Intensive Care Medicine
QUESTIONS
Before continuing, try to answer the following questions regarding heparin induced thrombocytopaenia (HIT). The answers can be found at the end of the article, along with an explanation.
- Which of the following statements is correct:
- In cases of HIT, it is recommended to transfuse platelets once below 20,000 × 109/L.
- Patients with HIT should be anticoagulated.
- HIT should only be treated if the patient tests positive for HIT antibodies.
- Patients with HIT are allergic to heparin.
- In a patient with HITT (which includes thrombosis), why should you reverse any existing
warfarin therapy? - Which of the following is not a direct thrombin inhibitor?
- Fondaparinux
- Lepirudin
- Argatroban
- Bivalirudin
- Danaparoid
INTRODUCTION
Part 1 of this tutorial discussed the aetiology, pathophysiology, and diagnosis of heparin induced thrombocytopaenia. In this part of the tutorial, we will discuss the treatment options for HIT and some of the alternative anticoagulants that can be used.
Heparin induced thrombocytopaenia (type II) is a life-threatening syndrome that may occur after exposure to unfractionated or (in rare cases) low-molecular-weight heparin. HIT is usually suspected after an incidental finding of thrombocytopaenia following administration of heparin, where there is no clear alternative cause for the fall in platelet count. Detection of the thrombocytopaenia and accurate diagnosis (or strong clinical suspicion), are key aspects in the management of HIT. Once the diagnosis has been committed to, treatment must be immediately initiated to combat some of the potentially catastrophic effects of the disorder. The principal goal of managing heparin induced thrombocytopaenia is to reduce the risk of thrombosis by decreasing further platelet activation and thrombin generation.
There are seven key aspects of treating hit:
- Discontinue heparin.
- Continue to monitor platelet counts.
- Avoid giving warfarin until platelet counts recover.
- Test for HIT antibodies (if not already tested for).
- Evaluate for the presence of deep vein thrombosis.
- Platelet transfusions are ONLY given to treat bleeding, NOT the thrombocytopaenia.
- Anticoagulate with a non-heparin regimen.
DISCONTINUE ALL HEPARIN
In most cases, patients with HIT have recently been exposed to a heparin source: heparin flushes, intraoperative anticoagulation, subcutateous injection for thrombophrophylaxis, or haemodialysis.
Whatever the situation, once your suspicion of HIT has been adequately confirmed; ensure all sources of heparin are discontinued, and notify all caregivers. Interestingly, a retrospective analysis2 demonstrated that timely discontinuation of heparin was no more effective in reducing morbidity and mortality than late heparin cessation. This demonstrates that, while ceasing heparin is a vital part of management, multimodal therapy is important in ultimately decreasing the morbidity and mortality. The thrombocytopaenia associated with HIT may continue to worsen, even after discontinuing heparin administration. Recovery of the platelet count is a good sign that HIT has regressed and is responding to therapeutic measures.
HIT is usually suspected and investigated after an incidental platelet count reveals thrombocytopaenia. Until the nadir of the patient’s platelet count has been established, and there is a clear rise in the platelet count, the patient’s platelet levels should be evaluated daily (when resources allow). In severe cases of thrombocytopaenia, haemoglobin levels should also be monitored for signs of subclinical bleeding.
WARFARIN AND LOW MOLECULAR WEIGHT HEPARIN (LMWH)
LOW MOLECULAR WEIGHT HEPARIN
Low molecular weight heparin is not an appropriate alternative for anticoagulation in HIT. There is an in-vivo cross-reactivity between UFH and LWMH in approximately 50% of cases.
WARFARIN
Warfarin monotherapy in active heparin induced thrombocytopaenia is contraindicated, based on multiple reports of warfarin induced skin necrosis and venous gangrene in the limbs. Affected patients typically have a supratherapeutic INR (typically above 4.0). The risk of thrombosis in HIT patients started on warfarin is 38-76% within the first month despite complete discontinuation of heparin. For this reason it is recommended that warfarin not be started until adequate resolution of the patient’s thrombocytopaenia has occurred (preferably, platelet counts above 150,000 × 109/L).
