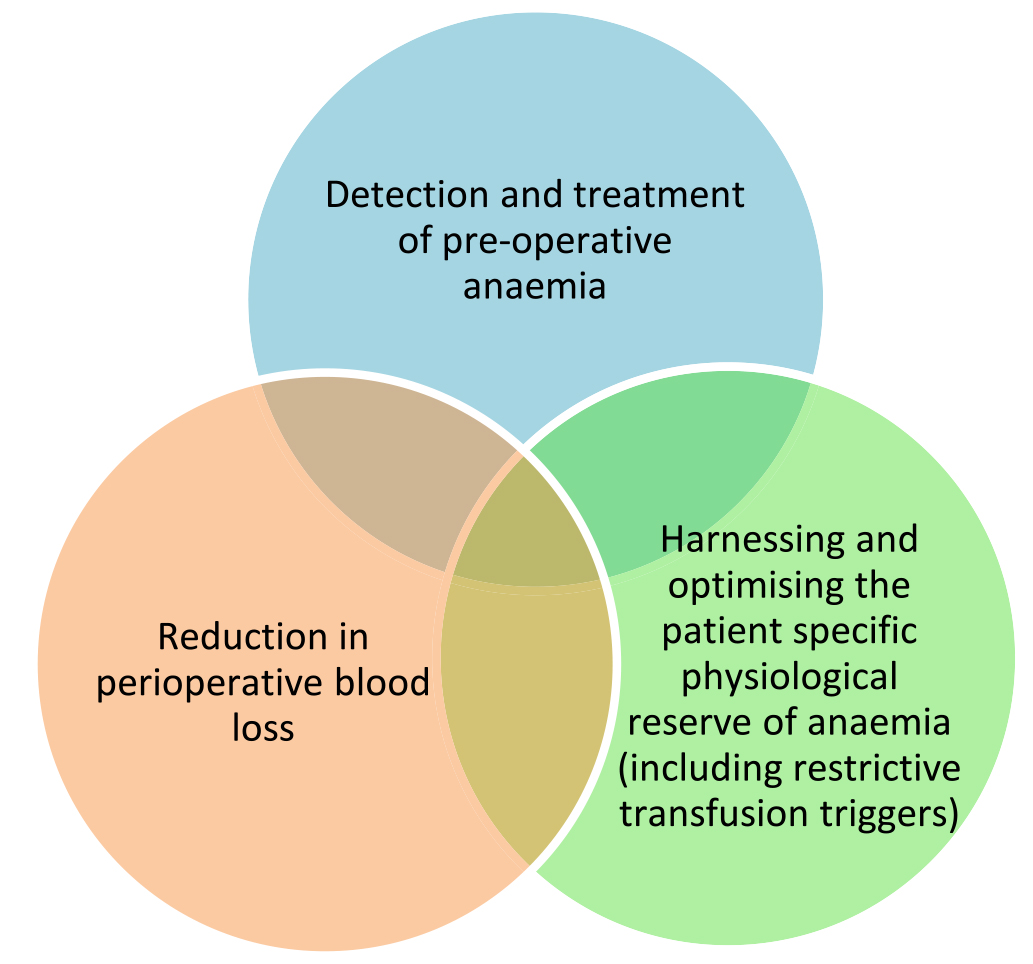
Figure 1. The 3 pillars of care in safe and rational use of blood products.2
PREOPERATIVE STRATEGIES
A patient-specific risk assessment for transfusion should be carried out for all patients but is especially important for those in whom there is potential for high blood loss during surgery, for example, cardiac, spinal, or major lower-limb procedures. Preoperative optimisation includes the identification and treatment of preoperative anaemia with consideration of patient comorbidities such as ischaemic heart disease and the appropriate management of perioperative anticoagulation.
IDENTIFYING AND TREATING ANAEMIA
Identifying the underlying cause of anaemia is key to effective treatment and optimisation. Causes include impaired red cell production, increased breakdown, or blood loss (Table 2). The cause can be identified by a combination of thorough history taking, examination, and simple blood investigations such as Hb level, RBC morphology, and haematinics.
