Obstetrics Anaesthesia
KEY POINTS
- Most peripartum neurological complications are due to compressive neuropathy secondary to obstetric causes, and most recover spontaneously.
- The incidence of permanent neurological injury following neuraxial anaesthesia is estimated to be 1:80 000 to 1:320 425.
- A patient with neuraxial labour analgesia who cannot straight-leg raise should be evaluated immediately by an anaesthetist.
- Patients unable to straight-leg raise 4 hours after administration of neuraxial surgical anaesthesia should be evaluated immediately by an anaesthetist, followed by appropriate and timely investigations.
- Patients should be advised when to expect resolution of the neuraxial analgesia or anaesthesia.
INTRODUCTION
The third National Audit Project (NAP3) published by the Royal College of Anaesthetists in the United Kingdom concluded that neurological complications arising from neuraxial procedures are rare.1 Failure to rapidly diagnose and manage serious neurological pathologies can have devastating consequences. Peripartum neurological injury is frequently obstetrical in origin, arising from pregnancy, labour or delivery, and is often incorrectly attributed to a neuraxial procedure.2 Therefore, it is essential for an obstetric anaesthetist to understand the potential mechanisms of nerve injury.
Neurological diseases in pregnancy can be classified into 3 categories:
- Preexisting chronic neurological diseases (eg, epilepsy, multiple sclerosis, spinal cord injury, raised intracranial pressure , and shunts)
- Diseases with onset during pregnancy or with exacerbation or deterioration during pregnancy (eg, brain tumours or prolapsed intervertebral disc with increasing symptomatology)
- Pregnancy-induced (acquired) disease (eg, eclampsia or posterior reversible encephalopathic syndrome_
This tutorial provides a comprehensive approach to the assessment, diagnosis and timely intervention(s) to ensure optimal outcomes if neurological pathologies occur and will predominantly focus on peripartum peripheral nerve injuries.
BACKGROUND
In 2017, Roderick et al3 published results of a national survey of neurological monitoring practice following obstetric anaesthesia in the United Kingdom. They found that only 63 of 112 (56%) units had a monitoring policy and assessment methods, and schedules were highly variable.
Intrinsic obstetric palsies are those attributed to the labour and delivery process itself. The incidence of these has fallen from 3.2% a century ago to less than 0.25%, likely because of a shortened second stage of labour.4 They are often sensory or motor, involving multiple lumbosacral nerve roots and typically resolve within a short time period of 6 to 8 weeks.
Data from the NAP3 report an incidence of permanent injury in patients who had a central neuraxial block (CNB) to be ‘pessimistically’ 4.2 per 100 000 cases (1:24 000) and ‘optimistically’ 2.0 per 100 000 (1:54 000).1 The NAP3 has garnered widespread interest across the specialty and has greatly informed practice with accurate estimates of risk and incidence, which for permanent harm following CNB is lower than reported in some smaller studies, which is reassuring.1
The summary of the ‘Safety Guideline: Neurological Monitoring Associated With Obstetric Neuraxial Block 20200, published by the Association of Anaesthetists and Obstetric Anaesthetists’ Association in the United Kingdom,5 concluded the following:
- During labour, the anaesthetist should be alerted if a woman is unable to straight-leg raise. Minor degrees of motor block are common, even with modern low-dose, low-concentration local anaesthetic regimens. However, any woman with profound motor and sensory block should be assessed by an anaesthetist and, where there is concern, management escalated.
- During the recovery phase after a spinal or epidural top-up, straight-leg raising should be used as a screening method to assess motor block. If the woman is unable to straight-leg raise at 4 hours from the last dose of epidural or intrathecal local anaesthetic, an anaesthetist should be called to assess whether the woman requires urgent investigation to exclude reversible causes of neurological injury.
- Women should be informed of how long neuraxial blocks are expected to last and encouraged to alert staff if recovery is delayed.
- Each maternity unit should have a policy in place to guide the escalation of care.
Peripartum Neurology
A thorough history and examination is essential in formulating a correct diagnosis when assessing a patient presenting with abnormal neurological signs or symptoms in the peripartum period.4 Careful consideration of the factors around labour and delivery such as obstetrical history, onset and duration of labour (in particular the second stage), patient positioning throughout labour (and in particular the second stage), along with any requirement for instrumental or operative delivery are important.