In some cases, HIT is diagnosed after warfarin therapy has already been initiated. In this situation, it is recommended to reverse warfarin anticoagulation with vitamin K. This reversal reduces the risk of warfarin necrosis, and minimizes the risk of direct thrombin inhibitor (DTI) underdosing, as warfarin prolongs the activated partial thromboplastin time (aPTT), which is used to monitor DTI levels, as well as the PT.
REVERSAL OF HEPARIN
The reversal of vitamin K antagonists (warfarin), should be conducted by administering vitamin K 5-10 mg orally or iv. Evaluation of the International Normalised Ratio (INR) and aPTT should be done roughly 6 hours later to evaluate the effectiveness of the vitamin K dosing. The does can be repeated, if necessary.
HIT ANTIBODIES
In a patient with clinically suspected HIT (where the pre-test probability is high), more definitive confirmation by testing for the presence of HIT antibodies helps to guide treatment and prevents missing an alternative diagnosis. Specifics on the types of testing available are detailed in part 1 of this tutorial.
DEEP-VEIN THROMBOSES
Anticoagulation is required for at least 2 to 3 months after the resolution of HIT (see below) to prevent and treat thrombosis. Thrombosis in HIT is one of the more serious complications, and as a result requires vigilance in monitoring and prompt treatment if detected. In patients with HIT who develop thrombosis, there is an associated mortality of approximately 20–30%, and an equal percentage will become permanently disabled by amputation, stroke or other causes.
Thromboembolic complications can be venous, arterial (or both) and include:
- Deep venous thrombosis (DVT)
- Pulmonary embolism (PE)
- Myocardial infarction (MI)
- Thrombosis and ischemia of limbs via arterial occlusion.
The type and site of thrombosis varies depending on the patient and some of their inherent risk factors. DVTs and PEs are most common in postoperative patients. While the incidence of deep vein thrombosis in orthopaedic patients who received heparin for thromboprophylaxis was 17.8%, those with HIT have a DVT incidence that approaches 89%. Even after discontinuation of heparin, patients with HIT have a 38-76% risk of developing thrombotic complications in the first month.
Therefore, an essential component to treatment of the patient with HIT is vigilance in monitoring for the development or existence of thrombi, and optimizing treatment to prevent further occurrence.
PLATELET TRANSFUSIONS
Even though patients with HIT can be severely thrombocytopaenic, the transfusion of platelets in an attempt to avoid bleeding is generally unnecessary. Even when severe throbocytopenia occurs in HIT, petechiae and other mucocutaneous bleeding that would typically be found in patients with non-HIT thrombocytopaenia are rare. Paradoxically, platelet transfusions have been linked with thrombotic events in some reports. When the diagnosis of HIT is uncertain and there is a high bleeding risk (as judged by the clinician), or if significant bleeding occurs, platelet transfusions may be appropriate.
ALTERNATIVE ANTICOAGULATION
As mentioned above, the thrombotic aspect of HIT is a serious comorbidity that should be prevented, monitored, and treated if discovered. The pathophysiology of HIT involves extensive platelet activation, and thrombocytopaenia secondary to platelet clumping/consumption. Hence, once the diagnosis of HIT is committed to, and heparin is discontinued, alternative thromboprophylaxis should be initiated.
Until it is safe to restart heparin or warfarin, a range of newer and developing anticoagulants may be utilized for thromboprophylaxis.
INITIATION OF ALTERNATIVE ANTICOAGULATION
Non heparin alternate anticoagulants should be given for at least 14 days to treat and prevent thrombosis. Preferred alternatives are lepirudin or argatroban.
- Special monitoring tests are required for these medications.
- Doses should be reduced in cases of renal and hepatic insufficiency.
- Once the platelet count rises above 150,000 × 109/L, oral warfarin may be initiated with careful overlapping with a nonheparin anticoagulant for at least 5 days.
- If a life saving procedure like coronary artery bypass graft (CABG), percutaneous coronary intervention (PCI) or haemodialysis is required during acute HIT, the alternative, nonheparin anticoagulant of choice will depend upon the resources at your facility (availability of medication and ability to monitor levels of anticoagulation).
