Covid-19
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article. Please answer True or False:
- Regarding the history and epidemiology of Ebola:
a. There are 5 known strains of ebolavirus that cause disease in humans
b. The virus is named after the Ebola River near where it was first discovered
c. The current outbreak is caused by the Zaire species
d. Ebola virus disease was first discovered during the 1950’s in outbreaks occurring in the Democratic Republic of Congo
e. Case fatality rates in West Africa are estimated to be as high as 70% - Regarding the transmission and presentation of Ebola:
a. Individuals exposed to the virus are immediately contagious
b. Ebola is an airborne disease similar to influenza
c. Anyone exposed to Ebola should monitor his or her health for 21 days post exposure
d. All patients develop coagulopathy and frank haemorrhage
e. Chest pain, shortness of breath, headache, confusion and convulsions are associated with a poor outcome - With regard to Ebola infection control and management options:
a. Any patient suspected of infection should be isolated immediately
b. Standard surgical masks are sufficient to reduce the risk of aerosol spread
c. Early aggressive supportive therapy improves outcomes
d. Mechanical ventilation and renal support significantly improves outcome in critically ill patients
e. Confirmed cases should be transferred to specialist isolation facilities for further management
Key Points
- Although the threat of imported cases to the UK and other international countries is low, there remains the distinct possibility that further cases may occur
- Consider Ebola virus disease (EVD) as a differential diagnosis in anyone with a positive exposure history and recent fever ≥37.5°C
- If deemed at risk, immediate isolation in a ward-level bed and discussion with local infectious disease service is paramount
- Diagnostic tests may take up to 8 hours to receive a result
- EVD has a high mortality but survival outcomes improve with early aggressive supportive care with intravenous fluid and electrolyte replacement
- Local escalation policies should be pre-agreed and followed
- All confirmed EVD cases should be transferred to a specialist High Level Isolation Facility for further management
INTRODUCTION
Ebola virus disease (EVD), previously known as Ebola haemorrhagic fever, is a rare, lifethreatening viral illness caused by infection with one of the Ebola virus strains.
The most recent epidemic of EVD, beginning in March 2014, has been the largest recorded outbreak, both in terms of geographical spread and case numbers. It has affected multiple countries in West Africa, amongst which the most affected are Guinea, Liberia, and Sierra Leone. In late July the World Health Organisation (WHO) declared the outbreak a ‘’Grade 3 emergency response’’ and, later in early August, a ‘’Public health emergency of international concern’’.
Cases have been reported in Nigeria, Senegal, Mali, the United States of America, Spain and the United Kingdom.
The management of EVD raises a number of practical, logistic and ethical issues. This tutorial aims to discuss the management of suspected and confirmed cases of EVD from a critical care perspective. It is based on recent publications from the World Health Organisation (WHO), Centers for Disease Control and Prevention (CDC), Public Health England (PHE) and North of England Critical Care Network (NoECCN).
Figure 1: An electron micrograph of an Ebola viral particle1
History
EVD is caused by the Ebolavirus, which is part of the filovirus family. Filoviruses can cause severe haemorrhagic fever in human and non-human primates. Four of the five known strains of Ebolavirus can cause disease in humans: Zaire, Sudan, Tai Forest, and Bundibugyo ebolavirus. The fifth species, Reston ebolavirus, has caused severe illness only in non-human primates.2
Ebola was first discovered in 1976 in almost simultaneous outbreaks occurring in the Democratic Republic of Congo near the Ebola River, from which the disease takes its name, and in South Sudan. The disease then disappeared after 1979 and did not reappear again until 1994 in Gabon. Since 1994 sporadic outbreaks have been occurring with increasing frequency.3
Current epidemiology
The current 2014 epidemic is caused by the Zaire species. It is by far the most widespread and intense outbreak recorded and, as of 6th February 2015, a total of 22,495 clinically compatible cases of EVD, including 8,981 deaths, have been reported globally. This number is believed to be an under-representation, as many cases will be cared for outside of the hospital setting.
At present, there have been six non-medically repatriated cases of EVD diagnosed outside of Africa; three imported cases (one in the United Kingdom and two in the US) and three incidents of local transmission to healthcare workers (one in Spain and a further two in the US).
