General Topics
KEY POINTS
- Cancer is a leading cause of death worldwide
- Anaesthetic agents and techniques might have long-term consequences on recurrence rates and mortality in patients undergoing cancer surgery.
- Laboratory in vitro findings have shown the effect of numerous anaesthetic agents on cancer cell lines; however, these results have not been easily translated into a clinical setting.
- Despite hope from early retrospective studies, the use of regional anaesthesia in cancer surgery has not been associated with any significant difference in either recurrence or survival in several randomised controlled trials.
- Laboratory and retrospective trials indicate that a propofol-based intravenous anaesthesia technique may confer a benefit over a volatile anaesthetic approach in cancer outcomes.
- At present, there is insufficient evidence to support the reduction of cancer recurrence with any specific anaesthetic agent or technique.
INTRODUCTION
Cancer is a leading cause of death worldwide, accounting for more than 10 million deaths (approximately 1 in 6 overall deaths worldwide), according to the World Health Organisation. Despite the significant developments in cancer therapy, tumour recurrence and metastasis continue to occur and account for a large proportion of cancer deaths. More than 80% of patients with cancer undergo either tumour resection or palliative surgery.1 Advances in healthcare and technology coupled with a growth in societal expectations translate to an increasing number of patients undergoing cancer operations. There is an expanding body of evidence that analgesic, anaesthetic, and other perioperative interventions mayaffect rates of recurrence, return to intended oncological treatment (RIOT) after surgery, and overall survival in cancer patients.
ANAESTHESIA AND CANCER SURGERY
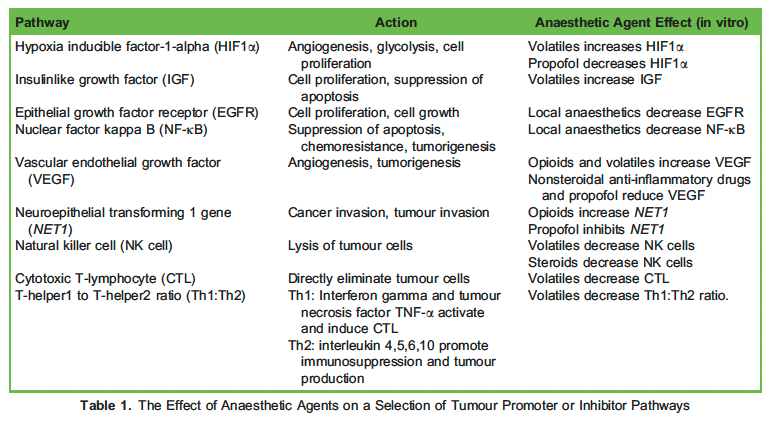
The perioperative period is characterised by physiological stress, which can affect tumour cell survival. Surgical stress response, inflammatory response, effects of anaesthetic techniques and anaesthetic pharmacological agents are all factors that contribute to a state of relative immunosuppression perioperatively. The immunosuppressed patient often has high levels of catecholamines, growth factors and prostaglandins, which can stimulate metastatic transition of cancer cells. Local tissue injury during surgery incites inflammation, promoting the release of cytokines such as interleukin-6 and prostaglandin E2 that typically inhibit the activity of natural killer cells that are usually prominent in the detection and destruction of circulating cancer cells during the surgical period.2 Multiple perioperative factors can lead to a state of reduced perfusion and local hypoxia. Hypoxia leads to the upregulation of hypoxia-inducible factor-1-α (HIF1α) and vascular endothelial growth factor. HIF1α encourages tissue repair and cell proliferation in injured cells but can inadvertently influence metastasis of cancer cells. Vascularendothelial growth factor promotes angiogenesis and lymphatic dilatation allowing cancer cells to spread via vascular and lymphatic systems.1,3 In vitro studies indicate that anaesthetic agents can alter levels and expression of numerous cancer pathways, summarised in Table 1.
