General Topics
KEY POINTS AND OBJECTIVES
- Intraoperative neurophysiological monitoring (IOM) can potentially reduce the risk of injury to neural structures during surgery.
- There are different IOM modalities, each monitoring a different neural pathway.
- A good understanding of the interactions between IOM and anaesthesia allows the anaesthetist to tailor the anaesthetic regimen to optimize IOM signals.
- IOM signal alternations may signify ongoing neurological injury, which may be related to surgical, anaesthetic, and physiological factors.
- Early coordinated team response to IOM signal change is the key to successful management of neural injury.
INTRODUCTION
Intraoperative neurophysiological monitoring (IOM) is evolving toward a standard of care to minimise the risk of injury to neural pathways during neurosurgical procedures. Its importance lies not only in diagnosing injury but also in creating a window of opportunity to salvage neural tissues at risk before damage becomes irreversible. To maximise its value, it is essential for the operative team to have a basic understanding of the principles of neuromonitoring and for the anaesthetist to understand how it can be affected by anaesthesia. Numerous modalities exist in the IOM armamentarium, and they can be classified into 2 types: 1) detection of spontaneous activity, such as electroencephalography (EEG) and electromyography (EMG), and 2) measurement of evoked electrical response of a specific neural pathway after an active stimulation. Examples of the latter type include somatosensory evoked potential (SSEP), motor evoked potential (MEP), and brainstem auditory evoked potential (BAEP). These modalities can be used in isolation or in combination depending on the structures that are at risk during surgery. In this tutorial, we will discuss the basic principles of the most commonly used IOM modalities, the factors that affect IOM signals, and clinical approaches to address signal changes.
WHY SHOULD WE CONSIDER INTRAOPERATIVE NEUROMONITORING?
Intraoperative neuromonitoring can be useful in the perioperative period for patients at high risk of developing neurological complications from surgery. The mechanisms of injury include direct mechanical disruption from surgical manoeuvres, thermal injury from surgical coagulation, pressure injury from patient positioning, and ischaemia due to local or global hypoperfusion.

Table 1. Characteristic of a Good IOM Modality
Because of anatomical and physiological variations in vascular collateral, anomalies, dominance, neurovascular autoregulation, and reserve, the ischaemic tolerance varies widely between individuals. An example can be seen amongst patients who exhibit variable shunt dependency during carotid endarterectomies. Furthermore, the nature of the surgery may expose the patients to different degrees of direct neuronal injury or vascular compromise. Systemic factors such as anaemia, hypotension, and hypoxia can contribute to secondary injuries.
In the face of these broad differences in neural susceptibilities, IOM is an attractive option to ascertain the well-being of the nervous system in real time. Table 1 summarises ideal characteristics of an IOM modality. False-negative results reflect a genuine failure of IOM, potentially leading to unnoticed neural damages and devastating consequences. False-positive results could trigger unnecessary interventions that might carry their own risks.
Examples of common IOM modality usage in various surgeries are shown in Table 2.