With regard to medically repatriated cases, a total of 18 confirmed EVD cases have been evacuated from Africa. Of this number, 14 have been discharged from hospital, and four have died.
The Zaire ebolavirus is one of the most virulent human pathogens known. The overall case fatality rates in the three intense transmission countries (Guinea, Liberia, and Sierra Leone) are estimated to be as high as 71% whilst, for those hospitalised, mortality rates are slightly lower at 60%. The mortality rate is seen to fall further in EVD cases being treated outside of Africa at approximately 20%.
The risk of further EVD cases being imported to Europe and the U.S. is still considered to be very low due to robust monitoring and surveillance measures in place. However, there remains a distinct possibility that additional cases may occur in the upcoming months raising concerns for the impact this may have on national health services.4
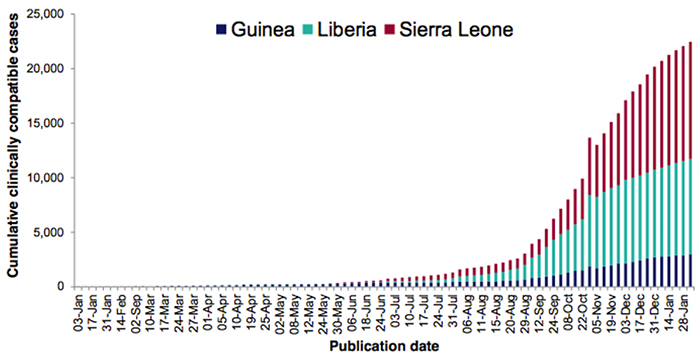
Figure 2: Cumulative clinically compatible cases in Guinea, Liberia and Sierra Leone as of 4 Febuary 20154
Pathogenesis
The natural reservoir of Ebola virus has not yet been identified. Non-human primates are a source of human infection but are not thought to be the reservoir as when infected they too develop fatal illness.
The natural reservoir of Ebola virus has not yet been identified. Non-human primates are a source of human infection but are not thought to be the reservoir as when infected they too develop fatal illness.
The first human case in an outbreak occurs through contact with the body fluids of an infected animal. Person-to-person transmission then follows through:
- direct contact with blood or body fluids (including but not limited to urine, saliva, faeces, vomit, breast milk, semen, and sweat) of an infected individual
- contact with objects contaminated with infected fluids in the absence of strict infection control measures
Traditional burial practices in West Africa, where mourners have direct contact with the bodies of the deceased, have significantly driven the transmission of EVD. Transmission via sexual contact with a convalescent case is also possible as the virus is present in semen and vaginal fluids for up to 3 months after recovery.3
Infection begins once the virus gains entry through unprotected mucous membranes, breaks in skin integrity or via the parenteral route. There is no evidence of transmission of EVD through intact skin or through small droplet spread, such as coughing or sneezing.
The virus migrates to regional lymph nodes and subsequently disseminates to the liver, spleen and adrenal glands affecting a number of target cells including monocytes, macrophages, dendritic cells, endothelial cells, fibroblasts, hepatocytes, adrenal cortical cells and epithelial cells.
Although not infected by Ebola virus, lymphocytes undergo apoptosis resulting in decreased lymphocyte counts and an altered immune response by modulation of gene expression. An early antibody response and high lymphocytic count during the infection is associated with survival. Hepatocellular and adrenocortical necrosis occur causing coagulopathy and shock from impaired steroid synthesis respectively.
Ebola virus appears to induce cytokine and other pro-inflammatory mediator release with subsequent loss of vascular integrity and coagulation defects ultimately resulting in multi-organ failure and shock.5 The pathogenesis of shock is multifactorial and includes fluid loss, fluid sequestration, coagulopathy and sepsis due to secondary infections.
Clinical presentation
Patients with EVD will develop symptoms within 21 days of infection and any symptoms following this period will not be due to Ebola virus. The onset of symptoms typically occurs after an incubation period of approximately 9-11 days.
