Paediatric Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding the anatomy and physiology of paediatric central neuraxial blockade:
a. Termination of the dural sac usually occurs at the level of L3
b. The intercristal line (between the iliac crests) occurs close to L5 in children
c. Hypotension following epidural blockade is rarely seen in children less than 8 years of age
d. Clonidine added to caudal solutions rarely causes hypotension
e. Pre-term infants have an almost absent autonomic response to spinal anaesthetics - Regarding paediatic epidural anaesthesia:
a. Lack of parental consent is a contraindication to performing neuraxial blockade
b. There is clear evidence of benefit of neuraxial anaesthetics over intravenous opioids in the provision of post- operative analgesia
c. Toxicity from bupivacaine can be treated with intravenous lipid emulsion
d. Epidural space depth is approximately 0.5mm/kg of body weight
e. Paediatric epidural insertion should be performed awake wherever possible - Regarding complications of epidural anaesthesia in children:
a. Complications of paediatric epidural blockade are similar to those seen in adults
b. Higher rates of complications have been reported in patients undergoing epidural anaesthesia asleep, compared to awake
c. Complication rates are lower in those performed under ultrasound guidance
d. The risk of serious long-term complications is approximately 1 in 10,000
e. Children under 6 months of age have a significantly greater risk of complications compared with older children
Key Points
- Epidural insertion continues to have a role in major paediatric surgery, particularly for perioperative and post-operative pain management
- It is important to understand the variations in anatomy and pharmacology in the neonatal and paediatric population and adjust technique and dosing accordingly
- For children above 6 months old, a widely accepted formula for estimating depth to the epidural space is 1mm/kg body weight
- Adequate post-operative management should be in place in order to improve the safety and efficacy of paediatric epidurals and to minimise potential complications
INTRODUCTION
Paediatric central neuraxial blockade is a well-recognised and valuable technique for achieving perioperative and post-operative pain control. Lumbar and thoracic epidural insertion remain important modalities for pain control and when working effectively, are considered the gold standard for post-operative analgesia in children (1).
Evidence suggests that pain control is at least comparable and in many cases improved when compared with intravenous techniques (2). Epidurals provide optimal analgesia whilst avoiding opioid-related side effects, with studies demonstrating fewer episodes of hypoxaemia or respiratory depression and a reduced need for postoperative ventilation and intensive care (3). There is also greater haemodynamic stability, improved gastrointestinal function, less nausea and vomiting and a reduced neuro-humeral stress response (4). It is worth remembering however that epidural opiates can also result in systemic complications.
The use of paediatric epidural anaesthesia is falling in the UK, with numbers between 2006 and 2011 having fallen by 40% (2). This can be attributed to an increase in laparoscopic surgery, as well as the development and success of other regional or local anaesthetic techniques (2). However epidural and other neuraxial techniques still remain in common practice, primarily employed as an adjunct to general anaesthesia.
The anatomy of the epidural space varies when comparing neonates and children to adults. The physiology and pharmacologic effects of drugs on this age group are similarly variable and these all need to be taken into consideration when performing paediatric epidural anaesthesia. Indications, contraindications and complications should be discussed and the decision for epidural insertion always made on an individual basis after a careful risk/benefit analysis. There continues to be debate regarding safe insertion practices (awake versus asleep), as well as recent developments in technique and performance with the introduction of ultrasound.
EPIDURAL INDICATIONS and CONTRAINDICATIONS
Indications
The majority of neuraxial blocks in children are used in combination with general anaesthesia or sedation, with relatively few situations prompting their use as a sole anaesthetic technique. There may be patients in whom it is preferable to avoid general anaesthesia, for example a hypotonic infant, a child with a history of apnoeas, bronchopulmonary dysplasia or other conditions that may require prolonged ventilation, patients with cystic fibrosis, those with a history of malignant hyperthermia, or occasionally older children who prefer to remain awake (5).
Decisions regarding epidural use should be made on an individual basis and should also consider the potential for additional benefits in children with comorbidities. For example children with Cerebral Palsy undergoing painful lower limb procedures would benefit from epidural use, particularly as pain assessment may be difficult in those with intellectual disabilities or poor communication (6). In patients with asthma or respiratory disease a functioning epidural will serve to block afferent pain pathways and thereby aid maintenance of respiratory muscle function and adequate ventilation (7).