SUMMARY OF CURRENT EVIDENCE
Total Intravenous Anaesthesia versus Volatile Anaesthesia
Recent research suggests that a volatile anaesthesia technique may have negative effects on patients with cancer, while a total intravenous anaesthesia (TIVA) technique may be beneficial. Inhalational agents increase tumour growth factors such as HIF1α and insulin like growth factor,which promote tumour cell growth, invasion and migration. In contrast, propofol appears to decrease HIF1α and displays antioxidant and anti-inflammatory properties, making TIVA preferable for the maintenance of anaesthesia for cancer operations.1,4 Unfortunately, clinical outcome data on propofol-based TIVA versus volatile anaesthesia is limited to retrospective cohort studies. To date there has been no prospective randomised controlled trial (RCT) comparing TIVA and volatile anaesthesia in cancer outcomes; however, there are some in progress (VAPOR-C, GA-CARES, TIVA/TCI- BC; ClinicalTrials.gov Identifiers: NCT04316013, NCT03034096, NCT02839668), which may hopefully yield definitive results.
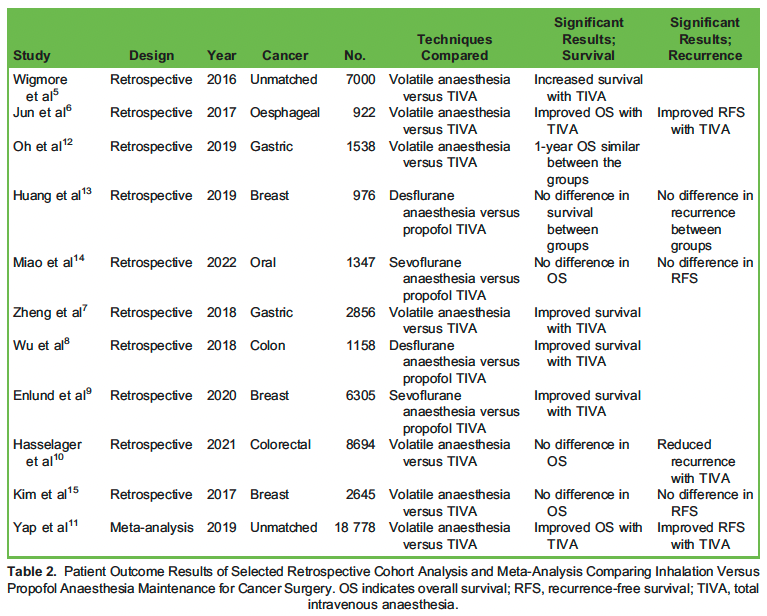
The current evidence investigating cancer recurrence and survival rates is summarised in Table 2. A landmark retrospective study carried out by Wigmore et al5 in 2016 included more than 7000 patients and compared TIVA and volatile anaesthesia. It showed an improved 5-year survival with a TIVA-based anaesthetic compared to a volatile technique (22.8% versus 15.6% P < .001).5 Several other observational studies have shown reduced mortality rates with a propofol TIVA technique.6–9 Additionally, a number of observational studies show a reduction in recurrence rates when TIVA is used compared to a volatile anaesthetic technique.6,10 A meta-analysis by Yap et al11 compared the effect of a propofol intravenous anaesthetic and volatile anaesthesia on cancer outcomes. Ten studies were analysed; 6 (n = 7866) showing a reduction in disease recurrence when TIVA was used compared to a volatile technique (pooled HR, 0.78; P <.01). Eight studies (n = 18 778) demonstrated a reduction in mortalitywith a TIVA technique (pooled HR, 0.76; P < .01).
However, several retrospective studies have been published that fail to show a difference in recurrence or mortality rates in these 2 groups.12–15. A retrospective cohort study by Huang et al13 of breast cancer surgery patients found no overall difference in disease-free survival or recurrence rates when a propofol TIVA technique was used compared to a desflurane anaesthetic technique (P = .449 and P = .454, respectively). A similar retrospective cohort study by Kim et al15 among breast cancer patients also found no significant difference in recurrence or mortality between TIVA and a volatile anaesthetic technique.
There are encouraging trends in current observational research that should motivate future prospective research to evaluate potential benefits for cancer recurrence prevention and long-term survival with propofol-based TIVA techniques in cancer surgery.