Anaesthetic-related considerations include the choice of neuraxial technique (spinal, epidural, or combined spinal-epidural) any difficulties (or patient-reported symptom) encountered during placement, duration of the block, and any additional doses and/or adjuncts administered. Other considerations include a history of preexisting back pain (including scoliosis and/or back surgery) or neurological condition.
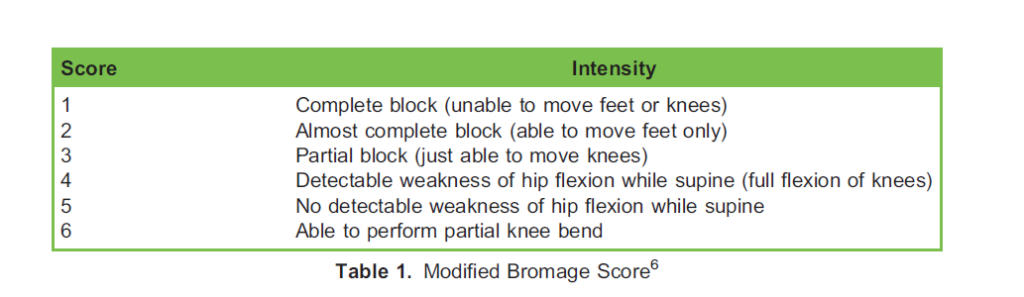
During neuraxial labour analgesia, all women should be assessed on a regular basis for motor block, and following surgery with neuraxial anaesthesia (for example, after a caesarean delivery), all women should be tested for the ability to straight-leg raise 4 hours after administration of the last dose.5 In addition to testing sensory block height, the Modified Bromage scale (Table 1) is a standardised method for motor assessment after neuraxial analgesia or anaesthesia.6
Obstetric-Related Neuropathy
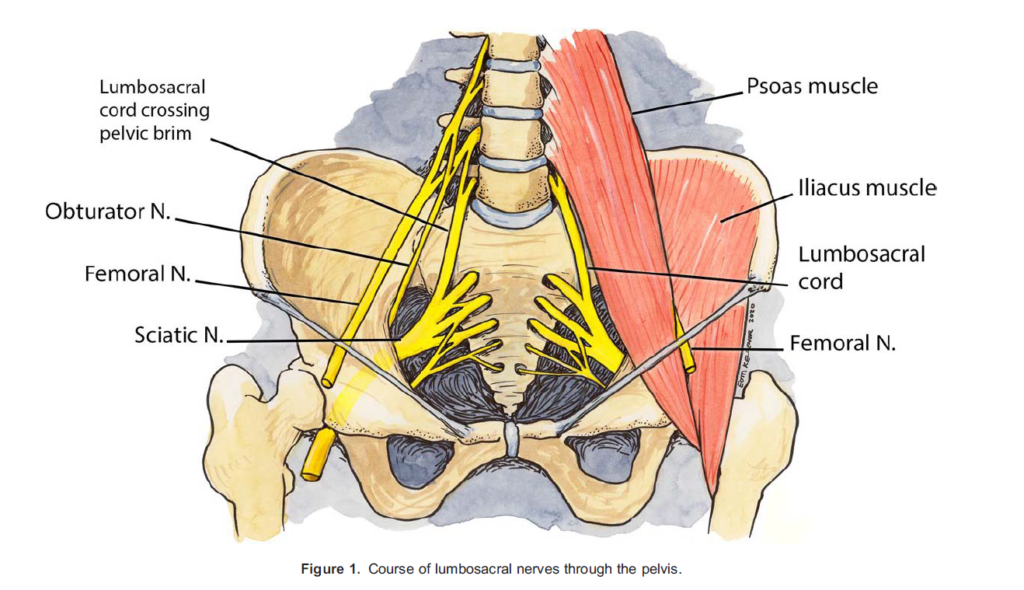
Most nerve palsies that present in postpartum patients are obstetric related in origin, for example, pressure encountered on nerves during labour and delivery or directly from the foetus can affect central, lumbosacral roots, or peripheral nerves (Figure 1). Risk factors for obstetric neuropathy (Table 2) include primiparity, positioning during and a prolonged second stage of labour, instrumental delivery and a large foetal head.7
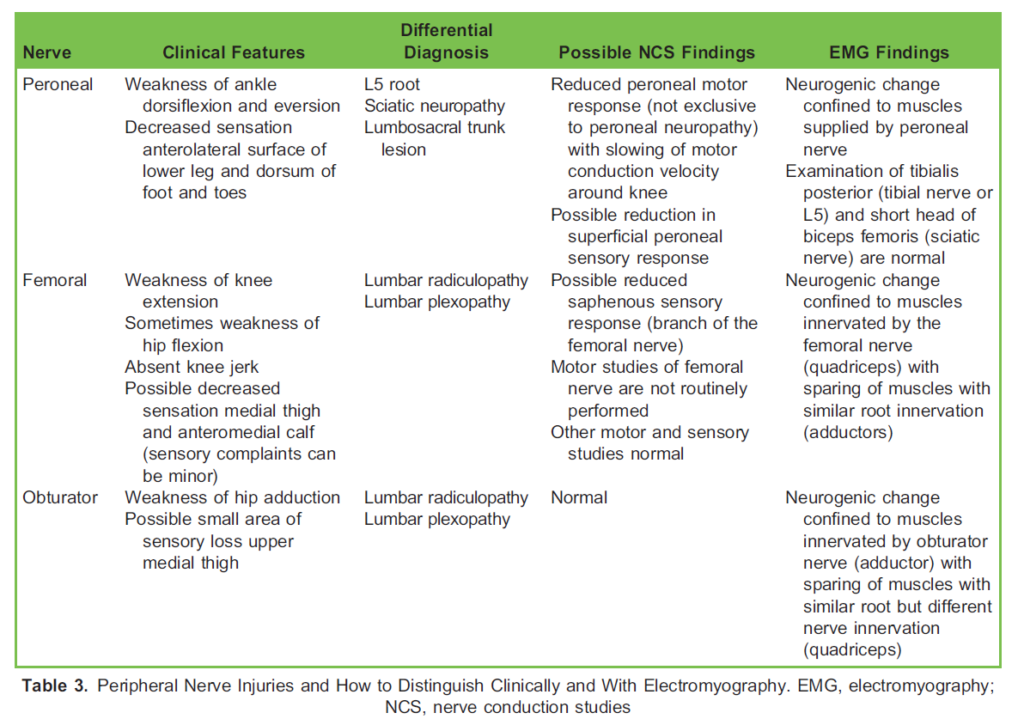
