Intensive Care Medicine
Introduction
Acute upper haemorrhage (UGIH) is a common medical emergency, with an incidence of up to 150 per 100,000. The incidence is highest in those from the lowest socio-economic groups. Although its incidence is declining, the mortality remains high. A large UK audit of UGIH in 1995 showed that in patients admitted with bleeding as their primary diagnosis the mortality was 11%. In contrast, in patients who had a ‘secondary’ UGIH (i.e. they had an UGIH whilst an in-patient for another reason) mortality was 33%. Most deaths from UGIH occur in the elderly population who have significant medical co-morbidities.
Depending on the site and rapidity of bleeding, patients can present with haematemesis, malaena, haematochezia (passage of blood PR) or syncope. There is a large range of clinical presentation, from minor UGIH that can be managed safely in the community to catastrophic exsanguination. All patients who have suspected UGIH should be assessed in hospital.
Initial Assessment
History
The presenting complaint, past history and medication history are all important in determining the aetiology of the UGIH. For example, a history of persistent vomiting followed by haematemesis in a young patient is strongly suggestive of a Mallory Weiss tear. There are a number of drugs (listed below) that increases a patient’s risk of UGIB. When taking the history it is important to ascertain if the patient has any past history of (or risk factors for) chronic liver disease, as bleeding from oesophageal or gastric varices can be significant and rapid.
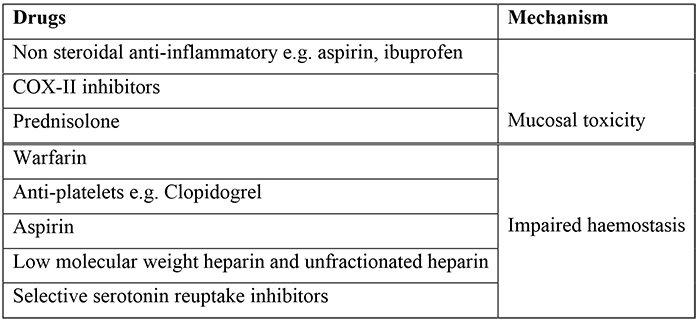
Table 1. Drugs associated with Upper GI haemorrhage
Examination
It is important to carefully assess any patient presenting with UGIH for haemodynamic instability or on-going bleeding. The assessment should include pulse and blood pressure (lying and standing), as well as a rectal examination to look for malaena. In addition, patients should be examined carefully for stigmata of chronic liver disease.
Investigations
At presentation blood should be taken for:
- Full Blood Count
- Urea and Creatinine (an elevated urea:creatinine ratio is a common finding in patients with significant UGIH)
- Liver function tests
- Clotting
- Cross match
Risk Assessment
It is important to identify those patients who are at risk of ongoing bleeding and death. There have been several scoring systems developed to help in this process. The Rockall scoring system is a validated scoring system used for risk categorisation based on simple clinical parameters. Rockall Scores can be calculated both before and after endoscopy, but the post endoscopy Rockall Score provides a more accurate risk assessment. It comprises independent risk factors which have been shown to accurately predict both re-bleeding and mortality (Table 2).
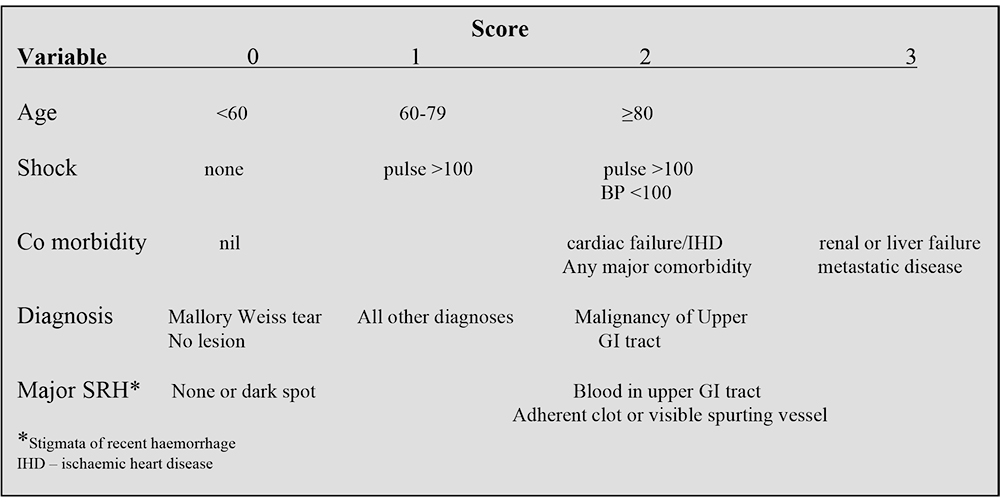
Table 2. Rockall scoring system (post endoscopy) for risk assessment of rebleedingand death in UGIH
With increasing age there is an increased risk of death. Mortality in those aged below 40 is negligible. Mortality increases to 30% in those aged over 90. Patients who have evidence of active bleeding and signs of shock have an 80% risk of death. Those with a non-bleeding visible vessel at endoscopy have a 50% chance of rebleeding.
