Intensive Care Medicine
MULTIPLE CHOICE QUESTIONS
- Regarding organ donation after circulatory death
a) This was previously known as non-heart beating donation
b) It can only occur if a decision has been made that ongoing treatment is futile
c) Confirmation of death requires brain stem death testing by two independent consultants
d) Renal transplant outcomes are equivalent to those from donors after brain stem death
e) Warm and cold ischaemia times affect outcomes of organ survival - With regards to ischaemic times and organ function after DCD
a) Donor warm ischaemia starts from asystole until cold perfusion starts
b) Recipient warm ischaemia starts from removal of organ from ice until reperfusion
c) Cold ischaemia refers to the time between the end of donor warm ischemia to the onset of recipient warm ischemia
d) Functional warm ischaemia time is a better predictor of outcomes of organ survival
e) Functional warm ischaemia starts once the donor systolic pressure drops below 80mm of Hg and arterial SaO2 less than 90% - Regarding the DCD process
a) Infection is a contraindication for organ donation
b) HIV infection is a contraindication for organ donation
c) Consent from family members is not essential in presence of organ donation card
d) Functional warm ischaemia time limit for liver and pancreas is 30 minutes
e) In lung transplantation time to lung re-inflation is more critical to outcome than cold ischaemia
INTRODUCTION
Organ transplantation has revolutionised the management of many chronic diseases. Whilst it is a complex process, it can offer individuals with end stage organ failure improved quality of life, freedom from the burden of on-going treatment and improved survival. For the families of deceased donors organ donation may offer the reassurance that some good has come from the passing of a loved one, whilst for society as a whole, organ donation and transplantation can prove a cost effective strategy in the management of chronic disease.1
Transplanted organs can come from living and deceased donors. Unfortunately there is a persistent gap between the numbers of patients on waiting lists for organ transplants and the availability of suitable organs (see Figure 1)2. This has fuelled renewed interest in deceased donation after circulatory death (DCD). As DCD is becoming a more frequent occurrence familiarity with the processes involved in successful donation is a requirement for all doctors and nurses working in critical care medicine, anaesthesia and emergency medicine. The focus of this article is organ donation after circulatory death, including its definition, classification and a review of the processes involved in successful DCD. References for further reading are provided.
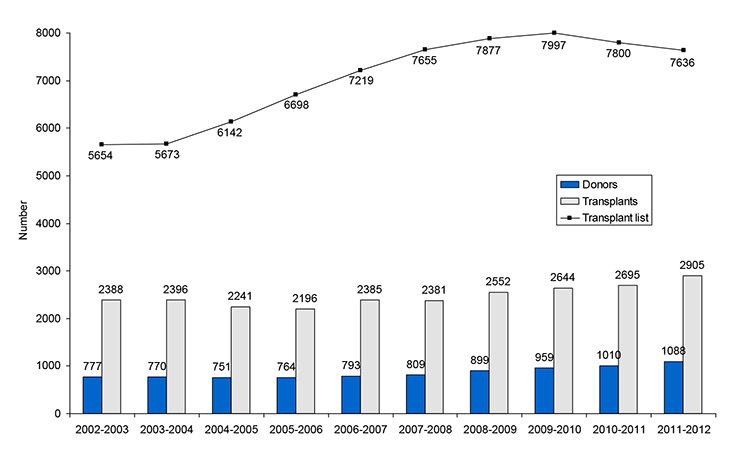
Figure 1. Number of deceased donors and transplants in the UK, 1st April 2002 – 31st March 2012, and patients on the active transplant list at 31st March each year.
DECEASED ORGAN DONATION
The definition and diagnosis of death are central to deceased organ donation. Death can be legally and formally defined as either circulatory death or brain death. Clear guidance on how both circulatory and brain death are defined has been provided by the Academy of Medical Royal Colleges.3 Circulatory death is defined as the irreversible cessation of circulatory function whilst brain death is the irreversible loss of function of the brain and brain stem. Organ retrieval and subsequent transplantation can occur after both brain death and circulatory death but this article will predominantly focus on donation after circulatory death (DCD).