In the early days of its development, IOM was occasionally performed by anaesthetists. It is now more commonly done by dedicated neurophysiologists, who together with the anaesthetists, surgeons, and operative staff, form a multidisciplinary intraoperative team. Success of IOM relies on close collaboration within the team: 1) clear common understanding of the location, nature, and timing of possible neurological injury so that suitable monitoring modalities can be chosen; 2) minimisation of anaesthetic and technical interference on signal attainment; 3) early recognition and interpretation of IOM signal change; 4) clear communication of IOM signal changes to the intraoperative team; and 5) timely and coordinated multidisciplinary team response to signal change.
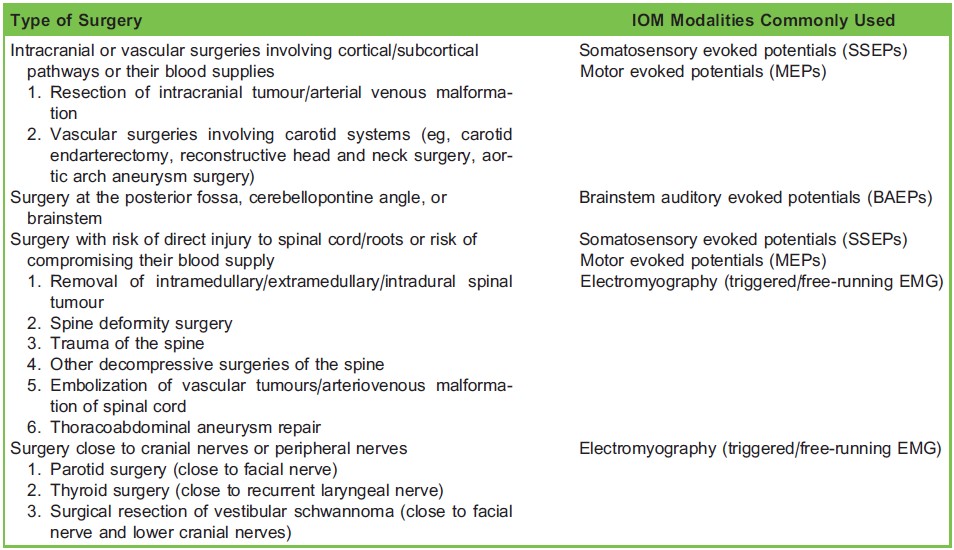
Table 2. Examples of IOM Modalities Used in Various Surgical Procedures
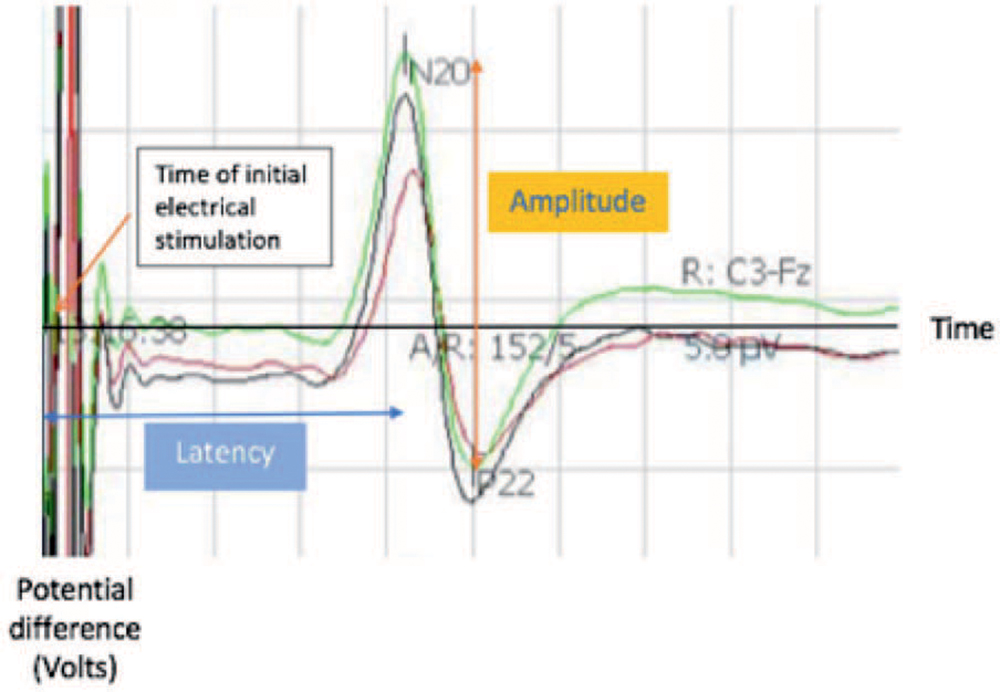
Figure 1. Examples of an evoked response. Latency (unit: seconds) and amplitude (unit: volts) are important parameters in describing evoked potentials.
EVOKED POTENTIALS
In evoked potential monitoring, the neural pathway is interrogated by applying electrical stimulation at one end of the pathway, with the response being measured at the other end in the form of a potential difference (volts). Evoked responses are expected to come in within a certain time (latency) from the stimulation and exhibit a certain strength (amplitude) and pattern (Figure 1). These features are important in describing the evoked response. Any major decrease in amplitude, increase in latency, or change in pattern suggest possible neural tissue compromise.
Somatosensory Evoked Potential (SSEP)
SSEP is one of the most commonly performed modalities, and it monitors the ascending sensory pathways through transcutaneous electrical stimulation of a peripheral nerve.1 The electrical signal travels to the dorsal nerve root after stimulation and ascends the spinal cord via the dorsal columns. It synapses at the cervicomedullary junction and decussates, ascending to the thalamus and finally the sensory cortex (Figure 2). Upper limb SSEPs are usually generated from the median or ulnar nerve stimulation at the wrist, while lower limb SSEPs are from the posterior tibial nerve stimulation at the ankle or popliteal fossa. The resulting signals will be recorded at several stations along the pathway and averaged across multiple stimulations. Standard electrode locations include Erb’s point, popliteal fossa (‘‘peripheral SSEPs’’), cervical spine (‘‘subcortical SSEPs’’), and the somatosensory cortex (‘‘cortical SSEPs’’; Figure 3). It can be performed continuously throughout the procedure and give a close-to-real-time monitoring of the sensory pathway.
SSEP has long been used to monitor dorsal column function and its integrity during scoliosis surgery, and it is increasingly used in major spine and aortic surgery when the chance of spinal cord injury is significant. It has an additional advantage of detecting malposition of the upper limbs causing brachial plexus injury, which can manifest as a signal loss across Erb’s point. It is also a more commonly chosen modality for monitoring middle cerebral artery and anterior cerebral artery perfusion during carotid endarterectomies and neurosurgical procedures.
Motor Evoked Potential (MEP)
MEP is used to monitor the integrity of the descending corticospinal and corticobulbar tracts. It starts with stimulation at the motor cortex, and the electrical activity descends along the motor tracts and synapses onto the anterior horn of the spinal cord or cranial nerve nuclei.2 It then travels along a peripheral or cranial nerve, across the neuromuscular junction to produce muscle contraction (Figure 4). MEP can be elicited either by transcranial electrical stimulation (TES) through the scalp or direct electrical stimulation (DES) on the brain. TES is used for functional monitoring of the motor system, while DES provides a higher spatial resolution of the motor cortex and subcortical tracts and is used for intraoperative motor mapping.