The table below outlines the symptoms of EVD. Such non-specific symptoms can be easily mistaken for other infectious diseases such as malaria or typhoid fever, or with any other viral or bacterial infection, so a comprehensive travel and exposure history must be obtained.
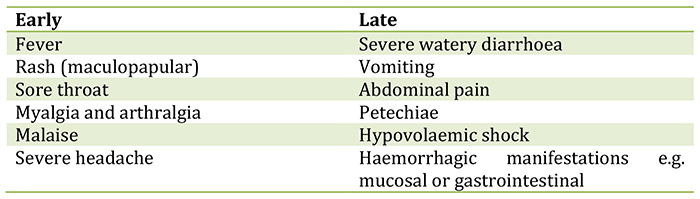
Table 1:Early and late signs and symptoms off Ebola virus disease6
Patients with fatal disease typically develop more severe clinical signs early during infection and usually succumb to the disease between days 6-16. In less severe cases, patients have fever for several days and improve around day 6. In the affected West African countries risk factors associated with a poor outcome are shown below.6

Table 2: Risk factors associated with a poor outcome in West African countries6
Infection control
Prior to isolation, if the patient has been in a public area, the hospital lead for infection control and the local health protection team must be notified, the area cornered off and decontaminated. All staff and public who may have come into physical contact with the patient or body fluids should be assessed.
Infection control measures are a critical part of clinical management. Person-to-person transmission of EVD requires direct contact with body fluids (e.g. blood, faeces or vomit) from a person who has developed symptoms. Therefore, in a secondary care setting, the rapid identification and subsequent isolation of high-risk patients is paramount. The patient should be isolated in a single room with an adjacent contained space to be used for the removal of personal protective equipment (PPE) and waste disposal.
Only staff trained in the correct use of PPE should have patient contact. Staff should attend the patient in pairs with a third member in the relevant PPE outside the room to assist as required. A fourth member of staff should act as a safety officer, ensuring correct PPE use and adherence to local protocols for the safe donning and doffing of PPE.7
The use of PPE is mandatory and guidance from the CDC sets out protection standards for dealing with suspected cases of EVD. Recommended PPE includes the following:
- hand hygiene with soap and water or an alcohol based hand rub
- single-use double gloves with extra-long cuffs
- fluid-resistant single-use coveralls extending to the mid-calf
- fluid-resistant single-use aprons if patients have vomiting or diarrhoea
- respiratory protective equipment (RPE) to reduce the risk of aerosol spread such as
- powered air-purifying respirator (PAPR) with a full face shield, helmet, or headpiece
- N95 or higher respirator in combination with disposable surgical hood extending to shoulders and single-use full face shield8
Please note: the above are recommendations and that individual facilities may elect to use PPE that varies from that outlined depending on local resources.
The key to effective and safe use of any PPE is through consistent implementation by repeated training and practice. Facilities should standardise the PPE in use and provide clear written protocols to avoid the risk of contamination.8
Cleaning and waste management
Contaminated equipment or surfaces should be cleaned and disinfected in a timely manner using standard hospital disinfectants (e.g. 0.5% chlorine solution).
Contaminated material must be immediately segregated at the point of generation, sealed in appropriately labelled containers and destroyed within 24 hours. The preferred method of waste disposal is autoclaving with the contents appropriately disposed of in a designated pit.
Handling of human remains should be kept to a minimum. Appropriately trained personnel must use full PPE when handling the remains of a suspected or confirmed case of EVD. The body should be placed in a leak-proof double bag, with the surface of each body bag being decontaminated with a suitable disinfectant. The body should then be immediately transported to the mortuary or the cemetery and buried promptly. Spraying, washing or embalming of remains in preparation for burial should be discouraged.
Due to the highly infectious nature of the remains post-mortem examinations should be limited to essential evaluations only where again full PPE by trained personnel must be utilised.9
Diagnosis
The diagnosis of EVD should follow a step-wise approach comprising of a comprehensive history, physical examination and relevant investigations. A high index of suspicion of EVD should be present when faced with any individual manifesting the signs and symptoms detailed above along with a positive exposure history.