Definition of Donation after Circulatory Death (DCD)
The term donation after circulatory death has replaced the previously used terms non-heart beating donation and donation after cardiac death. As expanded upon later different types of DCD exist, but in the UK, patients who have become DCD donors have usually died following withdrawal of organ support on an intensive care unit. Therefore, potential DCD donors in the UK are dependent on life-sustaining support. Critically, a decision needs to have been previously and independently made that continuation of such treatment or commencement of new treatments are not considered to be in the patient’s best interests.
Definition of Donation after Brain Death (DBD)
Donation after brain death, previously known as heart beating organ donation or donation after brain stem death, is defined as the retrieval of organs after confirmation of death using brain stem death testing criteria. This topic has been covered in a prior tutorial.4
Historical Context of DCD
Donation after circulatory death is not a new concept. As demonstrated in the timeline below, successful organ transplantation had been performed for many years prior to 1976, when clinical tests to confirm death using neurological criteria were first introduced. Before the publication of these guidelines the majority of organs were retrieved from DCD donors. However after 1976, most transplant centres switched rapidly to transplantation of organs from DBD donors. This was primarily because of concerns regarding the deleterious effects of the increased warm ischaemia times that are invariably associated with DCD. More recently, because of diminishing numbers of potential DBD donors, increasing demand for organ transplantation and advances in the management of both donors and recipients, there has been renewed focus on DCD donation. Evidence now shows that renal transplant outcomes after circulatory death are equivalent to those from donors after brain death, whilst the outcomes from liver, lung and pancreas DCD transplantation continue to improve.5 Many but not all European countries now have well-established DCD programmes6 and as can be seen in Figure 2, there has been a steady increase in DCD donation in recent years in the UK.2
Timeline: Historical Perspective of Organ Donation Worldwide:
1902 – Alexis Carrel paves the way for organ transplantation by demonstrating blood vessel anastomosis for the first time.
1905 – First reported cornea transplant takes place in Olmutz, Moravia
1954 – The world’s first successful kidney transplant is performed by Dr Joseph Murray in Boston, Massachusetts.
1960 – The UK’s first donor kidney transplant is performed by Sir Michael Woodruff at The Royal Infirmary of Edinburgh.
1963 – The world’s first liver transplant is performed by Dr Thomas Starzl in Denver, Colorado.
1965 – The first organ donation from a deceased donor takes place at the Royal Infirmary of Edinburgh in the UK.
1967 – The world’s first heart transplant is performed in by Dr Christiaan Barnard in Cape Town, South Africa.
1979 – The UK Transplant Service is formed through a merger between the National Tissue Typing and Reference Laboratory (NTTRL) and National Organ Matching and Distribution Service (NOMDS).
1979 – The UK’s first successful heart transplant is performed by Sir Terence English at Papworth Hospital.
1983 – The first combined heart and lung transplant in the UK is performed by Sir Magdi Yacoub at Harefield Hospital.
1994 – The NHS Organ Donor Register is set up to co-ordinate supply and demand following a five- year campaign.
1994 – The transplant of liver tissue from a living donor takes place for the first time in the UK, allowing both donor and recipients to have full liver function.
2001 – The first lung transplant from a ‘non-heart beating’ donor is performed by Dr Stig Steen in Lund, Sweden.
2005 – The first partial face transplant takes place in France.
2007 – The first altruistic living organ donation in the UK, with donors now being able to donate organs to people they do not know.
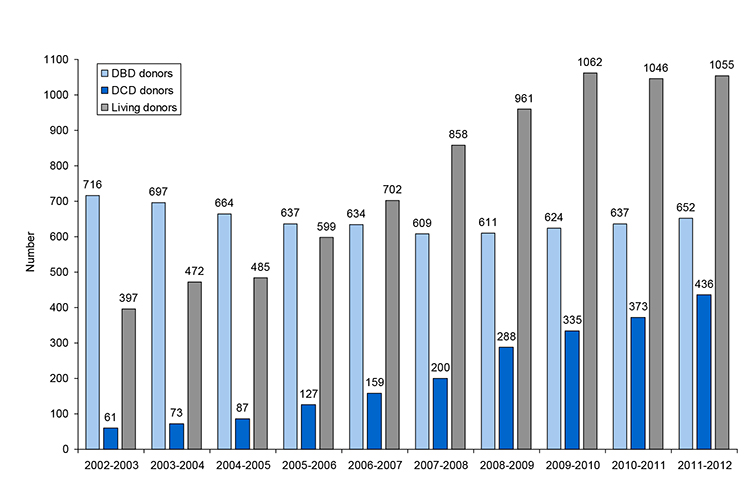
Figure 2. Number of deceased and living donors in the UK, 1st April 2002 – 31st March 2012
Classification of DCD
There are two broad categories of DCD donors.