Iron is the most common and widespread nutritional deficiency worldwide and occurs in association with chronic conditions.6 It plays a key role in erythropoiesis and is a critical component in cellular processes such as oxygen transport, mitochondrial respiration, and cellular immunity. Iron levels should be approximately 50 mg/kg body weight, and most of this is in Hb (65%). A further 10% is in myoglobin, and the remaining 25% is in liver, bone marrow, and macrophages. The iron required for daily RBC
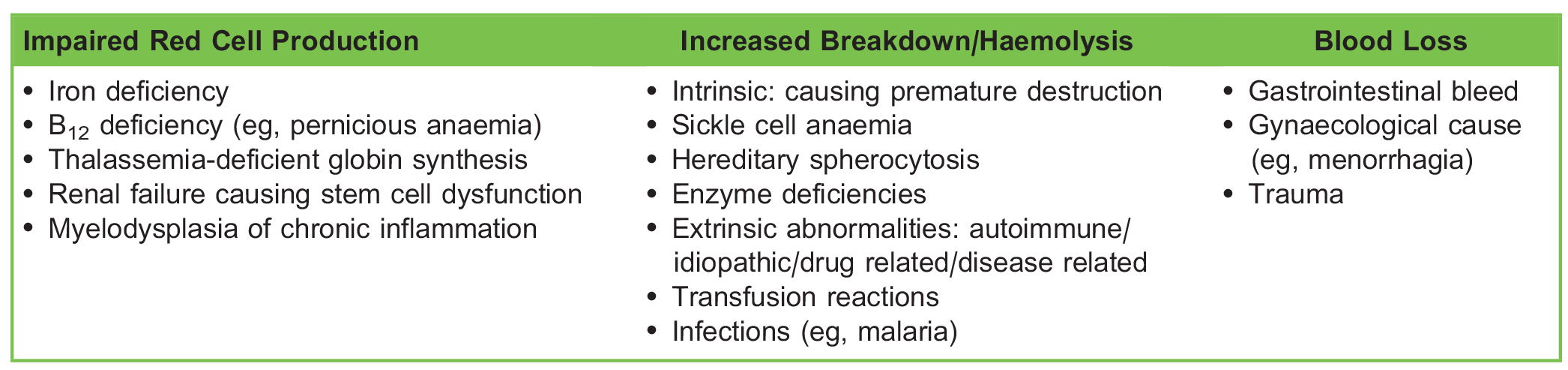
Table 2. Causes of Anaemia
production is provided mainly by macrophages recycling iron from degraded RBCs, whilst daily absorption of 1 to 2 mg replaces extra losses. Increased requirement, limited supply, and blood loss can all lead to a true deficiency. Patients can also have a functional iron deficiency, in which increased hepcidin levels, due to infectious or inflammatory diseases, ageing, or chronic disease, inhibit ferroportin and prevent iron release from macrophages despite adequate total body stores.7
Where there are true low iron levels, early correction with iron replacement treatment is optimal. This can be oral iron such as ferrous sulphate, ferrous fumarate, or ferrous gluconate, and the body can absorb up to 6 mg/d. Parenteral iron therapy can be used if there is a functional iron deficiency or if oral treatment is not tolerated or is ineffective or when time is limited for adequate replacement. An increase in Hb is seen within 2 weeks of parenteral iron therapy and is maximal at approximately 6 weeks.8
Erythropoiesis-stimulating agents (ESA) may also be of benefit to increase RBC production. A short preoperative regimen of ESA or even a single dose alongside parenteral iron has been shown to be successful in both cardiac and noncardiac surgeries for reducing transfusion rates.9
Other causes of anaemia once identified can be treated with replacement of the relevant component, for example, vitamin B12 or folic acid, or involve more specific management plans from the haematologist.
Anticoagulants
Many patients take medications that interfere with coagulation and can lead to a higher risk of perioperative bleeding. Examples in common use include aspirin, clopidogrel, warfarin, and direct oral anticoagulant drugs, such as rivaroxaban, apixaban, and dabigatran. It is important to balance the risks and benefits of stopping these medications perioperatively depending on the thromboembolic versus intraoperative bleeding risk, and this may require a multidisciplinary discussion between the anaesthetist, surgeon, haematologist, and cardiologist to make a detailed plan (Table 3). If the risk of stopping treatment is high, then bridging therapy can also be considered using a shorter-acting heparin to cover the immediate operative period.
Intraoperative
Reducing blood loss intraoperatively involves a multidisciplinary approach and ongoing communication with the surgical team. Individualised goal-directed interventions are important in minimising the need for transfusion. These include combined consideration of patient positioning, thermoregulation, regional anaesthesia, blood pressure management, operative techniques, haemodilution, cell salvage, and the use of antifibrinolytics.