At the spinal level, evoked responses can be measured in the epidural or intrathecal spaces as D (direct) waves or I (indirect) waves. D waves are negative peaks that are assumed to be elicited from direct corticospinal fibre activations, with subsequent peaks termed as I waves. However, compound muscle action potentials (CMAPs) at the effector muscles are more commonly
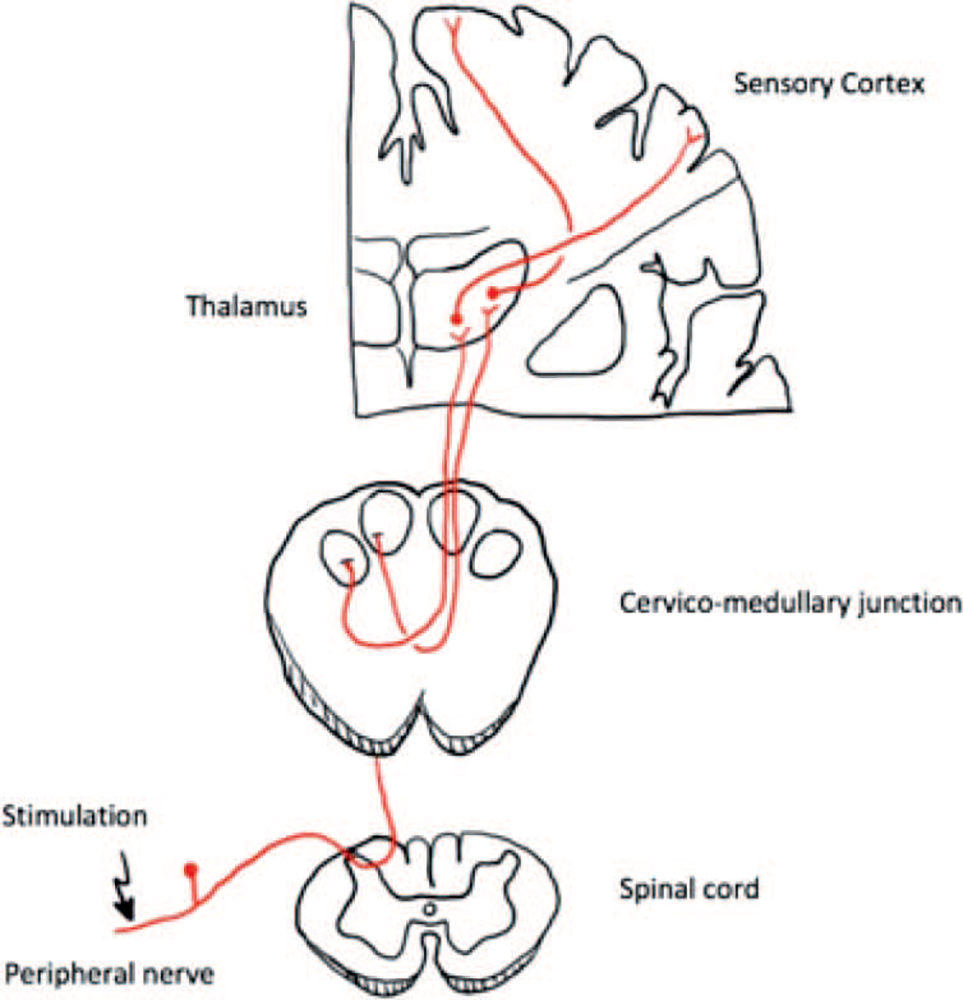
Figure 2. Ascending sensory pathway monitored by SSEP.
measured because of their sensitivity, specificity, and minimal invasiveness. These are the composite electrical activities within the effector muscles, resulting from synchronous activation of the corresponding motor neurons within a nerve bundle. Recording electrodes are usually placed at selected muscles innervated by the brain regions, nerve roots, or cranial nerves that are deemed at risk. Commonly monitored sites in the extremities include the thenar muscles, tibialis anterior, and abductor
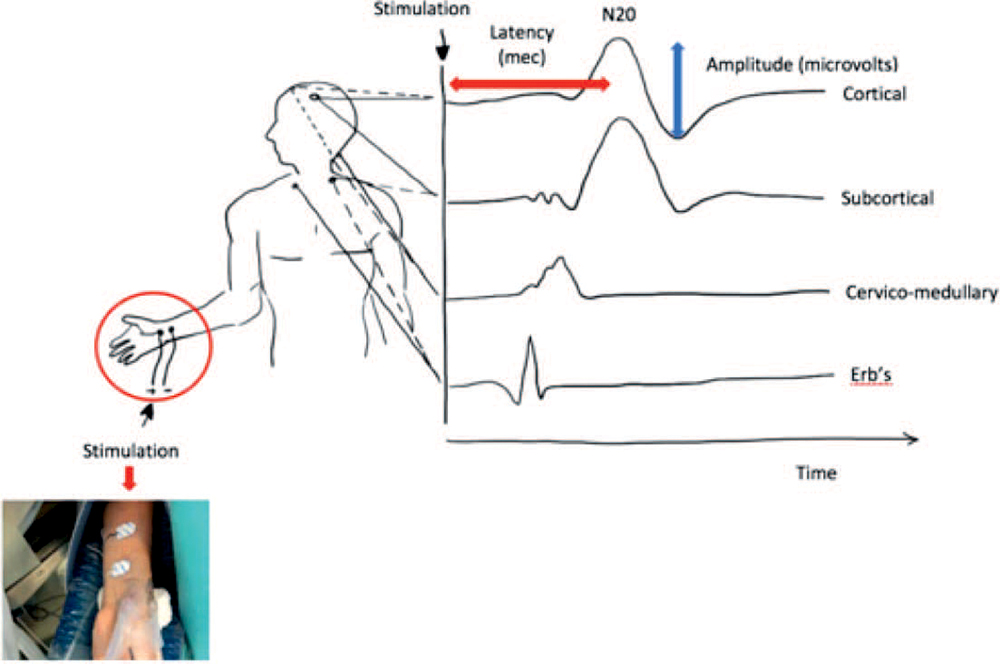
Figure 3. Placement of SSEP electrodes for upper limb (median nerve at the wrist [circled]) and the resulting waveforms. The nomenclature that is used for the peaks and valleys of SSEP waveforms uses N and P, respectively, to designate the polarity of the recorded signal (negative is up and positive is down) and an integer to denote the nominal poststimulus latency of the signal in normal adults. The prominent cortical SSEP signal for the upper limb is the N20 (upward deflection at around 20 ms) and for the lower limb is the N45 peak.