Patients who have a normal oesophagogastroduodenoscopy, Mallory Weiss tear or an ulcer with a clean base have a very low risk of rebleeding (fresh haematemesis +/- melaena associated with shock 24 hours after the initial event). Low risk patients are defined as those with a post endoscopy score of ≤2. These patients have a 4% risk of re-bleeding and 0.1% mortality.
However, the best risk assessment tool for identifying low risk UGIH was developed in Glasgow by Blatchford et al. The Blatchford score is calculated prior to endoscopy and is based on simple clinical and laboratory parameters (table 3). Its principle use is to identify low risk patients who do not require any intervention (blood transfusion, endoscopic therapy, surgery). Approximately 20% of patients presenting with UGIH have a Blatchford score of zero. Such patients can largely be managed safely in the community, as the mortality in this group is nil.
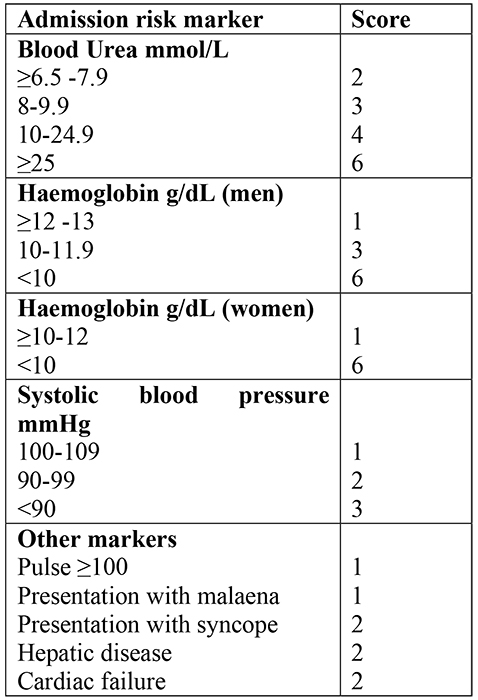
Table 3. Blatchford Score for assessing the severity of UGIH
Causes of upper GI haemorrhage
The cause for upper GI haemorrhage is identified in 80% of cases. The causes are shown in Table 4.
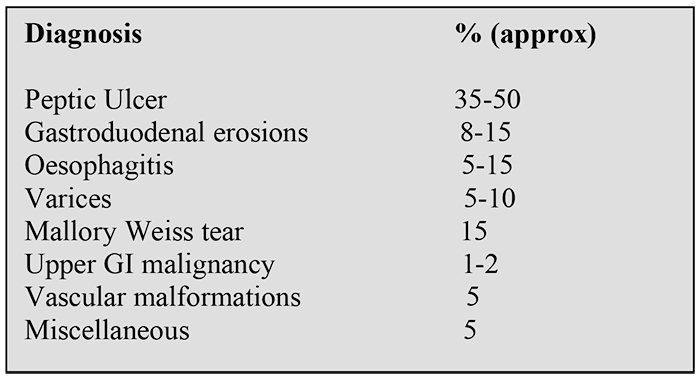
Table 4. Causes of upper gastrointestinal haemorrhage
Non-variceal upper GI haemorrhage: management
Resuscitation
All patients should be managed by the system of Airway, Breathing, Circulation. All patients should receive 100% oxygen (unless contraindicated) and intravenous access obtained. This entails the placement of at least one 18 gauge cannula and, if bleeding is significant, 2 large bore (16 or 14 gauge) cannulae should be sited. Either colloid or crystalloid can be used for volume replacement, aiming for a systolic BP >100mmHg. If, after 2 litres have been given, the patient still has signs of shock this implies that 40% or more of the patients circulating volume has been lost. Transfusion is necessary in if:
- Bleeding is extreme i.e. active haematemesis + shock
Or
- When Hb <10g/L in the presence of an acute bleed
If bleeding is severe patients with UGIH should be managed on a high dependency unit where fluid resuscitation should continue. A catheter should be inserted and hourly urine volumes measured. A central venous catheter should be inserted in those patients with a significant cardiac history to guide fluid management. The patient should be kept fasted until there is haemodynamic stability and an endoscopy performed.