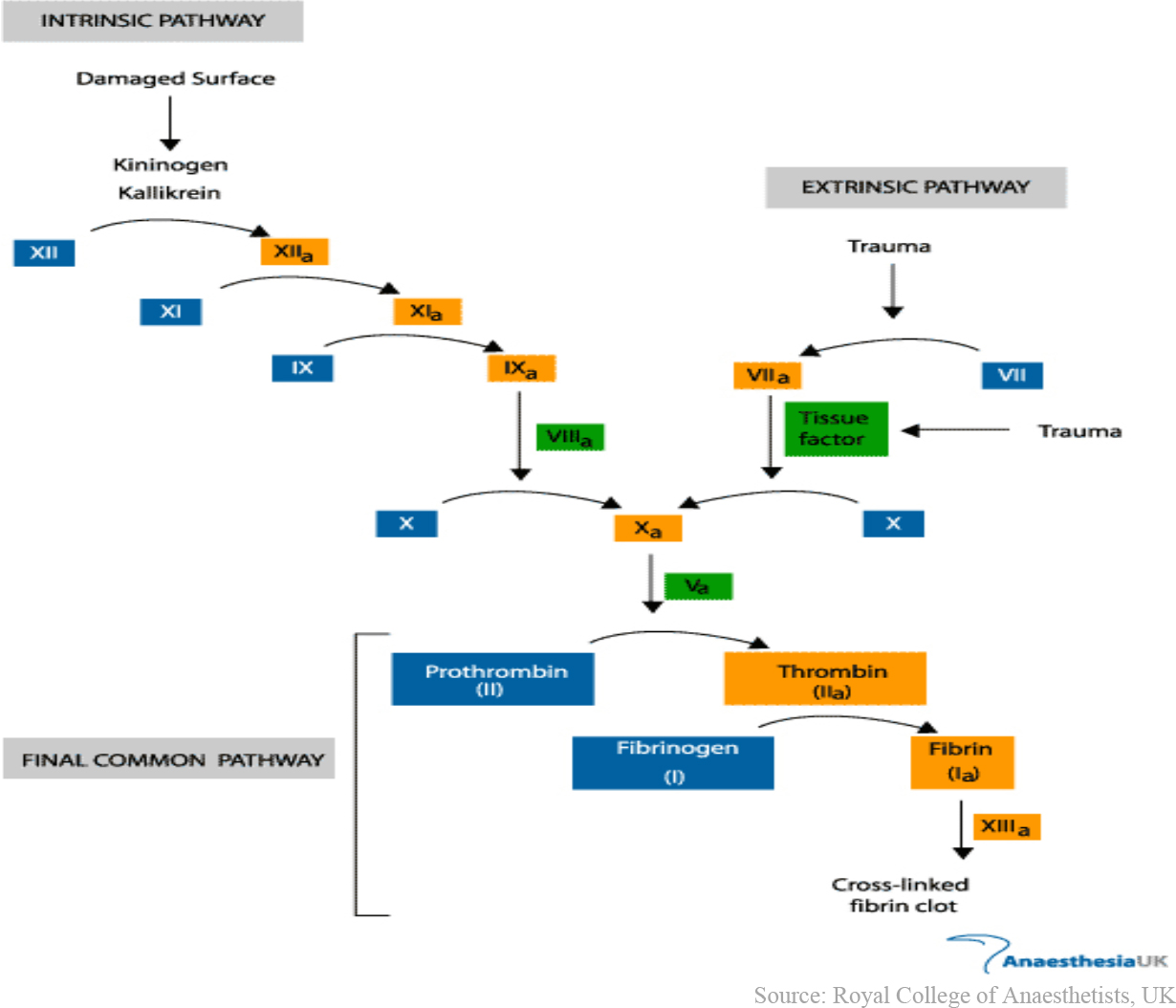
Figure 1: Schematic diagram of the clotting cascade.
DIRECT THROMBIN INHIBITORS
Direct thrombin inhibitors work in the final common pathway of the clotting cascade (see figure 1). They directly bind and inactivate thrombin (also known as factor IIa). This prevents the conversion of fibrinogen to fibrin (a key component to the production of a hemostatic plug or clot).
Direct thrombin inhibitors have short half-lives, show no cross-reactivity to heparin, and unlike heparin, do not require antithrombin to function.
Lepirudin
Lepirudin is recombinant hirudin (originally produced by leeches), which directly inhibits thrombin by irreversibly binding to it. In patients with HIT, studies have demonstrated that anticoagulation with lepirudin significantly lowered mortality, new thromboembolism, and limb amputation.