Lumbar or thoracic epidurals are effective in providing analgesia for thoracic, abdominal and orthopaedic procedures. There has been proven benefit of epidural analgesia for general surgery, major urological procedures, orthopaedics, spinal and plastic surgery (8). Compared with intravenous techniques, epidural analgesia, either continuous or patient controlled is associated with lower pain scores and a reduced incidence of post-operative nausea and vomiting (2,8)
Contraindications
Similar to adults, there are a number of absolute and relative contraindications to paediatric epidural insertion. A lack of parental consent or a lack of patient assent (in the older child > 12 years) should preclude insertion in these patients. Other absolute contraindications would include local infection at the site of insertion, coagulopathy or true local anaesthetic allergy (8). Relative contraindications may include anatomic abnormalities, neurological disease, sepsis, immunodeficiency, raised intracranial pressure, previous spinal surgery or the requirement for post-operative testing of motor or sensory function (9).
ANATOMY, PHYSIOLOGY AND PHARMACOLOGY
The anatomy of the epidural space and physiological responses vary with age. This has important implications when considering epidural anaesthesia in paediatric patients. The response to and handling of drugs by the body also varies, particularly in term and pre-term neonates.
The spinal cord terminates at L3 in term neonates, compared with L1 in adults, a difference that remains apparent up to approximately 12 months of age (3). Similarly the dural sac in neonates terminates at S3, but at S1-S2 in adults. An imaginary line drawn between the two superior iliac crests (intercristal line) should be below the level of the spinal cord at any age (3). This line is typically at the L3-4 interspace or the fourth lumbar vertebra in adults but tends to pass closer to the 5th lumbar vertebra in children and the L5-S1 interspace in neonates (3,10). Additional anatomical differences in paediatric patients include incomplete ossification of the vertebral bones, a thin ligamentum flavum and a larger, more compliant epidural space with less fat and fibrous tissue. This allows greater ease for insertion of epidural catheters to higher levels from lower approaches (3,11).
Epidural blockade in children produces significantly less haemodynamic disturbance than is seen in adults (3). Hypotension is rarely seen in children under 8 years of age, a product of a lower circulating volume in the lower limbs and splanchnic system, and a relative lack of resting peripheral vascular tone (3,11). In older patients, the block to sympathetic tone results in a small but consistent reduction in blood pressure, by up to 20-25% (10).
The pharmacology of local anaesthetic agents in neonates and children differs from adults and also with the age of the child. The relatively large cardiac output, high tissue blood flow and lower concentration of plasma proteins for binding (particularly in neonates) result in higher initial plasma concentrations and the potential for toxicity (12). They also have less mature hepatic and renal function until 3 months of age and potentially an increased permeability of the blood brain barrier although the latter remains controversial (3). This is more marked in premature neonates. It is as a result of these factors that paediatric patients are at greater risk of local anaesthetic accumulation and toxicity with epidural infusions (3). Incomplete myelination and an increased permeability of endoneurium in young children means that both duration and latency of nerve blockade are decreased (5).
EPIDURAL INSERTION TECHNIQUE
Principles of safe paediatric epidural insertion apply as they would for neuraxial anaesthesia in all patients and this tutorial will therefore not describe in detail the standard epidural technique. However, it is essential that the procedure includes application of appropriate patient monitoring, skin preparation, maintenance of sterile conditions and use of a test dose to reduce complications. During placement of the epidural needle, the child is typically placed in the lateral decubitus position with hips and knees flexed and spine arched to open the interlaminar space (10).
A midline approach is typically used and a loss of resistance technique applied as in adult patients. The thoracic vertebral spines remain almost horizontal until adolescence, thus a midline approach to the thoracic epidural space can be used (3). A large prospective study (5) by the French-Language Society of Paediatric Anaesthetists (ADARPEF) concluded amongst its findings that it is safer to use loss of resistance to saline rather than to air in neonates and infants (Figure 1).