Endoscopy
Once resuscitated, patients with UGIH should have an upper gastrointestinal endoscopy by an experienced endoscopist. In patients who are haemodynamically stable endoscopy can safely be performed in a semi-elective manner, ideally the morning after admission but certainly within the first 24 hours of admission. Not only does this aid diagnosis but it also identifies those patients who are suitable for early discharge.
Only a very small number of patients require endoscopy ‘out of hours’. Therapeutic endoscopy has been shown to improve prognosis in those who present with severe UGIH i.e. haemodynamic instability despite fluid resuscitation. It is these patients who benefit from an ‘out of hours’ service. Endoscopy in these patients should only be undertaken by a skilled endoscopist who has experience in the therapeutic endoscopic management of upper GI haemorrhage. Anaesthetic input is often required to protect the airway from aspiration of blood from the GI tract during endoscopy.
Haemostasis
Ideally all patients who show active bleeding, or who have endoscopic stigmata of recent haemorrhage, should receive endoscopic therapy. In general this comprises:
- Injection of adrenaline – 1:10,000 adrenaline injected into 4 quadrants around the bleeding point, up to a maximum of 16mls. This will achieve haemostasis in 95% of patients; however bleeding recurs in up to 20%.
- Application of heat – by means of a heater probe, Argon plasma coagulation or multipolar probe.
- i. The heater probe has a thermocouple at the tip of the probe and can heat up instantaneously to achieve tissue coagulation.
- ii. The Argon plasma coagulator (APC) is as effective as the heater probe. It uses argon gas to deliver evenly distributed thermal energy to a field of tissue adjacent to the probe. A high voltage spark is delivered at the tip of the probe, which ionizes the argon gas as it is sprayed from the probe tip
- iii. Multipolar probes achieve haemostasis by heating the tissue with electricity that passes through the tip of the probe rapidly from positive and negative electrodes.
- Mechanical clips – Endoscopically applied vascular clips are applied to bleeding vessels, however they can be technically difficult to apply, particularly in awkwardly placed ulcers.
When bleeding is seen it is recommended to apply dual modalities to stop bleeding i.e. adrenaline and thermal coagulation or adrenaline and clips. This is to decrease the risk of re-bleeding from use of adrenaline alone. All patients should be monitored for 4-6 hours post endoscopy. If haemodynamically stable after this period they should be allowed to eat and drink
All patients with ulcer disease should have a H. pylori (CLO) test taken at the time of endoscopy.
Medical management
- Proton Pump Inhibitors
- In patients with UGIH there is increased mucosal fibrinolytic activity, thus impairing haemostasis. Suppressing acid secretion blunts this response and makes any clot formation more stable. Clot lysis has been shown to occur at a pH <6. Proton pump inhibitors (PPI’s) are the only class of drug that consistently increase gastric pH to >6. Omeprazole has been shown to be of benefit in acute UGIH, in terms of incidence of re-bleeding, reduced occurrence of persistent bleeding, decreased need for blood transfusion and decreased duration of hospital stay. Therefore, a PPI is recommended for empirical treatment of UGIB prior to endoscopy. This can either be parenteral or oral. If the bleeding is severe the parenteral route should be considered.
The role of high dose omeprazole (80mg stat followed by and infusion of 8mg/hr for 72hours) is used in those patients who have had successful endoscopic therapy of major ulcer bleeding and remain at high risk of re-bleeding.
- In patients with UGIH there is increased mucosal fibrinolytic activity, thus impairing haemostasis. Suppressing acid secretion blunts this response and makes any clot formation more stable. Clot lysis has been shown to occur at a pH <6. Proton pump inhibitors (PPI’s) are the only class of drug that consistently increase gastric pH to >6. Omeprazole has been shown to be of benefit in acute UGIH, in terms of incidence of re-bleeding, reduced occurrence of persistent bleeding, decreased need for blood transfusion and decreased duration of hospital stay. Therefore, a PPI is recommended for empirical treatment of UGIB prior to endoscopy. This can either be parenteral or oral. If the bleeding is severe the parenteral route should be considered.
- Tranexamic acid
- A meta-analysis has shown that the use of tranexamic acid in UGIH may reduce the need for surgical intervention, although it has no effect on the ulcer re-bleeding rate. Tranexamic acid is not recommended as routine therapy and its role in UGIH requires further evaluation.
Repeat upper GI endoscopy
Repeat endoscopy is performed if:
- There is clinical evidence of re-bleeding, suggested by ongoing melaena or haematemesis, or haemodynamic evidence of blood loss. Most patients who re-bleed should have a repeat endoscopy not only to confirm the presence of blood prior to surgical intervention but also to attempt further endoscopic control of the haemorrhage.