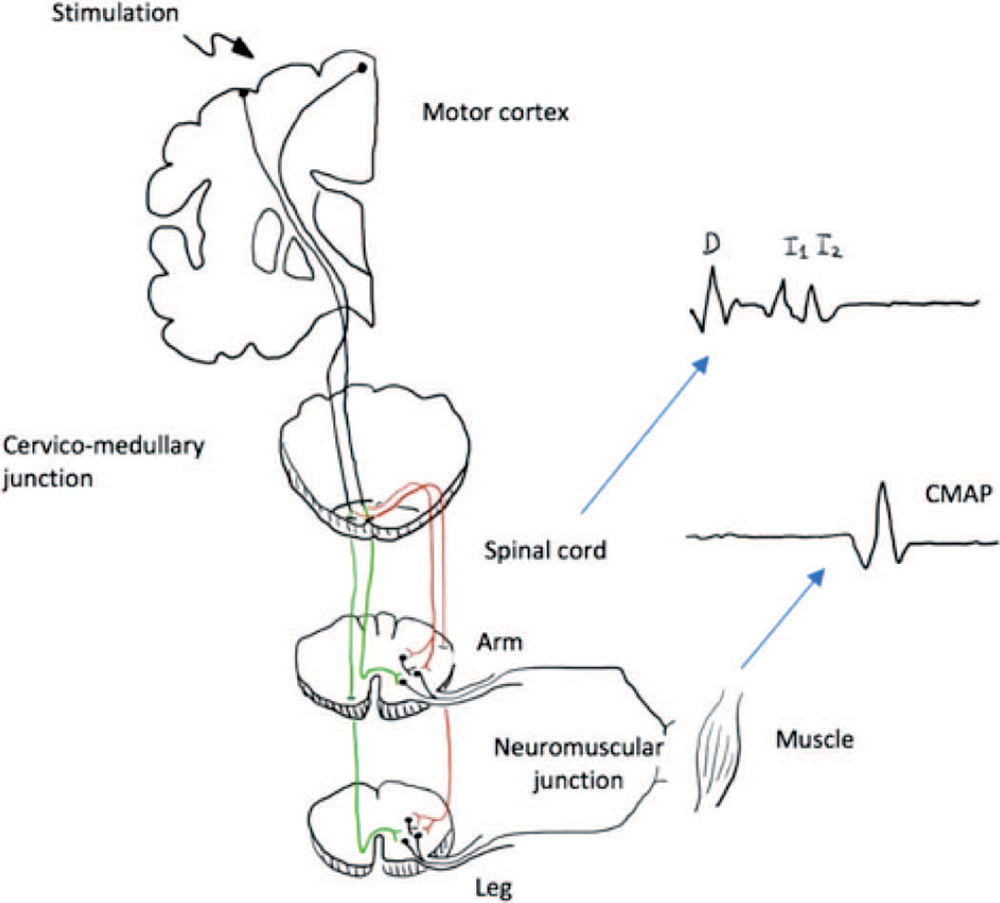
Figure 4. Motor pathway monitored by MEP with the waveform recorded from cord level (D and I waves) and muscle level (CMAP). Stimulation of the motor cortex generates evoked potentials that propagate through the brain and spinal cord to cause a muscle contraction. The response typically is recorded near the muscle as a compound muscle action potential (CMAP). The response can also be recorded over the spinal column as a D wave followed by a series of I waves (high-frequency repetitive discharges from the corticospinal fibers). At the spinal cord level, most motor fibres reside in the lateral corticospinal tract (red pathway) after decussation at the brainstem level. The anterior corticospinal tract (green pathway) contains fewer motor tracts.
hallucis. One disadvantage of MEPs is that they are performed only at intervals and therefore pose a potential risk for delayed detection of neural injury.
Motor deficits are a significant morbidity after surgery, and MEP can be used if the motor pathway is at risk. Motor neurons have a high metabolic demand, and at the spinal level they are nourished only by a single anterior spinal artery with variable reinforcement from spinomedullary arteries. MEP is used in major spine and intracranial surgeries to detect mechanical or ischaemic injury along the tract or at the end motor nerve. It is often used in conjunction with SSEP for spinal cord monitoring, as together they grossly cover the spinal cord anteroposteriorly. It also has a role in aortic surgery, in which the anterior spinal circulation may be compromised due to spinomedullary artery insufficiencies (eg, occlusion of the artery of Adamkiewcz).
Brainstem Auditory Evoked Potential (BAEP)
BAEP is used to monitor the vestibulocochlear nerve (cranial nerve VIII) and brainstem function.3 Acoustic stimulus is delivered by a device at the ear canal. Electrical signals, generated by the cochlear, travel along the vestibulocochlear nerve into the nucleus and various brainstem structures. The response is recorded by an electrode placed at the mastoid or ear lobe.
BAEP is used during posterior fossa surgeries (e.g. excision of vestibular schwannoma, microvascular decompression for hemifacial spasm or trigeminal neuralgia) to assess brainstem and hearing function, which can be compromised as a result of cerebellar retraction, vascular injury (e.g. injury to the posterior inferior cerebellar artery), or direct mechanical or thermal injury to the vestibulocochlear nerve and brainstem.