Uncontrolled DCD is when death occurs suddenly and unexpectedly.
Controlled DCD is when death occurs after the planned withdrawal of life-sustaining treatment.
The modified Maastricht classification further classifies DCD into five categories as seen in Table 1.7 Largely for logistical reasons, uncontrolled DCD is restricted to kidney-only retrieval within transplant centres. In the UK, it is currently practiced in a very limited number of centres.
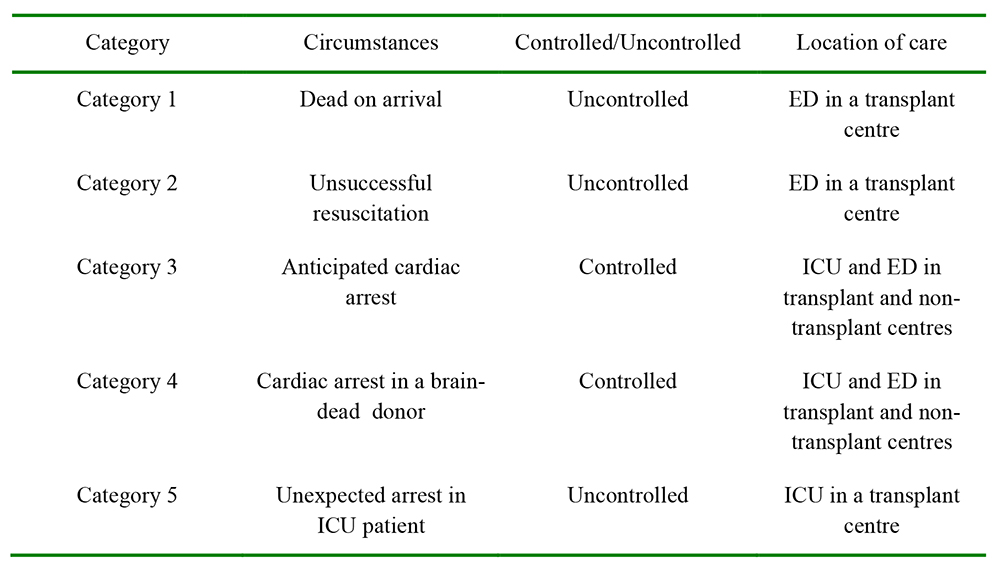
Table 1: Modified Maastrict Classification of DCD
Ischaemia and organ function after DCD
Successful organ function requires adequate perfusion. Different organs have differing susceptibilities to ischaemia induced injury and irreversible loss of function. Two types of ischaemia in retrieved organs have been described – warm ischaemia and cold ischaemia.8 Warm ischaemia may be further classified into donor and recipient warm ischaemia. Donor warm ischaemia starts from the time of asystole until cold perfusion begins. Unlike in DBD, where cold perfusion occurs prior to organ retrieval, donor warm ischaemia of over 10 minutes is inevitable in DCD. Recipient warm ischaemia starts from the removal of the organ from ice until reperfusion. Cold ischaemia refers to the time between the end of donor warm ischaemia until the onset of recipient warm ischaemia.
An understanding of the concept of functional warm ischaemia is important for all staff involved in the care of potential organ donors. Functional warm ischaemia commences once the donor’s physiological variables drop below certain thresholds. The current thresholds are a systolic blood pressure of less than 50 mm Hg an arterial oxygen saturation less than 70%, or both. Functional warm ischaemia ends with cold perfusion. Because different organs have differing susceptibility to warm ischaemia, organ specific thresholds for functional warm ischaemic time have been developed. DCD organ donation may not proceed if these thresholds have been or are due to be exceeded prior to organ retrieval. Recognition and recording of the onset of functional warm ischaemia is therefore vital. Table 2 provides current organ specific guidelines for maximum functional warm ischaemia times for the UK. However it is important to note that these are guidelines only. Each transplant centre and each transplant team may have their own acceptance criteria for organ suitability, including maximum duration of functional warm ischaemic time.