Of note, lepirudin is renally cleared, and caution should be taken in those with renal dysfunction. Antibodies to lepirudin develop in 30% of patients after initial exposure and in 70% of patients after repeated exposure. Because fatal anaphylaxis has been reported upon repeat administration of lepirudin, patients should not be treated with this agent more than once.
Argatroban
This is an arginine-based synthetic anticoagulant, which directly inhibits thrombin and reversibly binds the catalytic site of thrombin. Of note, Argatroban is hepatically cleared, and caution should be taken in patients with liver dysfunction. In these patients, it is recommended to reduce the dose by 75%.
Bivalirudin
Bivalirudin is another synthetic thrombin inhibitor that has been approved for percutaneous coronary intervention in patients who have, or are at risk for, heparin-inducedthrombocytopaenia (table 2). Because of its short half-life, bivalirudin shows promise as a potential anticoagulant in cardiac bypass surgery, but its use in treating heparin-induced thrombocytopaenia has not been studied in clinical trials.
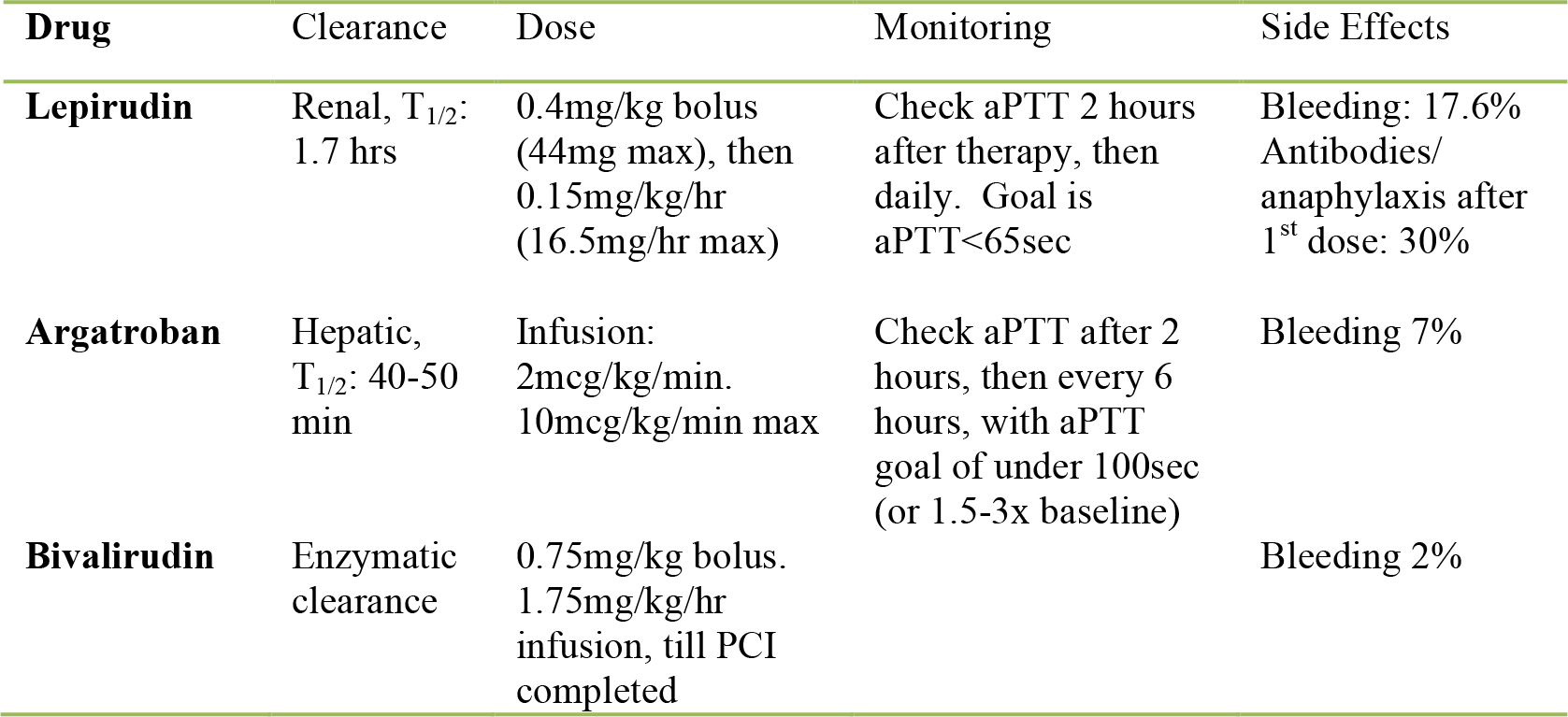
Table 1: Direct thrombin inhibitors; dosing and characteristics
FACTOR Xa INHIBITORS
Danaparoid
Danaparoid is a mixture of three glycosaminoglycans (heparin sulphate, dermatan sulphate and chondroitin sulphate) which, via antithrombin, inhibits anti-factor Xa activity. Recent studies in HIT associated thrombosis showed a 90% treatment success rate with danaparoid. Danaparoid has a long anti-factor Xa half-life of roughly 25 hours. Danaparoid can be given intravenously (see table) or subcutaneously if the intravenous route is not practicable. In this case it can be given as 1250U subcutaneously followed by 2000U twice a day.