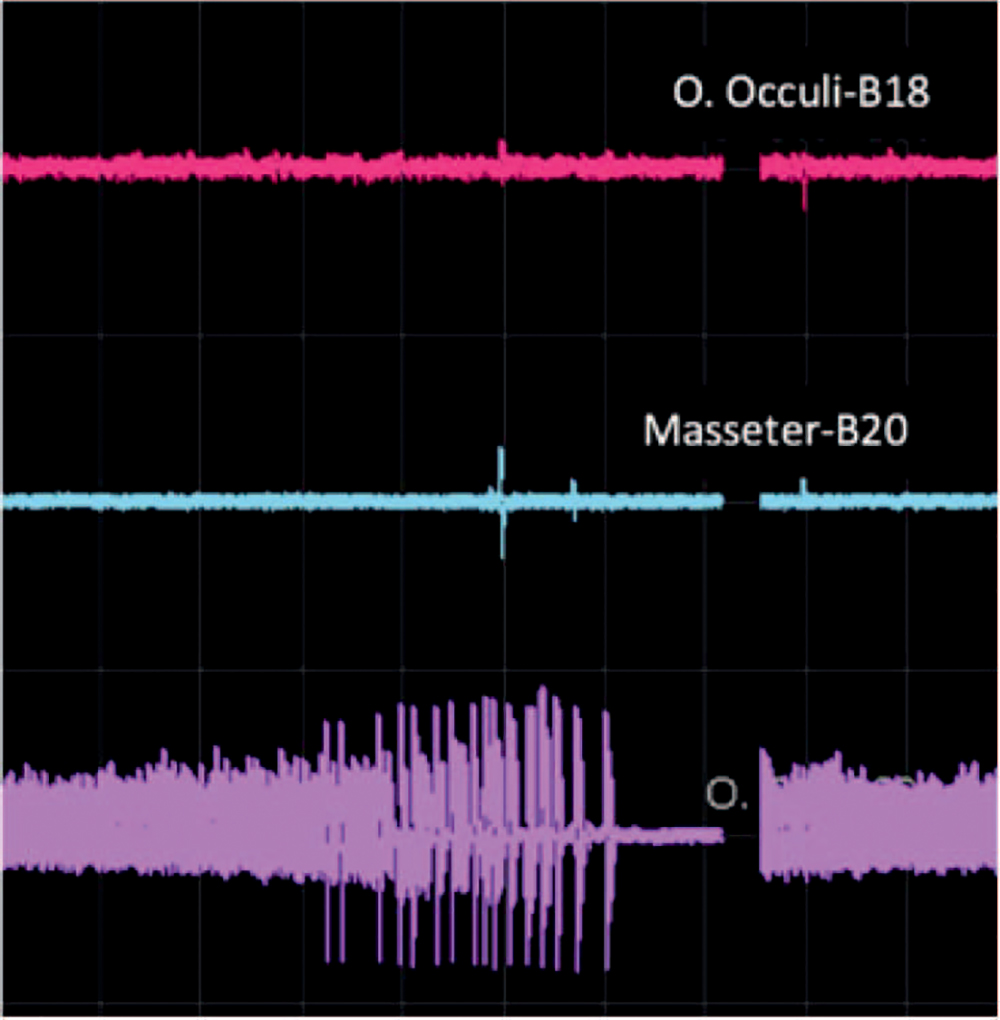
Figure 5. (Top 2 tracings) Normal spontaneous EMG activity in patient under general anaesthesia. (Bottom tracing) Neurotonic discharge during nerve irritation.
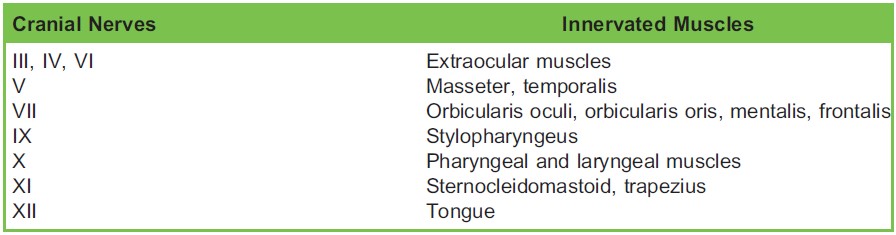
Table 3. Target Muscle CMAP Measured in Different Cranial Nerve EMG Monitoring
ELECTROMYOGRAPHY
Intraoperative EMG monitors motor cranial nerves, spinal or peripheral nerves at risk by looking at either the spontaneous or evoked CMAPs of the corresponding effector muscles.4
Spontaneous EMG records free-running electrical activities from muscles, which is normally of low frequency and amplitude. When the nerve is stretched or irritated, high-frequency neurotonic discharges will be produced, which alerts the surgical team of potential motor nerve damage (Figure 5). The absence of such discharges (i.e. a ‘‘negative response’’) suggests either an intact nerve or total denervation disrupting all nerve conduction.
Triggered EMG, on the other hand, is produced by direct, intentional stimulation of peripheral or cranial nerves, with the resulting CMAP recorded from the corresponding innervated muscles (Table 3). A positive CMAP from an appropriately applied stimulation will help to identify nerve structures when visual differentiation is difficult (e.g. due to tumour invasion) and to confirm integrity of the nerve distal to the stimulation. A common application of triggered EMG is facial nerve mapping and monitoring during vestibular schwannoma or parotid tumour resection.
ANAESTHESIA AND IOM
There are special anaesthetic considerations with IOM.
1. Many anaesthetic agents produce dose-dependent suppression of evoked potentials, making it essential for the anaesthetists to choose a regimen compatible with the proposed IOM modalities.

Table 4. Effect of Anaesthetic Agents on SSEP and MEP
2. Anaesthetists should provide a stable physiological and anaesthetic milieu to facilitate meaningful interpretation of signal changes and accurate surgical guidance.
3. When true signal change is confirmed, the anaesthetists should be aware of potential nonsurgical causes of neural injury and be ready to act promptly to ameliorate further insults and secondary injuries.
4. Using IOM may pose risks to the patient. Anaesthetists should exercise appropriate precautions (see the section ‘‘Complications with IOM and Precautions ’’ below).
Anaesthetic Agents
Most anaesthetic agents alter neural function by producing dose-dependent depression in synaptic activities. In general, inhalational agents have greater effects on the evoked potentials than intravenous anaesthetic agents (Table 4). Choice and dose of these hypnotic agents should be tailored to the modalities used.