Regional Anaesthesia
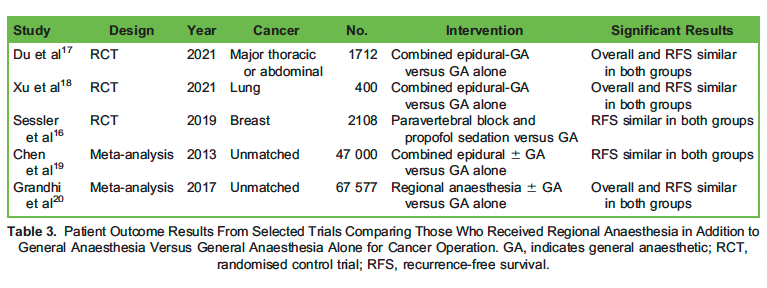
There have been conflicting trials examining the effect of regional anaesthesia on cancer outcomes over the last 20 years.3 Regional anaesthesia has several theoretical benefits on cancer recurrence. These include control of pain and potential for minimising opioid consumption,which is associated with immunosuppression, reduction of stress response to surgery and the direct immunomodulatory effect of local anaesthetics.1 Several randomised controlled trials do not show any benefit of regional anaesthesia in terms of cancer recurrence or survival (Table 3). A large RCT by Sessler et al16 examined the effect of regional anaesthesia in breast cancer patients. More than 2100 patients were randomly assigned to receive either regional anaesthesia (paravertebral block and propofol sedation) or volatile anaesthesia with opioid-based analgesia. The RCT failed to show any significant difference between the 2 groups in terms of cancer recurrence.16 A further RCT by Du et al17analysed patients undergoing major abdominal or thoracic cancer surgery, failing to show an improvement in cancer recurrence rates or survival in patients who received a combined epidural–general anaesthesia technique compared to general anaesthesia alone. Xu et al18 also showed similar findings in an RCT of lung cancer patients undergoing thoracoscopic surgery.
Lidocaine (Lignocaine)
Local anaesthetics may alter cancer outcomes as a direct result of their immunomodulatory effect of downregulation of epithelial growth factor receptor, interleukin-1, tumour necrosis factor alpha and nuclear factor kappa B. In addition, their opioid- sparing effect and sympathetic block (when used as part of a regional technique) can reduce the stress response to surgery and attenuate the associated immunosuppression.1 The literature is unclear, with conflicting evidence regarding overall survival and recurrence-free survival. Lidocaine reduces cell migration and cancer viability in laboratory studies. Retrospective cohort studies suggest that intraoperative use of intravenous lidocaine is associated with improved cancer outcomes; however, further studies are needed in this area to add clarity.2
Blood Transfusion
Cancer surgery can cause significant blood loss requiring blood transfusion. Laboratory studies have proven that transfusions cause inflammation and immunosuppression that can subsequently encourage cancer recurrence. Clinical studies suggest that perioperative blood transfusion in cancer surgery can have a detrimental effect on outcomes. A Cochrane review from 2006 including 12 000 patients concluded that blood transfusion was associated with risk of recurrence of colorectal cancer (odds ratio 1.42; 95% confidence interval, 1.20 to 1.67).21Other meta-analyses have confirmed significant recurrence rates associated with bladder, gastric and prostate cancer.22–24 Put in perspective, the blood transfusion data are currently limited to meta-analyses involving retrospective studies where associations but not necessarily causation can be implied.1,3
Opioids
Opioid analgesia is commonly used for perioperative pain relief in cancer patients but recent trends and advances in enhanced recovery programmes have fostered a move to a multimodal approach to pain management. Opioids can theoretically influence tumour growth and metastasis through a number of mechanisms. They have immunosuppressive qualities, including reducing activity of natural killer cells and neutrophils, which can accelerate cancer progression. Opioids can directly influence cancer cell growth in vitro through their effect on the mu opioid receptor, which is overexpressed in a wide range of cancer cells, including breast, colon and lung. Clinical studies in this area are limited and cannot offer conclusive evidence of detrimental effects of perioperative opioid use in cancer patients. Despite the paucity of evidence in this area, it is clear that equilibrium must be sought between the competing effects of opioid-related stress response reduction and cancer progression. On balance, it seems sensible to practice an opioid-sparing technique where possible.1–4
Alpha-2 Agonists
Alpha-2 adrenoceptors are known to impair release of noradrenaline to attenuate the sympathetic stress response; however, there are few data to date on effect of alpha-2 agonists involving immune system modulation and cancer recurrence. Alpha-2 receptor agonists (eg,dexmedetomidine and clonidine) have been increasingly used for sedation and opioid-sparing analgesia in recent years. Laboratory studies have mostly indicated increased tumour growth and metastasis with alpha-2 agonists and mostly detrimental effect in cancer cell lines. However, the potential tumour-potentiating consequences of alpha-2 agonists must be balanced against their secondary opioid- and volatile anaesthesia–sparing effect in cancer surgery. The clinical literature on the topic is limited to small retrospective studies that have failed to showa consistent benefit or detrimental effect of alpha-2-agonists in cancer outcomes.1–4
Steroids
Steroids have known immunosuppressive properties. It follows that they may affect immune system ability to detect and destroy circulating tumour cells and potentially increase tumour recurrence risk. Steroids also have anti-inflammatory properties that may attenuate surgical stressresponse and reduce the associated negative effects.1 Steroids are commonly used in the perioperative setting as antiemetics, anti-inflammatories and analgesic adjuncts. Studies investigating the clinical outcomes of the use of steroids in the perioperative setting are limited to retrospective cohort studies and offer mixed results, with most failing to show a difference in terms of survival or recurrence.3,4 Higher quality randomised trials are needed to ascertain the benefits or risks of steroid use in perioperative cancer patients. Current evidence involving steroid use in cancer surgery is insufficient to recommend a change in current clinical practice in this field.