The dose should be adjusted to maintain plasma anti-Xa levels within 0.5–0.8 U/mL (this requires facilities that have the ability to rapidly test factor Xa levels). If surgery or other invasive procedures are planned, danaparoid should be stopped until the anti-Xa level is less than 0.4.
Fondaparinux
Fondaparinux is a synthetic heparin pentasaccharide that does not cross-react with HIT antibodies, and is a favourable candidate for anticoagulation in patients with HIT. It has a long half-life of roughly 17 hours, and is recommended as prophylaxis for post-operative thrombosis by daily subcutaneous injection of 2.5mg. Despite its theoretical potential, there are few publications supporting the use of fondaparinux in HIT and other thrombocytopenic situations. Further studies are required before a definitive recommendation can be made on its use in HIT.
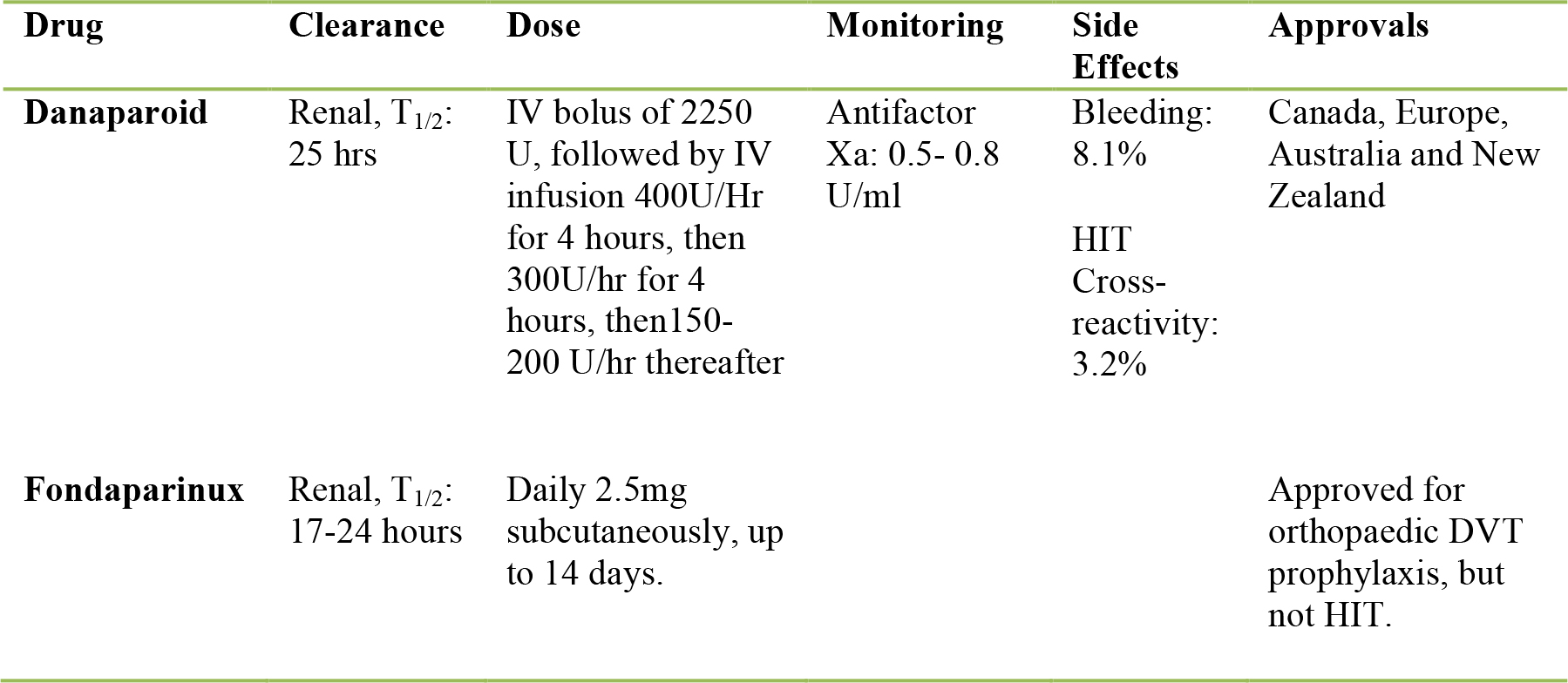
Table 2: Factor Xa inhibitors; dosing and characteristics
ALTERNATIVE ANTICOAGULATION IN LATER STAGES OF HIT
Subacute HIT
At this stage, platelets have generally recovered and are stable at levels of 150,000×109/L or above, but the patient is still HIT antibody positive.
- Avoid reintroduction of heparin for at least 100 days to minimize the increased risk of thrombosis.
- Alternative anticoagulation may be utilized in emergency situations (CABG, PCI, dialysis)
Previous HIT, but normal platelet count, and no detectable HIT antibodies
- Short-term exposure to heparin may be safe, but should be utilized only in emergency situations.
- Alternative anticoagulants may be used if there is a concern of HIT recurrence.
- Bivalirudin may be considered as drug of choice for PCI.
SUMMARY
- If HIT is confirmed or seriously suspected: stop all heparin.
- Follow platelet trends
- Evaluate and monitor for arterial and/or venous thrombosis
- Commence alternative anticoagulation
ANSWERS TO QUESTIONS