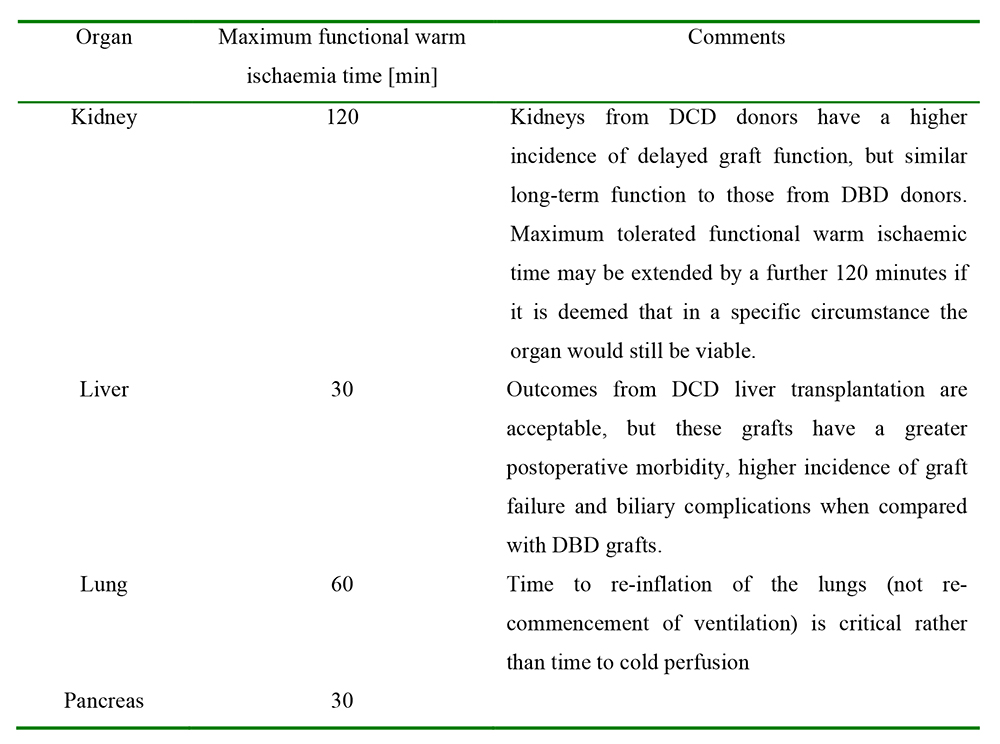
Table 2: UK functional warm ischaemia criteria for DCD organ retrieval 9
In addition to agreed organ specific limits for duration of functional warm ischaemia, there are also limits for duration of time between withdrawal of active medical treatment and asystole. Should these limits be exceeded then organ retrieval will not proceed. Table 3 outlines the stand down time for organ retrieval after withdrawal of life sustaining treatment (WLST) and onset of asystole.

Table 3: Stand Down time for Organ retrieval after WLST and onset of asystole9
Once again these times should be viewed as a guide rather than absolute values. The stand down times are organ and accepting centre specific.
CONTRAINDICATIONS TO DCD
The absolute contra-indications to donation of any organ are given below. Patients with one or more of these conditions should not be referred as potential DCD donors.
- Active invasive cancer in the last 3 years excluding non-melanoma skin cancer and primary brain tumour
- Haematological malignancy – myeloma, lymphoma, leukaemia
- Untreated systemic infection
- Variant CJD
- HIV disease (but not HIV infection)
If a patient fulfils the referral criteria for organ donation and there are no absolute contraindications it is best practice to discuss all potential donors with a specialist nurse for organ donation.
Legal and ethical background
Transplantation of human organs is now governed by The Human Tissue Act 2004. This Act replaces The Human Tissue Act 1961, The Anatomy Act 1984 and The Human Organ Transplants Act 1989. The Human Tissue Act was established to regulate activities concerning the removal, storage, use and disposal of human tissue and it covers England, Wales and Northern Ireland. Amongst other offences relating to un-authorised DNA analysis, the HTA makes the removal, storage or use of human tissue for scheduled purpose without appropriate consent, a criminal offence.10,11
The DCD Process
There are many different steps and staff groups involved in achieving successful organ donation and transplantation following DCD. The central person who co-ordinates the process is the specialist nurse for organ donation (SN-OD). Figure 3 provides a timeline for the organ donation process and outlines most of the numerous roles and responsibilities that the SN-OD has to undertake.