Current practice in the United Kingdom is based upon recent guidelines published by the College of Emergency Medicine, whereby if a patient is suspected to be at high risk of EVD in that:
- they have travelled from one of the affected areas (currently Guinea, Liberia and Sierra Leone) or have had contact with an individual with Ebola within the previous 21 days, and
- have a fever (≥37.5°C) or history of fever in the past 24 hours7
However, as the symptoms of EVD are non-specific and may mimic those of other diseases a definitive diagnosis can only be made through specific laboratory diagnostic tests including enzyme-linked immunosorbent assay (ELISA) testing, polymerase chain reaction (PCR) and virus isolation.
Ebola virus can only be detected in serum samples after the onset of symptoms, when the viral load is increasing. Diagnostic tests are usually performed on samples of serum and EDTA blood +/- urine.
In the United Kingdom, samples are sent to the Rare and Imported Pathogens Laboratory (RIPL) in Wiltshire, England. In addition to EVD, all samples are tested for other potential causes of fever that the patient may have been exposed to including but not limited to Marburg, Dengue and malaria.
For urgent samples, results may be available within 7-8 hours of sample receipt. Positive results are immediately telephoned to the referring clinician to aid in the timely patient management, infection control and public health response.10
It may take up to three days after the onset of symptoms for the virus to reach detectable levels. It is therefore essential, following an initial negative sample, to send a second sample for testing after at least 48 hours due to the possibility of a false negative result.11
Other supplementary laboratory investigations include haematological and biochemical tests. Findings may demonstrate low white blood cell counts followed later by elevated neutrophils. Serum amylase and hepatic transaminases may be raised, reflecting pancreatic and hepatic involvement. Blood coagulation tests may demonstrate a picture consistent with DIC such as prolonged prothrombin (PT) and partial thromboplastin times (PTT), elevated fibrin degradation products and a thrombocytopenia.6
Management
The increasing international response to the current outbreak has meant that a large number of international healthcare and military staff have been deployed to aid countries with widespread transmission. Such workers have been identified as being at obvious risk of EVD and the CDC and other national agencies have issued guidance for the monitoring, surveillance and, if indicated, escalation plans for returning staff based on their exposure risk.12
Individuals with suspected EVD are likely to initially present to a peripheral hospital so acute health services should have local management protocols in place. Discussion with infectious disease clinicians will help determine whether transfer to a HLIF is indicated.
In peripheral hospitals, it is recommended that cases be managed with the best available PPE, in a ward-level isolation room rather than a critical care setting. The decision to escalate or de-escalate care should be made on a case-by-case basis, taking into consideration the potential risks to the patient and staff. This decision-making process raises many ethical and practical issues so must be based on agreement between at least two local consultants (such as emergency department and critical care consultants) and advice from an infectious disease consultant.
In confirmed cases, a number of key teams are alerted including the outbreak control team, public health and senior government officials, and the clinical team at the nearest HLIF.13
Currently there is no licensed treatment or vaccine for EVD and the mainstay of treatment is primarily supportive. Care focuses on the early detection and supportive care of complications such as hypoxia, hypovolaemia, electrolyte and coagulation abnormalities, septic shock, multi-organ failure and disseminated intravascular coagulation (DIC). This standard supportive care should be provided by the hospital concerned within the limitations of the strict infection control measures.
Monitoring
The monitoring of patients’ vital signs, fluid balance and neurological status through non-invasive means (e.g. pulse oximetry, heart monitor, non-invasive blood pressure) should be carried out on a frequent basis guided by the patient’s clinical condition. Accurate documentation of fluid balance may be difficult particularly in the setting of vomiting and diarrhoea, therefore hourly monitoring of urine output via a Foley catheter and urometer is essential.
Invasive arterial blood pressure monitoring should be considered in those with haemodynamic instability requiring vasopressor support or when frequent blood samples are being taken. Insertion of a central venous line (CVL) solely for central venous pressure (CVP) monitoring is not recommended and in general the use of any form of invasive monitoring should be limited to reduce the risk of exposure to healthcare staff.
Airway management and ventilation
Respiratory involvement is not a common feature of EVD although in severe cases respiratory failure may occur. Airway management may otherwise be required to protect the airway from aspiration in those with reduced level of consciousness or upper gastrointestinal haemorrhage.