Patient Positioning
Circulatory changes induced by the perioperative positioning of patients can be used to reduce or increase blood loss by promoting or opposing venous drainage in a field. Venous drainage axes should not be compressed, as this can lead to

Table 3. Guidance on Withholding Anticoagulants Prior to Elective Surgery
increased blood loss. Prone positioning, for example, can increase the inferior vena caval pressure and therefore lead to increased risk of bleeding, so appropriate use of pillows under the chest and pelvis is essential to reduce intra-abdominal pressure. Maintaining the surgical field above the heart where possible, for example, in head and neck surgery, can also reduce bleeding at these sites.
Thermoregulation
Hypothermia, which is defined as a core temperature of less than 36°C, can lead to altered enzyme function in the clotting cascade, including effects on fibrinolysis and reduced platelet function.10 The combination of these factors may result in abnormal haemostasis with an increased risk of intraoperative bleeding. Hypothermia also leads to an increase in Hb oxygen affinity, reduced cardiac output, and ultimately a reduction in tissue oxygenation.
Regional Anaesthesia
Loss of sympathetic tone as a result of regional anaesthesia may help reduce the arterial and peripheral venous blood pressure in the surgical field, resulting in less intraoperative blood loss. A meta-analysis of 66 studies in 2006 showed that spinal or epidural anaesthesia was associated with a significant decrease in the estimated blood loss (at the end of the procedure) when compared with a general anaesthetic.11
Blood Pressure Management
Lowering the mean arterial pressure (MAP) in a controlled way reduces end-organ blood flow and may help decrease blood loss (eg, in ear, nose, and throat surgery). The risks of inducing hypotension must be tailored for an individual patient and balanced to ensure appropriate vital organ perfusion. A reduction of 30% below the patient’s usual MAP is thought to be clinically acceptable for patients with an American Society of Anesthesiologists classification of 1.12 A number of methods can be used to lower MAP, including deepening the anaesthetic (volatile or total intravenous [IV] anaesthetic) or using extra opioid such as remifentanil. Additional antihypertensives can also be considered in a closely controlled manner, for example, beta-blockers such as labetalol or esmolol, a potent and titratable arteriovenous dilator such as sodium nitroprusside, or a primary venodilator such as glyceryl trinitrate. Clonidine or magnesium could also be considered. An infiltration of local anaesthetic combined with vasoconstrictors such as adrenaline may be useful to help reduce blood loss in the area of local distribution.
Operative Techniques
A proactive approach to managing bleeding and preventing ongoing loss is crucial to blood conservation. Preventative measures such as tourniquets can be considered, especially with lower-limb surgery, ensuring that the duration of ‘‘tourniquet time’’ and pressure is clearly monitored to avoid muscle and nerve damage.13 Vessel ligation should be proactive rather than reactive. Other methods to stop bleeding early include pressure, ligation, diathermy, and topical vasoconstrictors (eg, use of adrenaline-soaked swabs and the use of fibrin glues).
Haemodilution
Haemodilution may be either normovolaemic or hypervolaemic. Normovolaemic haemodilution involves removing a patient’s blood and replacing it with crystalloid to maintain intravascular volume. The haematocrit is low; tissue perfusion and blood flow are high due to low plasma viscosity. Blood loss intraoperatively will contain fewer RBCs. Once the blood loss has stopped, the patient’s own blood can then be returned.
Hypervolaemic haemodilution is dilution of the blood volume without withdrawing blood first, aiming for a lower haematocrit to prevent overall loss of RBCs if bleeding occurs and to maintain tissue perfusion. Care to avoid circulatory overload must be taken, especially in patients with reduced cardiac function. Haemodilution is not commonly employed.
Cell Salvage
Where major blood loss might be expected, collection, processing, and reinfusion of autologous blood is becoming more widely used and accessible14 (Figure 2).
A recent study showed that 53% of UK hospitals now use cell salvage. Indications include anticipated loss of >1000 mL or 20% of estimated blood volume, increased risk factors for bleeding, patients with rare blood types or antibodies, and patients who refuse to receive donor blood.14
Drawbacks of cell salvage include appropriate training of personnel and the many relative contraindications (see Table 4).