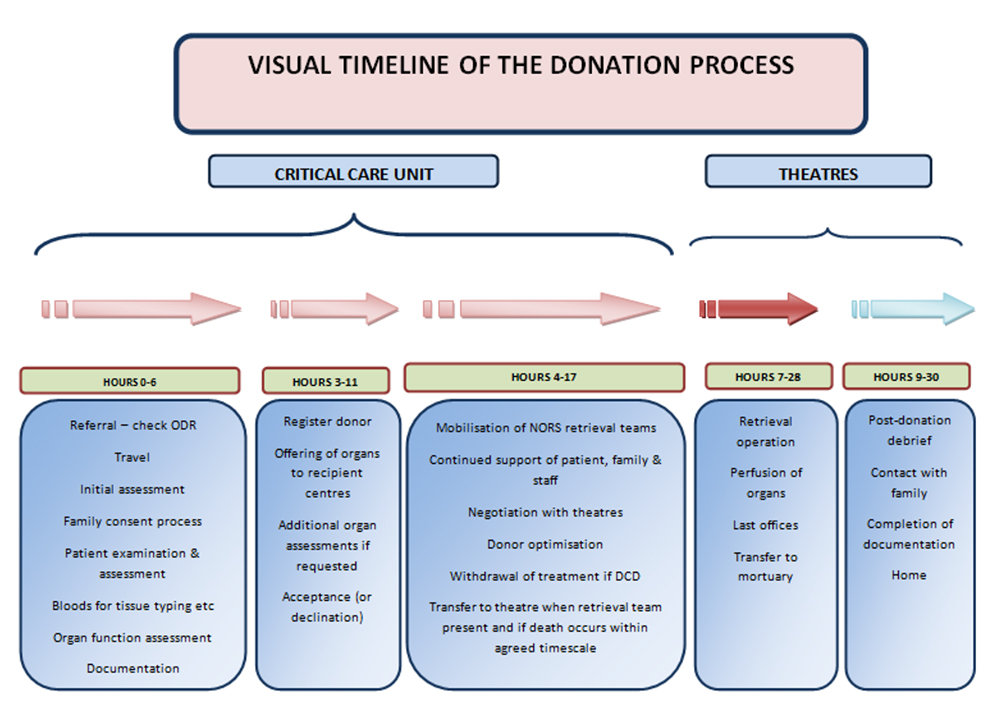
Figure 3: Timeline of the Donation process for Specialist Nurses for organ donation12 (Adapted with permission from the UK South West Organ Donation Team).
Key individuals and staff groups are:
- Specialist nurse for organ donation (SN-OD). Each acute trust in the UK has a nominated SN-OD. They often cover more than one trust so may not be resident in the hospital during normal working hours but there is always an on-call SN-OD that can be contacted at any time. There are 12 National Organ Donation Teams.
- Referring hospital team responsible for care of the potential donor and communication with the donor family. This is usually the referring hospital intensive care unit multi-disciplinary team and the SN-OD.
- The National Organ Retrieval Service, (NORS), Recipient Centre Points of Contact, (RCPoC) and transplantation teams.
- Theatre co-ordinator. The retrieval team will require a vacant theatre from the time of planned withdrawal of life-sustaining treatment for the donor. The retrieval team will bring their own theatre team but may require some additional anaesthetic support and assistance from the local operating theatres staff.
- Laboratory staff to perform tissue typing, cross matching, virology, microbiology, biochemistry and haematology.
- Additional staff including radiology/portering staff/chaplaincy/bereavement officers.
The individual steps involved in the DCD process are described below.