The most common intrinsic obstetric palsy is lateral femoral neuropathy.9 Palsies of the femoral, obturator, sciatic, common peroneal and lumbosacral plexus have all been reported. Femoral and obturator nerve injuries sometimes occur bilaterally and so can be mistaken for an intraspinal lesion. Femoral dysfunction may result in palsy of hip flexion (leading to an inability to climb stairs), decreased patellar reflex and sensory loss in the distribution of the femoral nerve. Neuropathy of the obturator nerve causes weakness of hip adduction and internal rotation and decreased sensation on the inner aspect of the thigh.
When patients are positioned in stirrups, it is particularly important to use padding to avoid injury to the common peroneal nerve from pressure on the lateral aspect of the knee, which will result in an area of sensory loss to the lower lateral aspect of the lower limb and dorsal aspect of the foot (Table 3).
Signs and symptoms of obstetric palsies usually recover over 6 to 8 weeks. Sensory changes secondary to neuraxial labour analgesia may contribute to obstetric-related nerve palsies by reducing the patient’s awareness of discomfort from positioning. It is important to always perform a thorough history and examination before block placement, and important information to provide to patients to reduce the risk of nerve injuries includes advice to regularly change position (especially during the second stage of labour), avoid prolonged hip flexion or extreme thigh abduction and external rotation and emphasise to patients with preexisting back pain and/or neurological conditions not to lie in positions that they anticipate could exacerbate or aggravate symptoms. Administration of low concentrations of labour neuraxial analgesia solutions can help minimize a dense motor and sensory block.