1a: FALSE. Platelet transfusion should be reserved for patients with documented bleeding, as further platelet loads are associated with thrombosis in HIT.
1b: TRUE. Because of the incidence and morbidity/mortality of HIT associated thrombosis, prophylaxis should be initiated after discontinuation of heparin.
1c: FALSE. Because of the sensitivity of the HIT antibody test, it serves as an excellent test to rule out HIT. Likewise, the availability of a HIT antibody test is not essential for diagnosing HIT. A diagnosis of HIT can be made on clinical grounds if an antibody test is not available.
1d: FALSE. HIT is not an allergic condition. While HIT is an antibody mediated condition, it does not involve systemic histamine release or anaphylactic/anaphylactoid characteristics.
2: When utilizing warfarin in HIT patients, a severe reduction in protein C can develop, resulting in a failure to control thrombin generation. As a result, thrombosis in HIT patients started on warfarin can reach 38-76% within the first month, despite complete discontinuation of heparin.
3: a and e. Fondaparinux and danaparoid are NOT direct thrombin inhibitors but anti-factor Xa agents.
REFERENCES and FURTHER READING
- Warkentin TE, Greinacher A. Heparin-induced thrombocytopaenia: recognition, treatment, and prevention: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004;126: Suppl:311S-337S. [Erratum, Chest 2005;127:416.]
- Wallis DE, Workman DL, Lewis BE, Steen L, Pifarre R, Moran JF.Failure of early heparin cessation as treatment for heparin-‐induced throbocytopenia. Am J Med 1999, 106:629-‐635
- Gowthami M. Arepally, M.D., and Thomas L. Ortel, M.D., Ph.D. Heparin-Induced Thrombocytopaenia. N Engl J Med 2006; 355:809-817
- Keeling D, Davidson S, Watson H. The management of heparin-induced thrombocytopaenia: On behalf of the Haemostasis and Thrombosis Task Force of the British Committee for Standards in Haematology. Brit Soc Haem 2006; 133: 259–269