Step 1- Decision of futility of treatment
The critical trigger in the DCD process is the decision that continuing life sustaining treatment is no longer in the patient’s best interest and should therefore be discontinued. This decision should be made by the Consultant responsible for the patient and must be independent of the DCD process. This decision then needs to be communicated to and then understood and accepted by the potential donor’s family, prior to the subject of organ donation being raised.13 As with all patients, potential organ donors must be treated with dignity and respect at all times. This last point has been emphasized in recent guidance from the GMC.14
Step 2- Seeking consent for organ donation
Discussions about organ donation should only occur once the potential donor’s family have accepted the withdrawal of active medical treatment. The governing body in the UK advocates that initial discussions with the family about the potential for organ donation should involve the specialist nurse for organ donation (SN-OD). However this is not always possible, in which case preliminary discussions may be undertaken by members of the referring hospital intensive care team. It cannot be emphasised enough how important the input of a SN-OD is, both in communication with the family and also in ensuring that the entire process is properly co-ordinated. In the UK the organ donor register should be checked prior to approaching the family for consent. This may help inform about the views of the patient, but organ donation will not proceed without the consent of the family. During the formal completion of consent for organ donation the SN-OD will discuss the requirement for and implications of virology testing. Following consent for organ donation, blood samples are obtained from the potential donor for a full virological screen and tissue typing.
Either the responsible Consultant or the SN-OD will liaise, when necessary, with the Coroner or the Procurator Fiscal in Scotland, to obtain permission to proceed with organ retrieval. The SN-OD will contact the appropriate retrieval centres.
Step 3- Allocation of Organs
The allocation of the organs to be donated is a lengthy process, focussing upon recipient prioritisation, safety and organ suitability. The SN-OD will assess the potential donor’s suitability in general and the suitability of individual organs. This will include a thorough review of individual organ function and an assessment of the current admission and past medical and social history. This information is gained by reviewing medical documentation, performing a physical assessment, interviewing the donor’s next of kin and completing an extensive medical questionnaire with the donor’s General Practitioner. Once the donor organs have been allocated, the SN-OD will arrange a vacant theatre and will mobilise the NORS.
Step 4- Management before withdrawal of life sustaining treatment
Supportive care should be continued in the period between considering organ donation and withdrawal of life sustaining treatment. Escalation of treatment to facilitate organ donation is an area of controversy. Legal guidelines only recommend measures that may facilitate donation if they do not cause the patient harm or distress. This may include increasing vasoactive drug infusions, increasing ventilatory support and inserting intravenous lines, but all proposed interventions should be carefully considered. More detailed guidance is available within the 2010 consensus document on organ donation after circulatory death.9
Step 5- Withdrawal of life-sustaining treatment (WLST)
Once retrieval teams are set up in a vacant theatre and all involved parties are informed, the SN-OD will request the withdrawal of life-sustaining treatment. Treatment withdrawal for DCD donors in the UK is often carried out in the intensive care unit, although many Trusts have a protocol to perform this in an anaesthetic room. Occasionally WLST may occur in the emergency department,15 however it is our recommendation that this is best performed, where resources allow, in an intensive care setting. It is vital that the SNOD documents the timing of WLST and haemodynamic deterioration, to allow clear communication of the onset of functional warm ischaemic time to the retrieval and transplantation teams.
Within the UK there are significant variations in how withdrawal of life-sustaining treatment is sequenced. This includes withdrawal in patients who are proposed organ donors. Each institution should follow their local protocols. Importantly practice should not be altered because of the potential for organ donation. Infusions of sedative or opioid medications may be continued or commenced as appropriate, in line with local and national end of life care guidance. Withdrawal will usually involve cessation of mechanical ventilation as well as any other forms of organ support. The endo-tracheal tubes of potential donors may be removed or left in place. If an endotracheal tube of a potential lung DCD donor has been removed, re-intubation and a recruitment manoeuvre will be required after confirmation of death and prior to organ donation. This is an area of considerable controversy and each trust should develop their guidelines on this. Importantly mechanical ventilation should not be commenced after confirmation of circulatory death.
Step 6- Diagnosis of circulatory death
Diagnosis of circulatory death
Circulatory death is defined as the irreversible cessation of circulatory function. The individual should be observed by the medical practitioner confirming death for a minimum of five minutes to establish that irreversible cardiorespiratory arrest has occurred. The absence of mechanical cardiac function is normally confirmed using a combination of the following:
- -absence of a central pulse on palpation
- -absence of heart sounds on auscultation
- -asystole on a continuous ECG display
- -absence of pulsatile flow using direct intra-arterial pressure monitoring
- -absence of contractile activity using echocardiography.
Any spontaneous return of cardiac or respiratory activity during this period of observation should prompt a further five minutes observation from the next point of cardiorespiratory arrest.