Causes Related to Neuraxial Analgesia or Anaesthesia
Nerve Injury
Nerve damage may be secondary to direct needle injury or intraneuronal injection, and signs and symptoms include paraesthesia, loss of sensation and/or motor weakness in the nerve distribution. Prevention of damage involves withdrawing the needle if the patient complains of paraesthesia during insertion and subsequent redirection of the needle prior to injection of local anaesthetic.
Spinal Cord Trauma
Direct trauma to the spinal cord or conus may occur when performing a spinal, epidural or combined spinal-epidural, and signs and symptoms include pain on needle insertion (which should prompt immediate withdrawal), and/or prolonged motor and sensory weakness at and below the level of injury should alert suspicion. While bilateral signs and symptoms suggest spinal cord pathology, they may be unilateral, and bladder or bowel symptoms may be present. In adults, the conus medullaris typically ends at L1 or L2, but in 10% of patients, it may extend to L2 or L3, and so preprocedural neuraxial ultrasound may offer a margin of safety by increasing the accuracy of identifying midline and the appropriate lumbar level when compared with surface anatomy.10
Cauda Equina Syndrome
Cauda equina syndrome (CES) occurs secondary to compression or trauma to the cauda equina fibres, and signs and symptoms include back pain, nerve root pain, saddle anaesthesia, paraplegia or bladder or bowel sphincter dysfunction. CES occurs commonly due to disc herniation in the lumbar region. Other causes include spinal lesions and tumours, spinal infections or inflammation of spinal cord, spinal arteriovenous malformations and, as a complication, post–lumbar spine surgery. Incorrect neuraxial drug administration (intrathecally or via the epidural catheter) may result in devastating consequences such as CES due to neural toxicity; therefore, it is prudent to perform a 2-person check during drug preparation.

Epidural Haematoma
An epidural haematoma11 can occur spontaneously in patients who have not had a CNB or in patients who have had a CNB with risk factors such as coagulopathy or a bleeding disorder (acquired or hereditary) and a difficult block placement, which is why it is important to obtain an accurate medical and drug history prior to block placement. An epidural haematoma causes neural ischaemia due to compression, and signs and symptoms include rapid-onset acute back pain, nerve root irritation and lower-limb motor weakness with paralysis as a late sign. A thorough preprocedure medical and drug history should decrease the risk along with appropriate timing of prophylactic and therapeutic doses of anticoagulants before and after block placement and epidural catheter removal (if applicable).12 The incidence of epidural haematoma in the obstetric setting has been reported to be 1:168 000.11
Neurotoxicity or Chemical Arachnoiditis
CNB placement is a sterile procedure, and skin preparation involves the application of an alcohol-based sterile solution while taking care to avoid contaminating the needle and epidural catheter with this solution. In the United Kingdom, 0.5% chlorhexidine with 70% alcohol is recommended for skin preparation with an adequate amount of time to dry13 to reduce the risk of neurotoxicity and chemical arachnoiditis (Table 4). Internationally, 0.5% and 2% chlorhexidine sponges in 70% alcohol (ChloraPrep) are available.
Vascular Injury
Anterior spinal artery syndrome (anterior spinal cord infarction) may result from trauma, atherosclerosis, aortopathies, vasculitis, prolonged hypotension or emboli in the artery of Adamkiewicz. The anterior spinal artery is the only arterial supply (with few collaterals), which makes the anterior cord very susceptible. The syndrome can present with paraplegia (motor and sensory loss to pain and temperature) below the level of the lesion due to interruption of the corticospinal tracts; however, proprioception and vibratory sensation are preserved via the dorsal columns. Autonomic dysfunction may present as hypotension, bladder or bowel dysfunction and areflexia.
Treatment is based on the cause, and prognosis is poor with mortality as high as 23%,14 and a further 24% of patients will have little or no improvement in long-term neurological function.15
Neurological signs and symptoms that warrant urgent review are listed below (Table 5).