- If there were suboptimal endoscopic views at the initial endoscopy. The reason for suboptimal views is often due to large amounts of blood in the upper gastrointestinal tract following severe UGIH. In such patients it is prudent to repeat the endoscopy 12-24 hours after the initial intervention.
Surgery
Active non variceal UGIH that is uncontrollable by endoscopic therapy needs surgical intervention.
Only one clinical trial has looked at the different surgical procedures for bleeding duodenal ulcers. Re-bleeding was lowest in those having a gastrectomy to include the ulcer with either a Billroth I or Billroth II reconstruction compared to more conservative measures. However, the study suggests that when a bleeding duodenal ulcer is under run with specific ligation of the gastroduodenal and right gastroepiploic arteries the re-bleeding rate is reduced to that of those in the gastrectomy group. Most patients are now treated with this more conservative approach wherever possible.
Gastric ulcers are best excised or treated by partial gastrectomy. There is no clinical evidence that favours either intervention however if there is a suspicion of a malignant ulcer it is best treated with partial gastrectomy.
The role of interventional radiology
There is a small group of patients with major non-variceal UGIH who continue to bleed despite endoscopic intervention who are unfit for operative surgery. Such patients are often the very elderly with major co-morbidities who would not survive major surgery. Such patients should be considered for mesenteric angiography. This allows the bleeding vessel to be identified and embolised with coils. This will often successfully arrest on-going UGIH, but the mortality of this group remains high.
Follow up
In patients with UGIH it is important that a management plan be put in place upon discharge from hospital so that the chances of recurrence of any further bleeding is minimised. The following guidelines should be considered prior to discharge:
- All patients who have peptic ulcer disease and who are H.pylori positive should receive standard eradication therapy.
- Ulcers should be treated with a PPI for an initial period of 6-8 weeks.
- Patients with ulcers associated with NSAID use should stop the use of these drugs.
- If a patient needs to continue taking a NSAID then the least gastro-toxic (ibuprofen) should be used with concomitant administration of a PPI.
- If a patient needs to continue taking an anti-inflammatory, consider a cyclooxygenase II inhibitor.
- If a patient needs long term therapy with aspirin then a PPI prescribed concomitantly reduces the risk of recurrent bleeding.
- All gastric ulcers need repeat endoscopy in 6-8/52 in order to confirm healing and exclude malignancy.
Variceal bleeding: management
Increased portal venous pressure (usually in the context of chronic liver disease) causes the development of a portosystemic collateral circulation, with resultant portosystemic shunts. Such shunts manifest clinically as oesophageal and gastric varices. Patients with varices have a 30% lifetime risk of variceal bleeding. The mortality for the first presentation of variceal bleeding is 50%. If a patient has bled once they have a 70% risk of recurrent bleeding. The mortality from subsequent bleeds is dependent on the severity of the underlying chronic liver disease (Table 5).
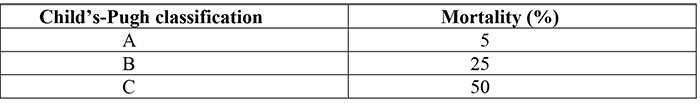
Table 5. Mortality from variceal bleeding in patients with differing severities of underlying chronic liver disease (Child-Pugh classification).
Resuscitation
The general principles of resuscitation are the same as non variceal UGIH (see above). In addition, the following need to be considered:
- Bilateral large bore intravenous access should be established in all cases of suspected variceal bleeding. Initial fluid resuscitation should begin with colloid or crystalloid (aim to avoid sodium chloride). Bleeding can be torrential. In such patients it is imperative to act quickly if bleeding varices are suspected.
- Cross match 6 units and transfuse as clinically required. The patient may require O-negative blood
- Ideally, central venous access should be established to guide fluid therapy
- Correct thrombocytopenia by platelet infusion if platelets <50x 109/L
- Correct clotting with;
- i. Vitamin K iv 10mg
- ii. FFP
- iii.Cryoprecipitate (only if fibrinogen <75mg/dL)
- iv. and/or prothrombin complex concentrate (Octaplex)
- Broad spectrum antibiotics (e.g. 3rd generation cephalosporins) should be given as they have been shown to reduce in hospital mortality by 20%
- Protect the airway by elective endotracheal intubation of any patient where there is:
- i. Severe uncontrollable variceal bleeding
- ii.Severe encephalopathy
- iii.Inability to maintain oxygen saturations >90%
- iv. Aspiration (pneumonia)
Endoscopy
Endoscopy should be performed at the earliest possible time when the patient is haemodynamically stable. There are a number of methods of treating varices. The choice of therapy depends on the site of the varices.