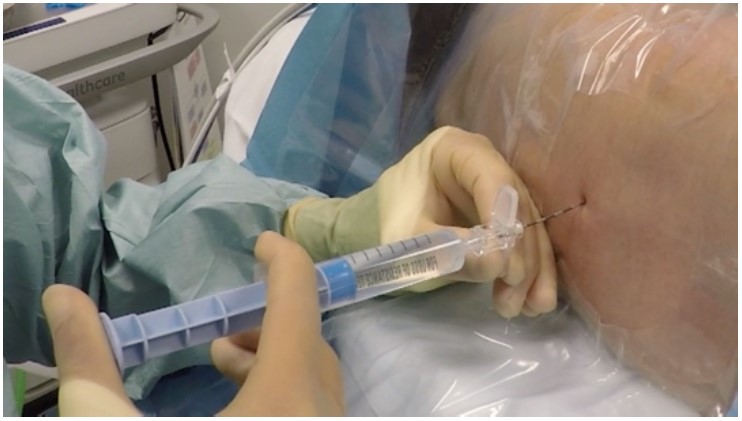
Figure 1: Midline approach for a lumber epidural using a loss of resistance technique with saline. Note the 0.5cm markings on the epidural Tuohy needle.
The epidural space is more superficial in children than in adults, with a number of guidelines available for estimating epidural depth. A widely accepted calculation of depth for lumbar epidural insertion in children between 6 months and 10 years of age is as below:

The mean depth of the epidural space in neonates is approximately 1cm, with a range of 0.4-1.5cm (10). Epidural needles for paediatric use are available as 18G or 19G, with shorter length and 0.5cm markings for easier handling (3).
It is recommended that a test dose together with careful aspiration be used to avoid inadvertent intravascular or subarachnoid injection (5). To assess for intravascular injection, a test dose of 0.1mL/kg (up to 3mL) of local anaesthetic solution such as 0.25% bupivacaine with 5mcg/mL of adrenaline should be given and adequate time allowed for a response. A positive test dose is defined as an increase in heart rate (>10 beats per minute), increase in systolic blood pressure (>15mmHg) or 25% change in T-wave amplitude (10).
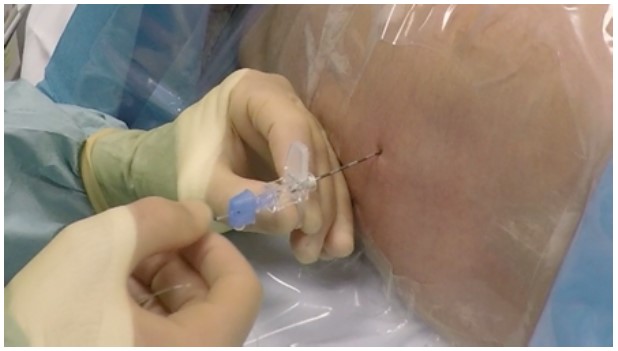
Figure 2: Insertion of the epidural catheter through the Tuohy needle
Drugs and Dosing
The determination of total volume for injection depends on the location of the surgery and the level of the epidural catheter. In younger children, the recommended dose is 0.04mL/kg/segment as an initial bolus. In children older than 10 years, a useful calculation is: Volume (in mL) per spinal segment to be blocked = 1/10 x age in years (10). Perhaps an easier way of calculating this would be to consider the surgery itself and load with 0.25-0.5mL/kg of 0.25% bupivacaine for lumbar epidurals and approximately half that for thoracic. Keeping in mind maximum dose recommendations, this dose of 0.25% can either be repeated with half the initial volume after 1-2 hours or a continuous low-dose infusion commenced, e.g. 0.2-0.4mL/kg/hour of 0.1% bupivacaine (5,10). Drug accumulation may occur in infants under 12 months of age, so infusion rates should be reduced (10).
Bupivacaine, Ropivacaine and Levobupivacaine have been shown to be effective in a variety of concentrations and dose rates, but lower concentrations are generally favored (8). Additive agents may be used in order to prolong the duration of analgesia, improve block quality and reduce unwanted side effects (3). The addition of epidural opioids or clonidine has shown to improve analgesia but can also increase the incidence of side effects, including nausea and vomiting, urinary retention and respiratory depression (8). However it is worth noting that clonidine is not recommended for children under 6 months of age (3). Other drugs considered include Ketamine and Tramadol but there is limited evidence for their safety, particularly in children (13).