Preexisting Neurological Condition
Pregnant women with preexisting neurological disease require a multidisciplinary team approach including neurology, obstetric medicine and anaesthesiology. The patient should be observed for any associated complications that may occur during pregnancy and a tailored plan for labour and delivery agreed upon.16 Following a detailed history and examination (with clear documentation of any findings), a patient with previous back surgery (or injury) and/or a preexisting neurological condition should be informed of the risks or benefits and alternative options for neuraxial analgesia or anaesthesia prior to undergoing a neuraxial procedure. Risk will vary significantly between individual patients, and it is preferable that patients are preassessed, informed and counselled in a controlled prelabour or presurgery setting.
Neuraxial labour analgesia is a safe and effective analgesic modality, but it may be contraindicated in specific cases, for example, patients with coagulopathy or with elevated intracranial pressure.16 If a neuraxial procedure is contraindicated, an opioid-based (eg, remifentanil) patient-controlled intravenous analgesia regimen is an appropriate alternative for labour analgesia and general anaesthesia for a surgical procedure (eg, caesarean delivery).
Recovery Time for Peripheral Nerve injuries
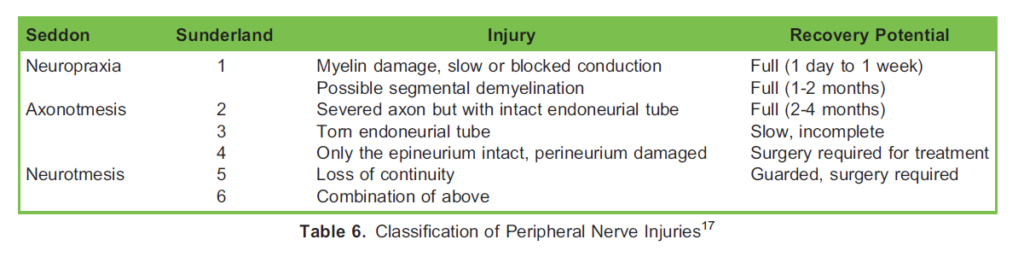
When a peripheral nerve is compressed, mild injury produces paranodal demyelination confined to the areas adjacent to the nodes of Ranvier, whereas severe damage causes loss of whole segments of myelin. Over a longer period of time, axonal disruption leads to Wallerian degeneration (Table 6).
Investigations and Management of Peripheral Nerve Injuries
Radiology
Magnetic resonance imaging and computed tomography are used to confirm or exclude central neuraxial lesions such as an abscess, haematoma or tumour (magnetic resonance imaging is the preferred imaging modality, as it enables excellent visualisation of soft-tissue pathologies and inflammation, although computed tomography may be more easily accessible). Signs and symptoms such as pyrexia, elevated white cell count, acute back pain, acute neurological deficit and/or bladder or bowel dysfunction require urgent examination and imaging.
Nerve Conduction Studies and Electromyography
Following neurologic examination of a peripheral nerve lesion, if signs or symptoms do not resolve within a few weeks, specific studies can be performed to identify the aetiology. Nerve conduction studies involve small electric shock stimulation of the peripheral nerve and the response recorded from the appropriate muscle or sensory point.
Electromyography involves placement of a needle electrode into a muscle to identify the neurogenic change from axonal loss. Denervated muscle fibres produce fibrillation potentials and positive sharp waves; however, these findings are not exclusive to nerve injuries, and other features help differentiate between myopathic and neurogenic aetiologies.
Neurosurgical Intervention
Prompt recognition of any neuropathy is essential, as urgent neurosurgical intervention may be required. Bos et al18 reported no difference in outcome between patients treated within 6 hours and 7 to 12 hours after the first symptoms, and further analysis indicated that neurological outcome was favourable in patients treated within the first 12 hours from symptom onset as compared with delayed intervention (after .12 hours).
Physiotherapy and Occupational Therapy
Early and continual input from physiotherapy and occupational therapy is essential to optimise recovery in patients with neurological deficit and may include occupational aids and exercise programs.
CONCLUSION
Neuraxial procedures are safe and recognised techniques for labour analgesia and surgical anaesthesia; however, they are associated with risks and complications. If a complication is identified, an immediate thorough neurological examination is essential with escalation to a radiologist and neurosurgeon if applicable, as it can be time sensitive to optimise the outcome.