The CDC strongly recommend that aerosol-generating procedures such non-invasive ventilation (NIV), bronchoscopy, sputum induction, intubation and extubation and open suctioning of airways be avoided if at all possible. In addition, ventilatory support has yet to demonstrate a significant improvement in survival rates and therefore may not be offered depending on local protocol.
If ventilatory support is to be offered, the Canadian Critical Care Society recommends early intubation with traditional mechanical ventilation in a negative pressure isolation room by highly experienced clinicians wearing appropriate PPE.14
Cardiovascular support and intravenous access
Careful attention to intravascular volume status and aggressive administration of fluids and electrolytes (with a special focus on potassium, calcium and bicarbonate supplementation) constitutes the first step in a series of supportive care interventions. In the non-critically ill patient this should be achieved via the oral route.
Intravenous access is required in those unable to tolerate the oral route or in the presence of haemodynamic instability. Hartmann’s or Ringer’s Lactate has been suggested as the fluid of choice for volume replacement. Large bore peripheral intravenous access is suitable for those with milder disease with central venous access required for those needing intravenous electrolyte replacement or with poor peripheral access.
In the event of needing to establish central venous access, the risk of injury and exposure can be minimised by having an experienced clinician conduct the procedure under ultrasound guidance. Needle-less systems may be used to avoid sharps injuries and the use of non-suture securing devices is advocated.
Cardiovascular support with vasopressors may be indicated, these may be administered either via the peripheral or central route depending on local protocol.
Vigilance must be taken with fluid replacement as with the systemic inflammatory response and loss of vascular integrity, profound third space losses have been observed. The correction of haematological and coagulation abnormalities with blood products may also be necessary.14
Renal support
Renal failure is common in severe cases. Dialysis for renal failure is considered a high-risk intervention for healthcare staff and the Royal Free Hospital in London has therefore ruled out offering this. In the US however, the CDC recommend that the care of patients with EVD should be provided in a hospital with the capacity to perform continuous renal replacement therapy (CRRT). This highlights the importance of regional and national guidelines in outlining clear escalation and management policies.15
Symptom management
Symptoms control is a significant component of EVD management.
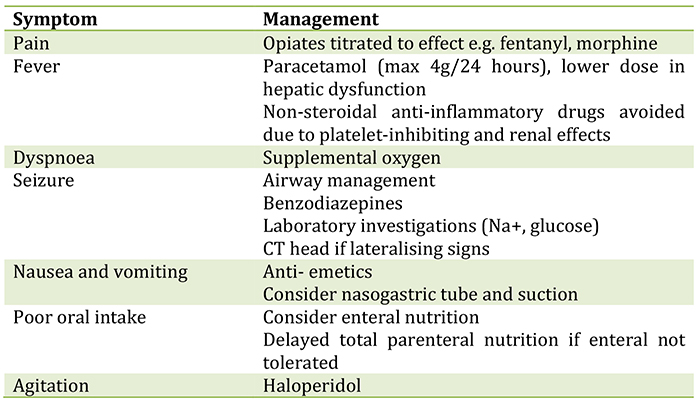
Table 3: Symptoms of EVD and the appropriate management14
Antibiotics and experimental therapies
The treatment of secondary bacterial infections and use of broad-spectrum antibiotics has been suggested in patients with evidence of septic shock and secondary infection. The early discontinuation of antibiotic therapy should be considered if microbiology results and other investigations do not reveal bacterial super-infection.
Other management options may include the early utilisation of experimental monoclonal antibody therapy such as ZMapp. The clinical benefit of ZMapp remains unproven and further trials are required to assess efficacy.
Beyond Zmapp, the development and testing of other antiviral therapies and experimental vaccines is also picking up pace.6,16
Patients who make a successful recovery from EVD develop antibodies that last for at least 10 years. It is not known if people who recover are immune for life or if they can become infected with a different strain of Ebola.6
Paediatric considerations
There are additional issues that must be considered when managing suspected or confirmed EVD in the paediatric population. There is limited information on the current outbreak and the impacts this has on children. However, due to their smaller circulating blood volumes, children are more likely to become fluid deplete from vomiting or diarrhoea, so without rapid intervention, they have the potential to deteriorate more rapidly as compared with adults. It may therefore be desirable to transfer children to a specialist HLIF at an earlier stage (i.e. prior to laboratory diagnosis of EVD) than for an adult.