After five minutes of continued cardiorespiratory arrest the absence of the pupillary response to light and corneal reflexes, together with the absence of any motor response to supra-orbital pressure should be confirmed.14
The time of death is recorded as the time at which these criteria are fulfilled.
Step 7- Arrangements for transfer to the operating theatre
During the consent process it should be clearly explained to families that after confirmation of death the deceased will remain in the intensive care unit for 5 minutes only prior to transfer to theatres. The family may wish to remain with the deceased for this time. Following the diagnosis and confirmation of death the deceased should be transferred to theatres in a swift, dignified manner, so as to minimise donor organ warm-ischaemia. The deceased will be accompanied by the SN-OD and a member of the intensive care team. The SN-OD is responsible for coordinating the safe transfer of the patient to the operating table, completion of the WHO checklist, documentation and communication of key timings.
Step 8- Organ retrieval and end of life care
The attending NORS team performs the retrieval operation. A thorough in situ assessment is performed by the NORS surgeons prior to organ removal. Following the end of the organ retrieval process care after death is performed by the SN-OD, in keeping with the religious and cultural practices of the deceased. Post donation, the SN-OD will contact the donor family to offer both additional support and information about the outcome of the donation.
CONCLUSION
Although often emotive and resource intense, the donation process frequently offers the opportunity of a much improved quality of life for recipients and long term comfort for donor families. Communication, multidisciplinary working, structured identification and referral processes are all crucial in the provision of high quality organ donation services. We must strive to incorporate all of these elements so that we can meet the long-term target of reducing the current organ donor waiting lists and reducing the numbers of individuals dying whilst waiting for a transplant.
ACKNOWLEDGEMENTS
The authors are very grateful to Ms Joanna Buckland, Specialist Nurse for Organ Donation, South West Organ Donation Team, for her valuable advice and assistance in preparing this manuscript.
ANSWERS TO QUESTIONS
- a)true b)true c)false d)true e)true
- a)true b)true c)true d)true e)false
- a)false b)false c)false d)true e)true
REFERENCES
- http://www.organdonation.nhs.uk/ukt/newsroom/fact_sheets/cost_effectiveness_of_transplantation.asp (accessed 11/11/2012)
- NHSBT Activity Report 2011/12 slide set, Overview of Organ Donation and Transplantation
- UK Donation Ethics Committee, (UKDEC), An Ethical Framework for Controlled Donation after Circulatory Death, December 2011
- http://totw.anaesthesiologists.org/2008/10/14/brainstem-death/ (accessed 11/03/2013)
- M.D.Doshi, L.G. Hunsicker et al, Short-and long- term outcomes with the use of kidneys and livers donated after cardiac death, Am J Transplant. 2007 Jan;7(1):122-9. Epub 2006 Oct 25
- Dominguez-Gil, Haase-Kromwijk et al, Current situation of donation after circulatory death in European countries, Transpl Int. 2011 Jul;24(7):676-86
- A.R.Manara, P.G.Murphy and G.O’Callaghan, Donation after circulatory death, BJA,vol 108,Supp1,Jan 2012
- Halazum K J, Al-Mukhtar, Ahmad N, Warm ischaemia in transplantation:search for a consensus definition, Transplant Proc 2007 Jun;39(5):1329-31.
- Department of Health. Organ Donation after Circulatory Death. Report of a consensus meeting. Inteisive Care Society, NHS Blood and Transplant, and British Transplantation Society, 2010, (accessed by visiting http://www.ics.ac.uk/intensive_care_professional/standards_and_ guidelines/dcd)
- http://www.hta.gov.uk/legislationpoliciesandcodesofpractice/legislation/humantissueact.cfm
- http://www.legislation.gov.uk/ukpga/2004/30/notes/contents
- South West Organ Donation Team 2011 Sharon Henry
- NHSBT, Timely Identification and Referral of Potential Organ Donors, A strategy for Implementation of Best Practice, September 2012
- http://www.aomrc.org.uk. The code of practice for the diagnosis and confirmation of death. Academy of the medical royal colleges.
- Genral Medical Council, Treatment and Care Towards the End of Life: Good Practice in Decision Making, May 2010
- British Transplantation Service and the College of Emergency Medicine,The Role of Emergency Medicine in Organ Donation; Report of a Workshop, October 4th 2010