All maternity units should have guidelines to facilitate timely referral of patients with neurological injury, and this also applies to patients who present to their general practitioner or emergency department. Most peripartum nerve injuries are obstetrical in origin and usually resolve within 6 to 8 weeks, but some can take months to years to completely resolve, and some may be permanent.4
REFERENCES
- Cook TM, Mihai R, Wildsmith JA; Royal College of Anaesthetists Third National Audit Project Working Group. A national census of central neuraxial block in the UK: results of the snapshot phase of the Third National Audit Project of the Royal College of Anaesthetists. Anaesthesia. 2008;63(2):143-146.
- Birnbach DJ, Hernandez M, van Zundert AA. Neurologic complications of neuraxial analgesia for labor. Curr Opin Anaesthesiol. 2005;18(5):513-517.
- Roderick E, Hoyle J, Yentis SM. A national survey of neurological monitoring practice after obstetric regional anaesthesia in the UK. Anaesthesia. 2017;72(6):755-759.
- Hosley CM, McCullough LD. Acute neurological issues in pregnancy and the peripartum. Neurohospitalist. 2011;1(2):104-116.
- Yentis SM, Lucas DN, Brigante L, et al. Safety guideline: neurological monitoring associated with obstetric neuraxial block 2020: a joint guideline by the Association of Anaesthetists and the Obstetric Anaesthetists’ Association. Anaesthesia. 2020;75:913-919.
- Breen TW, Shapiro T, Glass B, Foster-Payne D, Oriol NE. Epidural analgesia for labour in an ambulatory patient. Anaesth Analg. 1993;77:919-924.
- Boyce H, Plaat F. Post-natal neurological problems. Continuing Education in Anaesthesia Critical Care & Pain. 2013;13(2):63-66.
- Duncan A, Patel S. Neurological complications in obstetric regional anesthetic practice. J Obstet Anaesth Crit Care. 2016;6(1):3-10.
- Wong CA. Nerve injuries after neuraxial anaesthesia and their medicolegal implications. Best Pract Res Clin Obstet Gynaecol. 2010;24(3):367-381.
- Schlotterbeck H, Schaeffer R, Dow WA, Touret Y, Bailey S, Diemunsch P. Ultrasonographic control of the puncture level for lumbar neuraxial block in obstetric anaesthesia. Br J Anaesth. 2008;100(2):230-234.
- Ruppen W, Derry S, McQuay H, Moore RA. Incidence of epidural hematoma, infection, and neurologic injury in obstetric patients with epidural analgesia/anesthesia. Anaesthesiology. 2006;105(2):394-399.
- Leffert L, Butwick A, Carvalho B, et al. The Society for Obstetric Anesthesia and Perinatology consensus statement on the anesthetic management of pregnant and postpartum women receiving thromboprophylaxis or higher dose anticoagulants. Anesth Analg. 2018;126(3):928-944.
- Association of Anaesthetists of Great Britain and Ireland, Obstetric Anaesthetists’ Association, Regional Anaesthesia UK, et al. Safety guideline: skin antisepsis for central neuraxial blockade. Anaesthesia. 2014;69(11):1279-1286.
- Pearl NA, Dubensky L. Anterior cord syndrome. StatPearls. Treasure Island, FL: StatPearls Publishing; 2020.
- Cheshire WP, Santos CC, Massey EW, Howard JF. Spinal cord infarction: etiology and outcome. Neurology. 1996;47(2):321330.
- Chang LY, Carabuena JM, Camann W. Neurologic issues and obstetric anesthesia. Semin Neurol. 2011;31(4):374-384.
- Chhabra A, Ahlawat S, Andreseik G. Peripheral nerve injury grading simplified on MR neurography: as referenced to Seddon and Sunderland classifications. Indian J Radiol Imaging. 2014;24(3):217-224.
- Bos EME, Haumann J, de Quelerij M, et al. Haematoma and abscess after neuraxial anaesthesia: a review of 647 cases. Br J Anaesth. 2018;120(4):693-704.