Awake vs. Asleep
Paediatric epidurals have been safely placed for many years in anaesthetised patients, with sufficient expert opinion and evidence to support this (2). The factors to consider include limited cooperation; patient movement and discomfort; possible reduced reliability of a test dose under anaesthesia and limited feedback of potential neurological damage in terms of paraesthesia or pain (10). However on account of ethical and practical considerations, it is widely accepted practice for paediatric epidurals to be performed under anaesthesia. There appears to be no greater risk for paediatric regional insertion under general anaesthesia than with them being awake (1,14).
The Paediatric Regional Anaesthesia Network (PRAN) compared regional anaesthesia performed in children awake with those who were sedated or anaesthetized. With a database of over 14 900 blocks, they showed no differences in major or transient complications (15). There was however a lower transient neurological complication rate following epidural insertion in children under general anaesthesia who had not received neuromuscular blocking agents compared with those who had (15). Similarly the ADARPEF study of 24 400 children demonstrated a low complication rate with no serious or long-term sequelae (16). Separate consideration could be given to older children who may be able to cooperate, particularly in those undergoing thoracic epidural insertion.
Ultrasound guidance
With the inherent safety concerns regarding epidural insertion under anaesthesia, the advent of ultrasound has provided a way in which to potentially reduce this risk. Studies have demonstrated that ultrasound can be used effectively to directly visualise neuraxial structures, reduce insertion time and allow visualisation of the spread of local anaesthetic inside the epidural space in children aged 0-6 years (17). Pre-procedure scanning may be of benefit especially in smaller children, in order to localise the block level, assess ligamentum flavum depth and estimate distance to the epidural space (18). Ultrasound penetration and a largely cartilaginous posterior vertebral column mean that epidural placement can be performed real-time in neonates and infants (18).
COMPLICATIONS
Paediatric epidural insertion is not without risk, although recent studies have confirmed the incidence of serious complications to be very low. A follow-up to the original study by the ADARPEF group in 2010 showed an overall complication rate of 0.12% for all regional anaesthesia techniques; however the rate was 6 times higher for central neuraxial blockade than for peripheral blocks (19). Less severe complications include block failure, unilateral or incomplete block, post-dural puncture headache (PDPH) and opioid-related side effects including nausea, pruritis, urinary retention and respiratory depression. PDPH is discussed in TOTW “Effective management of the Post Dural uncture Headache” (20)
The UK National Paediatric Epiural audit collected data on epidural complications over a 5-year period. A total of 96 incidents were reported in over 10 000 epidurals (~1:100) performed, of which 56 were directly associated with the insertion or maintenance of epidural infusion analgesia (21). The remaining 40 incidents were felt to be associated with the use of epidural analgesia, 33 of which were the development of pressure sores (21). The majority of these 56 incidents were of low severity with five incidents (~1:2000) graded as serious. Only one of these children had residual neurological deficit 12 months after surgery, giving an incidence of 1:10 000, comparable to adult rates (21). Considering all epidural performed 28 children experienced post-epidural infection (~1:300), PDPH occurred in 6 (~1:2000), nerve injury in 6 (~1:2000), inadvertent spinal anaesthesia in 2 (~1:5000), drug error in 13 (~1:1000) and local anaesthetic toxicity in 1 (~1:10000) (21). Of the 28 post-epidural infections reported, 25 of these were of local skin infection with 1 case of meningism and 2 diagnosed epidural abscesses (21). Whilst severe complications following paediatric epidural insertion are rare, it should be recognised that the incidence is higher in neonates and infants, with a rate of complications in the under 6 month age group being 4 times that of older children (2).
