Basic Sciences
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation.
- Regarding the anatomy of the stellate ganglion:
a) It receives contribution from the first thoracic ganglion
b) It is situated anterior to the transverse process of C7
c) It supplies sympathetic efferent fibres to the hand, neck, head and heart
d) The ganglion lies superiorly and posterior to the dome of the pleura
e) The vertebral artery lies lateral the ganglion - Stellate ganglion block is useful in the treatment of :
a) Thrombo-angitis obliterans
b) Refractory angina
c) Phantom limb pain
d) Migraine
e) Scleroderma - Possible complications of stellate ganglion block are:
a) Pneumothorax
b) Oesophageal perforation
c) Seizures
d) Mydriasis
e) Tachycardia
INTRODUCTION
The sympathetic nervous system (SNS) directly controls involuntary human homeostatic activities and has a major role in neuropathic, vascular, and visceral pain. Sympathetically maintained pain occurs in a variety of vascular pathologies such as occlusive arterial diseases, diabetes mellitus or venous ulceration and neuropathic conditions such as complex regional pain syndrome (CRPS), postherpetic neuralgia and after peripheral nerve lesion. CRPS type I (formerly known as reflex sympathetic dystrophy) occurs following an initiating event such as trauma or injury but with little or no nerve injury while CRPS Type II (formerly causalgia) has nerve injury as its causative factor.
The complexity of vascular (ischaemic) pain can be traced to a dysfunctional microcirculation. Neuropathic pain can be due to neural ischaemia with resultant neurogenic inflammation1,2. The underlying pathophysiological mechanism is unclear, and there are suggestions indicating the presence of an abnormal coupling between the sympathetic and the somatosensory nervous system. This can occur either in the peripheral nerves or in the dorsal root ganglion. CRPS is associated with cortical, sensory and motor dysfunction along with peripheral inflammatory changes.
It has been proposed that a temporary sympathetic block can be useful in the discrimination of sympathetically maintained and sympathetically independent pain, however the role of these blocks in providing effective analgesia is undetermined with evidence coming from very few placebo controlled, blinded studies3-4.
ANATOMY