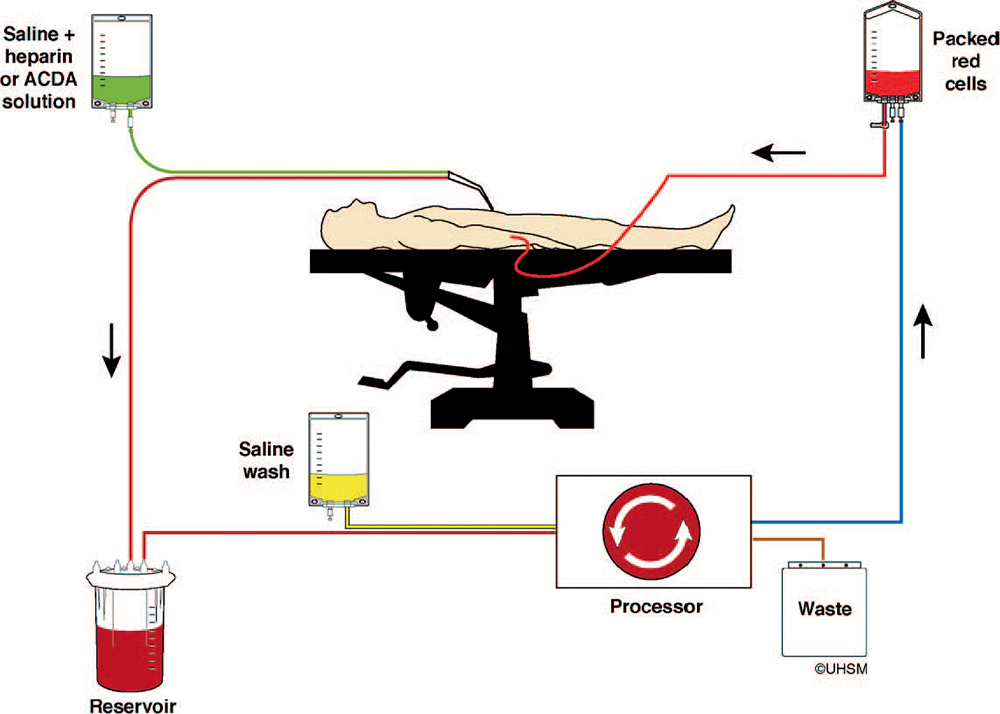
Figure 2. Picture from the Association of Anaesthetists of Great Britain and Ireland intraoperative cell salvage guideline (https://www.aagbi. org/sites/default/files/cell%20_salvage_2009_amended.pdf).

Table 4. Relative Contraindications to Cell Salvage
Antifibrinolytics
Tranexamic acid is an antifibrinolytic used increasingly during surgery to decrease blood loss by helping to prevent the breakdown of fibrin and promoting the maintenance of blood clots. Many trials have demonstrated its effectiveness; the WOMAN trial published in April 2017 was an international randomized controlled trial looking at postpartum haemorrhage in more than 20 000 women. It found a significant reduction in death due to bleeding in patients randomly assigned to receive tranexamic acid compared with placebo (1.5% vs 1.9%), especially when given within 3 hours of delivery.15 The risk of adverse events, including thromboembolism, was not different between the two groups. Across other surgical specialties, a systematic review and cumulative meta-analysis in 2012 looked at the effect of tranexamic acid on surgical bleeding and found that the probability of receiving a blood transfusion was reduced by as much as a third.16 The CRASH-2 trial in 2011 assessed tranexamic acid in trauma patients and found that early treatment with tranexamic acid (<1 hour) significantly reduced the risk of death due to bleeding from 7.7% in the placebo group to 5.3% in the tranexamic acid group.17 However, when given after 3 hours, this effect was reduced.
Postoperative
Blood conservation postoperatively includes close observation and maintaining a high index of suspicion if there are concerns about ongoing blood loss. Pressure dressings can be useful. Cell salvage, using the appropriate aseptic collection systems, can also be considered for collections in drains.