POST-OPERATIVE MANAGEMENT
A working epidural can be of extraordinary value in providing post-operative analgesia (9). Due to the potential for serious complications and the need to optimise the analgesia provided, there need to be appropriate protocols, monitoring and care in place for all children with post-operative epidurals. Physicians trained in epidural management, educated nursing staff as well as order sheets outlining infusion solutions and dosing regimens help to increase epidural safety and success (10). The presence of a hospital acute pain service should be advocated particularly in centres where high volumes of epidurals are performed.
Post-operative pain should be assessed regularly via patient self-reporting or observational assessment, combined with changes in physiological status. Assessment of the block, its quality and level, can prove more challenging, particularly in smaller children. It should however be performed regularly and in conjunction with an assessment of the patient’s observations, temperature, motor function and an examination of the epidural site. In younger non-verbal children and those with intellectual disability or poor communication skills, for example those with Cerebral Palsy, interpretation of pain and block level can be particularly difficult (1, 6). Attempts to establish pre-operative motor function is very useful to act as a comparison. It is also important to appreciate that, in some patients, the usual behavioural indicators such as moaning, grimacing or altered sleep patterns may often be present in the non-pain state (6). These children may be unsettled for other reasons such as fear, hunger, lack of understanding of the surgery or discomfort with the unusual sensation or impaired mobility in their limbs (15). Despite challenges in assessing these patients, they stand to benefit significantly from reduced pain and spasm after extensive lower limb orthopaedic surgery (6).
Whilst recognising the difficulties in paediatric epidural assessment, it is important to monitor for inadequate block as well as those complications outlined above, and be familiar with how to manage these. According to PRAN, 2-9% of blocks are inadequate or fail and operators need to be aware of methods to manage an “imperfect” block (15). Anatomical variability, presence of visceral pain, variations in epidural site and dosing and inter-patient variability in response to local anaesthetics all contribute to the perceived efficacy of epidural block (15). The regular assessment of observations, temperature, block level, motor function and a review of the epidural site should be performed as outlined above. Significant complications such as severe hypotension, seizures, unexplained fever, signs of local infection, bladder or bowel dysfunction or persistent neurological deficit should warrant urgent physician or anaesthetist review. Non-epidural related causes of such physiological disturbances must also be considered where appropriate.
SUMMARY
Epidural insertion continues to have a role in major paediatric surgery, particularly for perioperative and post-operative pain management. Studies have demonstrated significant benefits and reduced morbidity with epidural use and complication rates remain low. It is important to understand the variations in anatomy and pharmacology in the neonatal and paediatric population and adjust technique and dosing accordingly. With increased availability and use of ultrasound, the safety of epidural insertion under anaesthesia may be further improved. Adequate post-operative management should be in place in order to improve the safety and efficacy of paediatric epidurals and to minimise potential complications. When there are issues post-operatively, although they may be related to the epidural, it is important to consider other more likely causes.
Answers to MCQ questions
- ..
a) False: Termination of the dural sac occurs around the level of S3
b) True: The intercristal line (between the iliac crests) occurs close to L5 in children
c) True: Hypotension is rarely seen in children less than 8 years of age, a product of a lower circulating volume in the lower limbs and splanchnic system, and a relative lack of resting peripheral vascular tone
d) False: Clonidine may cause hypotension when used as an additive in caudal solutions
e) True: There is an almost absent autonomic response to spinal anaesthesia up to 5-6 years of age - ..
a) True: Lack of parental consent is a contraindication to performing neuraxial blockade
b) False: Whilst considered the gold standard in terms of pain management, there is a lack of evidence supporting neuraxial techniques over intravenous analgesia
c) True: Local anaesthetic toxicity can be treated with lipid rescue therapy in order to improve resuscitation outcomes [7]
d) False: Epidural space depth is approximately 1mm/kg of body weight up to 10 years
e) False: Given practical and ethical considerations, it is widely accepted practice to perform paediatric epidurals under anaesthesia - ..
a) True: Complications of paediatric epidural blockade are similar to those seen in adults
b) False: There is no evidence to support a lower risk of complications with either awake or asleep insertion or ultrasound guidance
c) False: There is no evidence to support a lower risk of complications with ultrasound guidance
d) True: The risk of serious long-term complications is approximately 1 in 10 000
e) True: Children under 6 months of age have approximately 4 times the risk of complications from epidural insertion than that seen in older children
REFERENCES and FURTHER READING
- Marhofer P, Ivani G, Suresh S, Melman E, Zaragoza G, Bosenberg A. Everyday regional anesthesia in children. Pediatric Anesthesia 2012; 22: 995-1001.