Nonsteroidal Anti-Inflammatory Drugs
The inflammatory response to surgery is linked to cancer recurrence, so theoretically anti-inflammatory agents may alter this effect. Potential beneficial effects of nonsteroidal anti-inflammatory drugs include an opioid-sparing effect, altered expression of epithelial growth factor receptor and nuclear factor kappa B, and inhibition of prostaglandin-induced immunosuppression. Despite the theoretical advantages of nonsteroidalanti-inflammatory drugs, clinical perioperative studies after cancer surgery are inconclusive.1–4
Return to Oncological Treatment
RIOT is a new endpoint in onco-surgical research. Reduced surgical recovery times allow for earlier RIOT, thereby increasing the likelihood ofrecurrence-free survival.25 Postoperative complications commonly linked to anaesthetic technique may reduce or prolong patient recovery with implications for recommencement of oncologic therapy. Hayden et al26 conducted an RCT investigating the effect of intraperitoneal infiltration of ropivacaine on readiness for postoperative oncologic therapy. They found a reduction in time to RIOT with infiltration of ropivacaine compared to the control. This was the first RCT in onco-anaesthesia using RIOT as a primary endpoint. Although the use of RIOT as an endpoint in onco-anaesthesia research is to date limited, it may become a potentially useful endpoint in the future.25
CONCLUSION
Surgery is the commonest treatment modality with curative intent for cancer patients. The idea that anaesthetic technique can have long-lasting implications beyond the perioperative period is an exciting prospect, yet anaesthesia is one of many factors influencing recovery from cancer and its potential for recurrence. Adding regional techniques to general anaesthesia does not appear to improve prognosis for cancer progression and recurrence, while the evolution of a number of current RCTs may confirm that propofol TIVA techniques for anaesthetic maintenance may offer an advantage to volatile anaesthesia. The potentially harmful effects of blood transfusion require confirmation with prospective research. Influence of individual drugs coadministered during anaesthesia will take longer to clarify because the individual effects of any drug on inflammatory and immunefunction must be weighed against ability to supress stress response and reduce need for other potentially harmful drugs.
REFERENCES
- Buddeberg BS, Seeberger Anesthesia and oncology: friend or foe? Front Oncol. 2022;12:802210.
- Sherwin A, Wall T, Buggy DJ. Anaesthesia and cancer recurrence: UpToDate; 2022. Accessed June 28, 2022. https:// uptodate.com/contents/anesthesia-and-cancer-recurrence#H2875951382
- Wall T, Sherwin A, Ma D, Buggy DJ. Influence of perioperative anaesthetic and analgesic interventions on oncological outcomes: a narrative review. Br J Anaesth. 2019;123(2):135-150.
- Evans MT, Wigmore T, Kelliher The impact of anaesthetic technique upon outcome in oncological surgery. BJA Educ. 2019;19(1):14-20.
- Wigmore TJ, Mohammed K, Jhanji Long-term survival for patients undergoing volatile versus IV anesthesia for cancer surgery: a retrospective analysis. Anesthesiology. 2016;124(1):69-79.
- Jun IJ, Jo JY, Kim JI, et al. Impact of anesthetic agents on overall and recurrence-free survival in patients undergoing esophageal cancer surgery: a retrospective observational study. Sci Rep. 2017;7(1):14020.
- Zheng X, Wang Y, Dong L, et Effects of propofol-based total intravenous anesthesia on gastric cancer: a retrospective study. Onco Targets Ther. 2018;11:1141-1148.