Children depend on their parents or caregivers for their physical needs and psychological support so the isolation and quarantine of children poses a challenge.
With most infectious diseases, children are often isolated with a parent. However with EVD and the risk of parental exposure, parents may need to be separated from their child. This may impact on the child’s compliance with treatment especially when confronted with clinical staff in full PPE.
The decision to allow parents to accompany a child or to administer sedation to aid with management must be made on a case-by-case basis. Hospital protocols, public health advice and the level of exposure between parent and child that may have occurred before seeking medical care must all be taken into consideration.17
Ethical issues of escalation
The prioritisation and allocation of finite critical care resources occurs routinely throughout the health service. The provision of critical care is based on clinical decisions allowing for the most effective and ethically sound allocation of resources, free from external, political and public influences.
In EVD the most likely situation is that small numbers of patients will require simultaneous treatment. However, the treatment of a single EVD patient will require extraordinary resources. In the event of situations where exceptional demand is placed on critical care, such that resources cannot be provided to all patients who have the ability to benefit, the threshold for accessing critical care consequently rises.
Despite EVD’s current prominence in the media and in public interest, the clinical and ethical principles underpinning such decision-making processes remain unchanged. This must be done on an individual case-by-case basis, with the patient with the higher clinical likelihood of benefit being given precedence.
In the case of EVD, there are two main conflicting ethical stances regarding the provision of scarce critical care resources. The first approach attempts to weigh the potential benefits against harm and in doing so, a balanced judgement can be made which results in the greatest net benefit. Taking into consideration the high mortality rate, the high risk of secondary contamination to healthcare staff and the subsequent denial of effective treatments for other patients due to redistribution of resources, the refusal of level 3 care for patients with confirmed EVD can be justified as the overall perceived harm outweighs the limited expected gains.
The opposing view considers our duties and obligations and how best they may be met. Here the argument is that with strict adherence to infection control measures, the risk to staff and other patients is acceptably low, the reduction in mortality rates with relatively simple supportive care considerable, and the redistribution of work and resources possible with good organisation, planning and communication. This approach would support the provision of critical care interventions to patients with EVD.13
Practical issues of escalation
Currently, there is limited evidence to support the provision of critical care in the management of EVD as, unlike optimum supportive care, the addition of renal and ventilatory support has yet to demonstrate a significant improvement in survival rates.
This attitude may be set to change however, as increasing clinical experience is continually being gained from treating EVD cases in the US and Europe. Practically, the escalation of EVD patients to a critical care setting in a peripheral hospital presents many challenges.
In Texas for example, a 25 bed critical care unit was closed for several weeks when treating an imported case of EVD. The closure of a critical care unit would have a significant impact on the ability of other patients to access a higher level of care. This may result in the restriction of surgeries, conversion of theatre recovery areas and of Level 1 and 2 beds to Level 3 and the rapid retraining and redeployment of medical and nursing staff. Devoting such resources to patients with EVD is likely to affect the hospital’s ability to staff other services.
It is therefore recommended that no peripheral hospital should be required to provide Level 3 care to a patient with confirmed EVD. Such patients should ideally be managed in an isolated ward-level bed prior to transfer to a specialist HLIF.13
At present, in the United Kingdom there is only one HLIF at the Royal Free Hospital in London comprising of two specialist beds available for the management of highly infectious diseases. Three additional infectious disease units in the cities of Newcastle, Sheffield and Liverpool provide surge capacity where EVD patients could be transferred in the event of a larger outbreak, making a total of 26 beds available in the UK.
Answers
- FTTFT
- FFTFT
- TFTFT
References
- Centers for Disease Control and Prevention. (2014). Think Ebola. Available: http://www.cdc.gov/vhf/ebola/pdf/could itbe-ebola.pdf. Last accessed 10th Feb 2015.