Appropriate replacement of blood products is also an important consideration. Laboratory or point-of-care testing of coagulation can provide useful guidance, for example, giving cryoprecipitate if fibrinogen is low. Where red cell transfusion may be required, an optimal transfusion threshold trigger should be used. In the absence of bleeding, the National Institute for Health and Care Excellence have issued guidance for restrictive transfusion thresholds, which are 70 g/L or 80 g/L in the presence of cardiac disease.6,7 Other management strategies are similar to preoperative techniques and should include, for example, iron replacement. Where the gain from oral iron administration may be limited because of poor gastrointestinal absorption in the postoperative period, IV administration should be considered. Single-dose parenteral iron according to iron requirements and body weight has been shown to be effective, and most patients show a rapid Hb response (50% at 5 days, 75% at 10-14 days, maximal at 3 weeks).6 The safety of parenteral (IV) iron replacement was considered in a recent meta-analysis of 103 trials published from 1965 to 2013 (including 19 253 patients); this analysis concluded that IV iron therapy was not associated with an increased risk of serious adverse events or infection when compared with oral or intramuscular iron or placebo.18
SUMMARY
With such a high incidence of perioperative anaemia and reduced availability and rising costs of donor blood, blood conservation techniques are essential to improving patient care and reducing morbidity and mortality. Strategies should begin well in advance of the day of surgery and should include a multimodal, multiprofessional approach throughout the patient’s perioperative journey.
REFERENCES
- Musallam KM, Tamim HM, Richards T, et al. Preoperative anaemia and postoperative outcomes in non-cardiac surgery: a retrospective cohort study. Lancet. 2011;378:1396-1407.
- Spahn D, Theusinger O, Hofmann A. Patient blood management is a win win: a wake-up call. Br J Anaesth. 2012;108(6):889-892.
- Herbert P, Wells G, Blajchman M, et al. A multicentred randomized controlled clinical trial of transfusion requirements in critical care. N Engl J Med. 1999;340(6):409-417.
- Annual serious hazards of transfusion report. 2016. https://www.shotuk.org/wp-content/uploads/SHOT-Report-2016_ web_11th-July.pdf.
- Kotze´ A, Carter LA, Scally AJ. Effect of a patient blood management programme on preoperative anaemia, transfusion rate, and outcome after primary hip or knee arthroplasty: a quality improvement cycle. Br J Anaesth. 2012;108:943-952.
- Munoz M, Acheson A, Auerbach M, et al. Peri-operative management of anaemia and iron deficiency. Anaesthesia. 2017;72:233-247.
- Thomas D, Hinchliffe R, Briggs C, et al. Guideline for the laboratory diagnosis of functional iron deficiency. Br J Haematol. 2013;161(5):639-648.
- Spahn D, Zacharowski K. Non-treatment of preoperative anaemia is substandard clinical practice. Br J Anaesth. 2015;115(1):1-3.
- Yoo YC, Shim JK, Kim JC, et al. Effect of single recombinant human erythropoietin injection on transfusion requirements in preoperatively anemic patients undergoing valvular heart surgery. Anesthesiology. 2011;115:929-937.
- Rajagopalan S, Mascha E, Na J, et al. The effects of mild peri-operative hypothermia on blood loss and transfusion requirement. Anesthesiology. 2008;108:71-77.
- Richman J, Rowlingson A, Maine D, et al. Does neuraxial anaesthesia reduce intra-operative blood loss? A meta-analysis. J Anaesth. 2006;18(6):427-435. http://www.sciencedirect.com/science/article/pii/S0952818006001371
- Beck J, Johnston K. Anaesthesia for functional maxillofacial surgery. BJA Educ. 2014;14(1):38-42. https://academic.oup. com/bjaed/article/14/1/38/336413/Anaesthesia-for-cosmetic-and-functional.
- Tai T, Chang C, Lai K, et al. Effects of tourniquet use on blood loss and soft tissue damage in total knee arthroplasty: a randomised controlled trial. J Bone Joint Surg. 2012;94(24):2209-2215.
- Blood transfusion and the anaesthetist: intra-operative cell salvage. AAGBI safety guideline. https://www.aagbi.org/sites/ default/files/cell%20_salvage_2009_amended.pdf.
- Shakur H, Roberts I, Fawole B, et al. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post partum haemorrhage (WOMAN): an international, randomised, double-blind, placebocontrol trial. Lancet. 2017;389(10084):2105-2116.
- Ker K, Edwards P, Perel P, et al. Effect of tranexamic acid on surgical bleeding: systematic review and cumulative metaanalysis. BMJ. 2012;344:e3054. http://www.bmj.com/content/344/bmj.e3054.
- Roberts I, Shakur H, Afolabi A, et al. The importance of early treatment with tranexamic acid in bleeding trauma patients: an exploratory analysis of the CRASH-2 randomised controlled trial. Lancet. 2011;377(9771):1096-1101.
- Avni T, Bieber A, Grossman A, et al. The safety of intravenous iron preparations: systematic review and meta-analysis. Mayo clinic proceedings. 2015;90(5):695-696