- Moriarty A. Pediatric epidural analgesia (PEA). Pediatric Anesthesia 2012; 22: 51-55.
- Patel D. Epidural analgesia for children. Continuing Education in Anaesthesia, Critical Care and Pain 2006;6(2): 63-66.
- Marhofer P, Keplinger M, Klug W, Metzelder M. Awake caudals and epidurals should be used more frequently in neonates and infants. Pediatric Anesthesia 2015; 25(1): 93-99.
- Oechsner H, Ehlers M. Central Neuraxial Blocks in Paediatrics. Department of Anaesthesiology, Albany Medical Centre. AMC Anaesthesiology.
- Darcey M. Anaesthetic management of patients with Cerebral Palsy. AAGBI Anaesthesia Tutorial of the Week 2010; 196. https://www.aagbi.org/sites/default/files/196-Anaesthetic-management-of-patients-with-Cerebral- Palsy.pdf
- Liston DE. Childhood asthma and anaesthesia. AAGBI Anaesthesia Tutorial of the Week 2010 (187). https://www.aagbi.org/sites/default/files/187-Childhood-asthma-and-anaesthesia.pdf
- PA Guideline. Good practice in postoperative and procedural pain management. 2nd edition, 2012. Pediatric Anesthesia 2012; 22 (1): 39-73.
- Sawardekar A, Suresh S. Neuraxial blockade in children. Anaesthesia and Intensive Care Medicine 2010; 11:6:
- Suresh S, Wheeler M. Practical paediatric regional anaesthesia. Anesthesiol Clin North America 2002;20(1):83-113.
- Muller M. Pediatric neuraxial anesthesia from caudals, to thoracic epidurals, to awake spinals. Iowa Symposium XIII, May 2013.
- Skinner A. Paediatric pharmacology. AAGBI Anaesthesia Tutorial of the Week 2007. https://www.aagbi.org/sites/default/files/48-Paediatric-pharmacology1.pdf
- Howard R, Carter B, Curry J, Morton N, Rivett K, Rose M, Tyrrell, Walker S, Williams G. Analgesia Review. Pediatric Anesthesia 2008; 18(1): 64-78.
- Shabana A, Shorrab A. Paediatric neuraxial anaesthesia asleep or awake, what is the best for safety? The Internet Journal of Anesthesiology 2008; 21(1).
- Polaner D, Martin L. Quality assurance and improvement: the Pediatric Regional Anaesthesia Network. Pediatric Anesthesia 2012; 22: 115-119.
- Giaufre E, Dalens B, Gombert A. Epidemiology and morbidity of regional anaesthesia in children: a one-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists. Anesth Analg 1996; 83:904-912.
- Willschke H, Marhofer P, Bosenberg A, Johnston S, Wanzel O, Sitzwohl, Kettner S, Kapral S. Epidural catheter placement in children: comparing a novel approach using ultrasound guidance and a standard loss-ofresistance technique. British Journal of Anaesthesia 2007; 97(2): 200-207.
- Sawardekar A, Szczodry D, Suresh S. Neuraxial anaesthesia in paediatrics. Anaesthesia and Intensive Care Medicine 2013; 14(6): 251-254.
- Eccofey C, Lacroix F, Fiaufre E et al. Epidemiology and morbidity of regional anaesthesia in children: a followup on-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists (ADARPEF). Pediatric Anesthesia 2010; 20:1061-1069.
- Campbell NJ. Effective management of the post dural puncture headache. AAGBI Anaesthesia Tutorial of the Week 2010; 181. https://www.aagbi.org/sites/default/files/181-Post-dural-puncture-headache.pdf
- Llewellyn N, Moriarty A. The national paediatric epidural audit. Pediatric Anesthesia 2007; 17: 520-533.
Images in this tutorial have been reproduced with kind permissions from Dr Natasha Woodman, Great Ormond Street Hospital