- Centers for Disease Control and Prevention. (2013). Viral Hemorrhagic Fevers (VHFs): Filoviridae. Available: http://www.cdc.gov/vhf/virus-families/ filoviridae.html. Last accessed 14th Dec 2014.
- Public Health England. (2014). Ebola: overview, history, origins and transmission. Available: https://www.gov.uk/government/publications/ebola-origins-reservoirs-transmission-and-guidelines. Last accessed 14th Dec 2014.
- Public Health England. (2015). Ebola virus disease: epidemiological update: 6 Febuary 2015. Available: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/402217/ EVD_Epidemiological_Update_6_February.pdf. Last accessed 13th Feb 2015.
- Sullivan, N. Yang, Z. Nabel, G. (2003). Ebola Virus Pathogenesis: Implications for Vaccines and Therapies. Journal of Virology. 77 (18), 9733-9737.
- Centers for Disease Control and Prevention. (2015). Ebola Virus Disease Information for Clinicians in U.S. Healthcare Settings. Available: http:// www.cdc.gov/vhf/ebola/hcp/clinician-information-us-healthcare-settings.html. Last accessed 14th Jan 2015.
- The College of Emergency Medicine, Public Health England. Ebola guidance for emergency departments. CEM, 2014.
- Centers for Disease Control and Prevention. (2014). Guidance on Personal Protective Equipment To Be Used by Healthcare Workers During Management of Patients with Ebola Virus Disease in U.S. Hospitals, Including Procedures for Putting On (Donning) and Removing (Doffing). Available: http://www.cdc.gov /vhf/ebola/healthcareus/ppe/guidance.html. Accessed 10th Feb 2015.
- World Health Organisation. (2014). Interim Infection Prevention and Control Guidance for Care of Patients with Suspected or Confirmed Filovirus Haemorrhagic Fever in Health- Care Settings, with Focus on Ebola. Available: http://apps.who.int /iris/bitstream/10665/130596/1/WHO_HIS_SDS_2014.4_eng.pdf?ua=1&ua=1&ua=1. Accessed 26th February 2015.
- Public Health England. (2014). PHE Microbiology Services VHF Sample Testing Queries. Available: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/388011/PHE_ Microbiology_Services_VHF_Sample_Testing_Queries.pdf. Last accessed 12th Jan 2015.
- World Health Organisation. (2014). Laboratory diagnosis of Ebola virus disease. Available:
http://apps.who.int/iris/bitstream/10665/134009/1/WHO _EVD_GUIDANCE_LAB_14.1_eng.pdf. Last accessed 26th February 2015. - Centers for Disease Control and Prevention. (2014). Interim U.S. Guidance for Monitoring and Movement of Persons with Potential Ebola Virus Exposure. Available: http://www.cdc.gov/vhf/ebola/exposure/monitoring-andmovement- of- persons-with-exposure.html. Accessed 11th Feb 2015.
- North of England Critical Care Network, NHS England. Guidelines for Escalation of Ebola Virus Disease. NoECCN, 2014.
- Canadian Critical Care Society. (2014). Ebola clinical care guidelines. Available from: http://www.ammi.ca/media/69846/Ebola%20Clinical%20Care%20Guide lines%202%20Sep%202014.pdf. Accessed 26th February 2015.
- Centers for Disease Control and Prevention. (2015). Recommendations for Safely Performing Acute Hemodialysis in Patients with Ebola Virus Disease (EVD) in U.S. Hospitals. Available: http://www.cdc.gov/vhf/ebola/healthcare-us/ hospitals/acute-hemodialysis.html. Accessed 13th Feb 2015.
- Lyon GM, Mehta AK, Varkey JB, Brantly K, Plyler L, McElroy AK et al. Clinical Care of Two Patients with Ebola Virus Disease in the United States. N Engl J Med. 2014 Dec 18;371(25):2402-2409.
- American Academy of Pediatrics. (2014). Ebola FAQs. Available: http://www.aap.org/en-us/advocacy-and policy/aaphealth-initiatives/ Children-and-Disasters/Pages/ebola_faqs.aspx. Accessed 11th Feb 2015.