- Wu ZF, Lee MS, Wong CS, et al. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in colon cancer surgery. Anesthesiology. 2018;129(5):932-941.
- Enlund M, Berglund A, Ahlstrand R, et al. Survival after primary breast cancer surgery following propofol or sevoflurane generalanesthesia—a retrospective, multicenter, database analysis of 6305 Swedish Acta Anaesthesiol Scand. 2020;64(8):1048-1054.
- Hasselager RP, Hallas J, Gogenur I. Inhalation or total intravenous anaesthesia and recurrence after colorectal cancer surgery: a propensity score matched Danish registry-based study. Br J Anaesth. 2021;126(5):921-930.
- Yap A, Lopez-Olivo MA, Dubowitz J, Hiller J, Riedel B, Global Onco-Anesthesia Research Collaboration Anesthetic technique and cancer outcomes: a meta-analysis of total intravenous versus volatile anesthesia. Can J Anaesth. 2019;66(5):546-561.
- Oh TK, Kim HH, Jeon YT. Retrospective analysis of 1-year mortality after gastric cancer surgery: total intravenous anesthesia versus volatile anesthesia. Acta Anaesthesiol Scand. 2019;63(9):1169-1177.
- Huang YH, Lee MS, Lou YS, et al. Propofol-based total intravenous anesthesia did not improve survival compared to desflurane anesthesia in breast cancer surgery. PLoS One. 2019;14(11):e0224728.
- Miao L, Lv X, Huang C, Li P, Sun Y, Jiang H. Long-term oncological outcomes after oral cancer surgery using propofol- based total intravenous anesthesia versus sevoflurane-based inhalation anesthesia: a retrospective cohort study. PLoS One. 2022;17(5):e0268473.
- Kim MH, Kim DW, Kim JH, Lee KY, Park S, Yoo Does the type of anesthesia really affect the recurrence-free survival after breast cancer surgery? Oncotarget. 2017;8(52):90477-90487.
- Sessler DI, Pei L, Huang Y, et al. Recurrence of breast cancer after regional or general anaesthesia: a randomised controlled trial. Lancet. 2019;394(10211):1807-1815.
- Du YT, Li YW, Zhao BJ, et Long-term survival after combined epidural-general anesthesia or general anesthesia alone: follow-up of a randomized trial. Anesthesiology. 2021;135(2):233-245.
- Xu ZZ, Li HJ, Li MH, et al. Epidural anesthesia-analgesia and recurrence-free survival after lung cancer surgery: a randomized trial. Anesthesiology. 2021;135(3):419-432.
- Chen WK, Miao The effect of anesthetic technique on survival in human cancers: a meta-analysis of retrospective and prospective studies. PLoS One. 2013;8(2):e56540.
- Grandhi RK, Lee S, Abd-Elsayed The relationship between regional anesthesia and cancer: a metaanalysis. Ochsner J. 2017;17(4):345-361.
- Amato A, Pescatori Perioperative blood transfusions for the recurrence of colorectal cancer. Cochrane Database Syst Rev. 2006(1):CD005033.
- Cata JP, Lasala J, Pratt G, Feng L, Shah Association between perioperative blood transfusions and clinical outcomes in patients undergoing bladder cancer surgery: a systematic review and meta-analysis study. J Blood Transfus. 2016;2016:9876394.
- Agnes A, Lirosi MC, Panunzi S, Santocchi P, Persiani R, D’Ugo D. The prognostic role of perioperative allogeneic blood transfusions in gastric cancer patients undergoing curative resection: A systematic review and meta-analysis of non- randomized, adjusted studies. Eur J Surg Oncol. 2018;44(4):404-419.
- Li SL, Ye Y, Yuan Association between allogeneic or autologous blood transfusion and survival in patients after radical prostatectomy: a systematic review and meta-analysis. PLoS One. 2017;12(1):e0171081.
- Finnerty DT, Buggy Return to intended oncologic therapy: a potentially valuable endpoint for perioperative research in cancer patients? Br J Anaesth. 2020;124(5):508-510.
- Hayden JM, Oras J, Block L, et al. Intraperitoneal ropivacaine reduces time interval to initiation of chemotherapy after surgery foradvanced ovarian cancer: randomised controlled double-blind pilot Br J Anaesth. 2020;124(5):562-570.