Volatile Agents
Inhalational agents cause a dose-dependent decrease in amplitude and increase in latency of evoked responses. For SSEP, such effects are more pronounced on cortical responses than on subcortical and peripheral responses. Adequate SSEPs can usually be recorded at <1 minimum alveolar concentration. However, for patients with baseline neurologic impairment or neuropathy from systemic disease, lower levels of inhalational agents may abolish potentials and make monitoring impossible. MEP is affected by even lower concentrations of volatile agents as they suppress lower motor neuron excitability more profoundly. BAEPs and EMG are resistant to inhalational agents.
Intravenous Agents
SSEP signals are unaffected by high barbiturate doses, whereas MEP signals are sensitive to barbiturates. Benzodiazepines used at premedication doses would not suppress SSEPs and MEPs. Ketamine can enhance SSEP and MEP responses, which may be beneficial for monitoring when used to supplement anaesthesia or analgesia. Etomidate can augment the amplitude of cortical SSEP recordings without affecting the peripheral evoked potentials or subcortical responses. There are limited studies on the effect of dexmedetomidine on evoked potentials. SSEPs and MEPs are shown to be recordable at low clinical doses, but MEP recordings may be lost at higher doses.
Known for its favourable pharmacokinetic profile and titratability, propofol suppresses SSEPs and MEPs in a dose-dependent manner but to a much smaller extent, allowing adequate measurements at commonly used clinical doses. It is considered the anaesthetic agent of choice to facilitate IOM, especially when MEP monitoring is used.
All anaesthetic agents take time to equilibrate after dose adjustment. Therefore, it is undesirable to alter the dose unnecessarily during critical steps of surgery, as that may confound the interpretation of IOM signals.
Opioids
Evoked potentials can be recorded even at very high doses of opioids, so infusions of short-acting potent opioids such as remifentanil are commonly used to reduce the anaesthetic requirement during surgical procedures involving evoked potentials monitoring.
Muscle Relaxants
MEP and EMG monitoring are affected by muscle relaxants, which inhibit electrical activities across the neuromuscular junctions, affecting signal recording as muscle contraction can no longer be generated upon neural stimulation.
If MEP or EMG recording is necessary shortly after endotracheal intubation, one can administer a cautious dose of short-acting muscle relaxant before laryngoscopy and allow its effect to wear off before monitoring commences. If necessary, reversal agents can also be given prior to monitoring. Intubation without neuromuscular blockers is possible with adequate depth of anaesthesia from propofol and remifentanil infusion.
Occasionally, a mild degree of neuromuscular blockade is desirable in some surgeries that also monitor MEP or EMG. For instance, paraspinal muscle relaxation often facilitates the initial dissection during scoliosis corrective surgery. If a muscle relaxant has to be used intraoperatively while monitoring MEP or EMG, the level of paralysis should be carefully controlled and monitored, ideally guided by a nerve stimulator with an accelerometer rather than visual inspection. Neuromuscular blockade should be maintained at a stable and mild level throughout the monitoring period with a neuromuscular blocker infusion, aiming for not more than two twitches on train-of-four stimulation.
With current anaesthetic techniques (e.g. total intravenous anaesthesia with propofol and remifentanil infusion), it is possible to avoid the use of neuromuscular blockers altogether in most cases, providing an anaesthetic environment for more reliable MEP and EMG recording.
Physiological Parameters
Several physiological factors can affect evoked potential monitoring. Inadequate oxygen supply to meet the metabolic demand of neural tissues is the main cause of pathological signal changes seen in the evoked potentials (i.e. increase in latency or decrease in amplitude). Oxygen supply to the brain is dependent on adequate blood oxygen content and cerebral perfusion, which in turn is affected by systemic blood pressure and caliber of the intracranial vessels.
- Reduction in systemic or regional blood pressure can depress cortical evoked potential monitoring. SSEP and MEP latency increase and amplitude drop are associated with a reduction in cerebral blood flow and possible embolic events. Patients with baseline abnormal cerebral or spinal perfusion or impaired autoregulation are at higher risk of ischaemia with fluctuations of blood flow.
- Hyperventilation and the resulting hypocapnia can constrict cerebral vasculature and diminish oxygen delivery. The effect is more significant in patients with compromised vascular anatomy or when compensatory autoregulation is already maximised.
- Anaemia and hypoxaemia reduce the oxygen content of blood. Latency of evoked potentials starts to increase and amplitude decreases when haematocrit falls to 10% to 15%. IOM signal change may occur at a higher haematocrit level if there is concurrent hypoperfusion.
Hypothermia decreases central and peripheral nerve conduction velocities and hence increases SSEP latency. Therefore, the core temperature should be maintained within about 2°C of baseline. Regional hypothermia, for example, by exposing the spinal cord or cerebellopontine angle to cold irrigation, can also affect evoked potential monitoring. Limb cooling (e.g. from cold infusion of fluids) may increase the SSEP stimulation threshold in that extremity and alter the degree of peripheral nerve stimulation.
Patient positioning for surgery can cause vascular or neurological compromise, leading to signal changes. For example, lateral patient position can cause direct brachial plexus compression or vascular insufficiency of the dependent arm. Neck flexion for spinal or posterior fossa surgeries may cause mechanical pressure on the cord, nerves, or important vasculatures, especially if these structures are already predisposed because of underlying pathologies. In patients with a high risk of positional neural injury, it may be desirable to measure evoked potentials before and after patient positioning. If the potentials diminish after adopting the operating position, patient posture should be readjusted before surgery begins. It is also important to note that positional neural injury can lead to signal changes gradually and insidiously; hence, it should remain as a differential diagnosis of intraoperative signal change.