In patients with bleeding oesophageal varices band ligation is the method of choice as it has the greatest chance of controlling bleeding. However, in patients who are actively bleeding views of the lower oesophagus can be so poor that it is impossible to place the bands endoscopically. In such patients, injection sclerotherapy may be easier to apply. If haemostasis is not achieved by these therapies balloon tamponade using the Sengstaken-Blakemore tube is used (see below). This tends to be a temporary measure prior to either TIPSS or surgery.
In patients with gastric varices, if varices are present at or around the gastro-oesophageal junction they are treated as oesophageal varices and either banded or sclerosed. However, if gastric varices are present in isolation initial therapy should be by the injection of cyanoacrylate (tissue glue). If this fails to control bleeding balloon tamponade is used as a temporary measure with a view to performing a TIPSS procedure.
Balloon Tamponade
Balloon tamponade is only used in intubated patients who continue to bleed despite attempted endoscopic therapy. The most widely used technique is by the deployment of a Sengstaken-Blakemore tube. This is a temporary orogastric tube consisting of 2 inflatable balloons (gastric and oesophageal) and 2 lumens (gastric and proximal oesophagus). Once inserted the gastric balloon is inflated and the tube pulled back to achieve traction/compression of the feeding vessels at the gastro-oesophageal junction and gastric fundus. This controls bleeding in 90% of patients. The oesophageal balloon is rarely used and can cause ischaemic necrosis of the lower oesophageal mucosa, particularly if left inflated for protracted periods.
Lowering portal pressures by the use of drugs
Vasopressin or its derivative, terlipressin (either alone or in combination with nitro-glycerine) are used in patients with variceal bleeding. These drugs reduce portal blood flow and variceal pressures. However, these agents also increase systemic vascular resistance and therefore decrease cardiac output and coronary blood flow. Nitro-glycerine is therefore sometimes used to offset these negative cardiovascular effects. Terlipressin has been shown to be as effective as balloon tamponade in stopping bleeding.
Alternatively, somatostatin or its analogue octreotide can be used as they cause selective splanchnic vasoconstriction and hence reduce portal blood flow. Neither has been shown to reduce mortality.
Transjugular intrahepatic portosystemic shunt (TIPSS)
This procedure is used when variceal bleeding is not adequately controlled by endoscopy and/or balloon tamponade and terlipressin. It is performed by an interventional radiologist under local anaesthesia and sedation. The jugular vein is cannulated and from there the hepatic vein. Once in the hepatic vein, a tract is created through the liver parenchyma into the portal vein under fluoroscopic guidance. This tract is then dilated and an expandable metal stent inserted to create a shunt between the portal and systemic system. This causes a reduction in portal pressure. TIPSS is an effective means of achieving haemostasis in uncontrolled variceal haemorrhage. However, 25% of patients become encephalopathic post procedure and 50% of stents block after 1 year. TIPSS is therefore regarded by many as a rescue therapy when endoscopic therapy fails and a bridge to transplantation.
Surgical procedures
Surgical shunt procedures are an alternative to TIPSS and perform essentially the same task. They create a shunt between the portal and systemic system by surgical means (e.g. spleno-renal shunt) and are occasionally used in an acute situation to control variceal bleeding. Oesophageal transection and gastric devascularisation are rapidly becoming obsolete, but can be of benefit in those with both portal and splenic vein thrombosis who are not therefore candidates for a surgical shunt procedure.
Liver transplantation is the treatment of choice for patients with decompensated chronic liver disease who have had a life-threatening bleed. This rapidly reverses any portal hypertension and other consequences of liver failure.
Long term follow up
This is aimed at decreasing the patient’s risk of sustaining any future variceal bleeds. All patients should be started on a beta blocker after the initial bleed. Beta blockers decrease portal pressure and have been shown to reduce mortality by decreasing the risk of re-bleeding. Propranolol is commonly used. Low doses should be used in the first instance, as propranolol normally has a high ‘first pass’ metabolism. In patients with chronic liver disease this is lost and such patients are often sensitive to even paediatric doses. The dose is gradually titrated upwards with the aim of reducing the resting pulse rate by 25% (this is a good surrogate marker for effective reduction in portal pressure).
All patients who have bled should have repeated endoscopic treatment of their varices, ideally using band ligation. It is recommended that each varix be banded with a single band at 1 week intervals until eradicated. Patients should then be endoscoped at least every year to check for recurrent varices.