COMPLICATIONS WITH IOM AND PRECAUTIONS
During MEP stimulation, muscles for mastication are activated and can result in bite injury if bite blocks are not meticulously placed.5 Both SSEP and MEP stimulations will result in patient movement, especially when the stimulation current is high. If the patient moves vigorously during MEP acquisition, anaesthetists should inform the neurophysiologists to check if a lower stimulating current can be used. Any patient movement can be deleterious if it occurs during a critical stage of surgery (e.g. application of clips during intracranial aneurysm surgery). Hence, good communication between the anaesthetists, surgeons, and neurophysiologists is imperative. During SSEP acquisition in an unparalysed patient, distal limb movements can also affect blood pressure or pulse oximetry measurement if the arterial line or pulse oximeter are on the same extremity.
Subdermal needle electrodes may be placed in close proximity to vascular access (Figure 6) or even the endotracheal tube (e.g. during CN VII, IX, X, XII monitoring), so communication with the neurophysiologist is desirable when siting and readjusting these electrodes intraoperatively. Local guidelines on safe handling and disposal of sharps should be followed, as the corkscrew and subdermal needle electrodes may be contaminated with the patient’s blood. On removal of electrodes, the sites should be compressed until haemostasis is achieved.
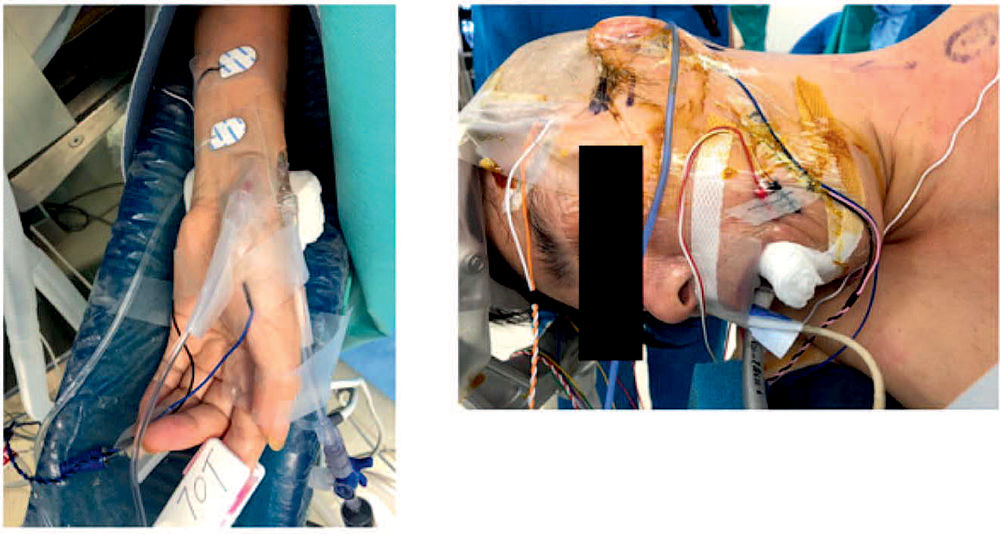
Figure 6. (A) Hypodermic needles used for upper limb MEP and percutaneous electrode for median nerve stimulation (upper limb SSEP) can be in close proximity to arterial lines and peripheral vascular access, and (B) those used for cranial nerve monitoring can be close to the patient’s airway.
MANAGEMENT OF IOM SIGNAL CHANGE
It is important that an optimal and stable physiological and anaesthesia milieu is present during the acquisition of a baseline reading, as this will be referenced for the rest of the procedure. Different criteria of what would constitute a significant signal change exist, in part because of variations in study protocol, anaesthetic regimen, and neuromonitoring setup. A true, sudden total loss of signals should always prompt remedial interventions, while a 50% reduction in amplitude and 10% increase in latency for evoked potentials are considered significant and should be communicated to the whole operating team (anaesthetists, surgeons, and neuromonitoring personnel) such that the cause can be identified and treated promptly to prevent further damage to neural tissues.
When a significant signal change is detected, the persistence of this change should be confirmed, and technical issues (falsepositives) should be excluded. This can involve rerunning the evoked potentials, ensuring both the recording and stimulation parameters are correct, or cross-checking with other modalities for associated changes.
Management varies depending on the likely cause. The list of differential diagnosis for signal change can be narrowed down by considering the pattern of change, the location on tract at which the signal drop occurs, and the laterality of change. Anaesthetic and physiological aetiologies may cause global changes in signals, while single-sided or one-limb signal alteration usually points toward surgical, technical, or positional factors. In the case of SSEP, more than one station along the pathway is usually monitored, and the station at which the signal begins to drop off can help to localise the site and cause of injury.
The temporal relationship between the change in monitoring signals and surgical manoeuvres may suggest the likely cause for the change. Examples of such surgical manipulations include an excessive brain retraction or spinal distraction, prolonged or misplaced vascular clipping or cross-clamping, or localised vasospasm during surgical interventions. If signal change occurs during clipping of an aneurysm or carotid cross-clamping, and the pattern of change is consistent with the interventions, then a surgical cause of ischaemia is very likely to account for the change.
In response to signal change, the surgeons may transiently withhold any manipulations and observe for any spontaneous signal recovery. Irrigation of the surgical field with warm saline helps to promote regional blood flow and remove irritating metabolites that may have axonal blocking properties. Preemptive local application of papaverine and increasing the arterial blood pressure facilitate local perfusion to resist ischaemia. The anaesthetist should promptly optimise perfusion and oxygen supply to the neural tissues by targeting at normal or supra-normal blood pressure, correcting anaemia, and fine-tuning the depth of anaesthesia.
If the evoked potentials do not recover, a joint decision has to be made whether the surgical approach should be modified, such as lessening of spinal distraction, removing the spinal implant, or discontinuing tumour resection. It may also warrant review of a postoperative management plan and physiological targets. In some spine centres, methylprednisolone administration would
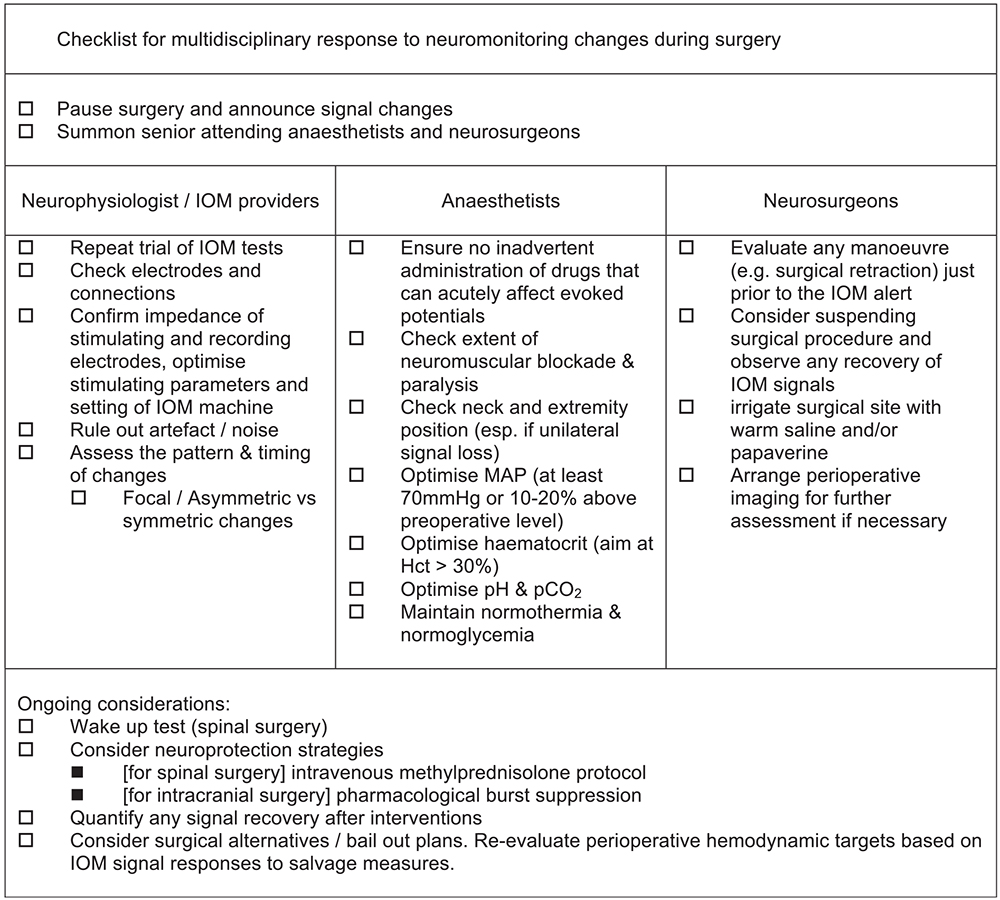
Figure 7. Example of a multidisciplinary checklist to respond to intraoperative IOM signal changes.
be considered if spinal cord injury is suspected. Good communication within the operative team is imperative, and a specially designed checklist has been used to promote coordinated resuscitation effort in such situations (Figure 7).6,7
CONCLUSION
IOM offers an attractive option for real-time monitoring of the nervous system, but successful implementation must be built on a team approach to facilitate IOM and appropriate corrective measures when major signal changes happen. The anaesthetist plays an important role in evaluating the impact of anesthesia and physiology on IOM signals and optimising these factors intraoperatively. There is growing evidence that IOM reduces neurological disability from surgery, but a universal protocol and guideline for its use and interpretation are still lacking. This is in part due to variations in patients’ premorbid neurological status, the type of anaesthesia, the setup of the neuromonitoring, and the nature and approach of surgery. Each institution may develop its own protocol on what and how to monitor and what degree of signal alteration amounts to a significant change criterion, making communication among intraoperative personnel vital.
REFERENCES
- Toleikis JR. Intraoperative monitoring using somatosensory evoked potentials: a position statement by the American Society of Neurophysiological Monitoring. J Clin Monitor Comput. 2005;19:241-258. doi:10.1007/s10877-005-4397-0
- MacDonald DB, Skinner S, Shils J, Yingling C; American Society of Neurophysiological Monitoring. Intraoperative motor evoked potential monitoring: a position statement by the American Society of Neurophysiological Monitoring. Clin Neurophysiol. 2013;124:2291-2316. doi:10.1016/j.clinph.2013.07.025
- Martin WH, Stecker MM. ASNM position statement: intraoperative monitoring of auditory evoked potentials. J Clin Monitor Comput. 2008;22:75-85. doi:10.1007/s10877-007-9108-6
- Leppanen RE. Intraoperative monitoring of segmental spinal nerve root function with free-run and electrically-triggered electromyography and spinal cord function with reflexes and F-responses: a position statement by the American Society of Neurophysiological Monitoring. J Clin Monitor Comput. 2005;19:437-461. doi:10.1007/s10877-005-0086-2
- MacDonald DB. Safety of intraoperative transcranial electrical stimulation motor evoked potential monitoring. J Clin Neurophysiol. 2002;19:416-429.
- Skinner S, Holdefer R, McAuliffe JJ, Sala F. Medical error avoidance in intraoperative neurophysiological monitoring: the communication imperative. J Clin Neurophysiol. 2017;34:477-483. doi:10.1097/WNP.0000000000000419
- Vitale MG, Skaggs DL, Pace GI, et al. Best practices in intraoperative neuromonitoring in spine deformity surgery: development of an intraoperative checklist to optimise response. Spine Deform. 2014;2:333-339. doi:10.1016/j.jspd.2014. 05.003