The stellate ganglion is a sympathetic ganglion situated on either side of the root of the neck. It is formed on each side of the neck by the fusion of the inferior cervical ganglion with the first, and occasionally second, thoracic ganglion. The stellate ganglion is only supplied by efferent sympathetic nfibres from the ipsilateral sympathetic chain (which lies inferiorly), along with the first and second thoracic segmental anterior rami.
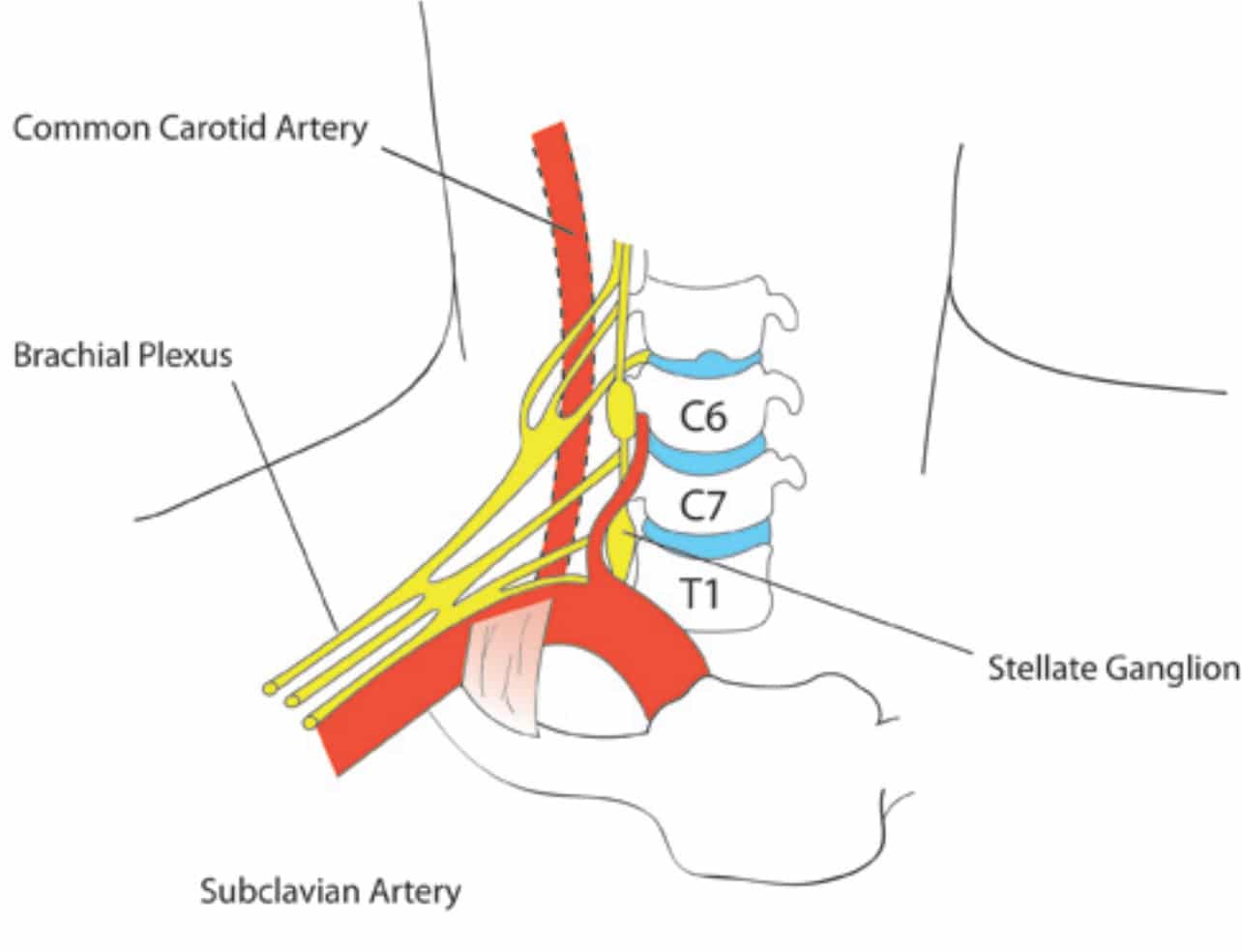
Figure 1. Anatomy of the Stellate Ganglion
The anatomical relations are:
anteriorly:
- subcutaneous tissue
- sternocleidomastoid muscle
- subclavian artery
- carotid sheath
posteriorly:
- anterior scalene muscle
- sheath of the brachial plexus
- neck of first rib
- transverse process of C7
- vertebral artery
- longus colli muscle
laterally:
- superior intercostal vein
- superior intercostal artery
- ventral ramus of first thoracic nerve
medially:
- prevertebral fascia
- vertebral body of C7
- oesophagus
- thoracic duct
inferiorly:
- pleural dome over the lung apex
INDICATIONS
Chronic Pain conditions
- CRPS 1 and 2
- Herpes zoster affecting the face and neck
- Refractory chest pain or Angina
- Phantom limb pain
Vascular Disorders of upper limb
- Raynaud’s phenomenon
- Obliterative vascular disease
- Vasospasm
- Scleroderma
- Trauma
- Embolic ph
CONTRAINDICATIONS
- Recent myocardial infarction
- Anti-coagulated patients or those with coagulopathy
- Glaucoma
- Pre-existing contralateral phrenic nerve palsy ( may precipitate respiratory distress)
TECHNIQUES
1) Landmark technique
The patient is in a supine position with slight extension of the neck. The head is turned to the opposite side. The needle is introduced between the trachea and the carotid sheath at the level of the cricoid cartilage and Chassaignac’s tubercle (C6) to avoid any potential injury to the pleura. The sternocleidomastoid muscle and carotid artery are pushed laterally while simultaneously palpating the Chassaignac’s tubercle. The skin and subcutaneous tissue are pressed firmly onto the tubercle, the needle is directed medially and inferiorly towards the body of C6, to hit it and then withdrawn by 1-2 mm to rest outside the longus colli muscle. We inject 10 mls of 0.25% L-Bupivacaine after a small test dose of 0.5 mls and repeated negative aspiration for blood to rule out intravascular placement of the needle. Pain specialists use Bupivacaine (0.125-0.5%) or Ropivacaine, 0.2% in a volume ranging from 5-15mls depending upon their approach for the block, local guidelines and protocols and clinical judgement.
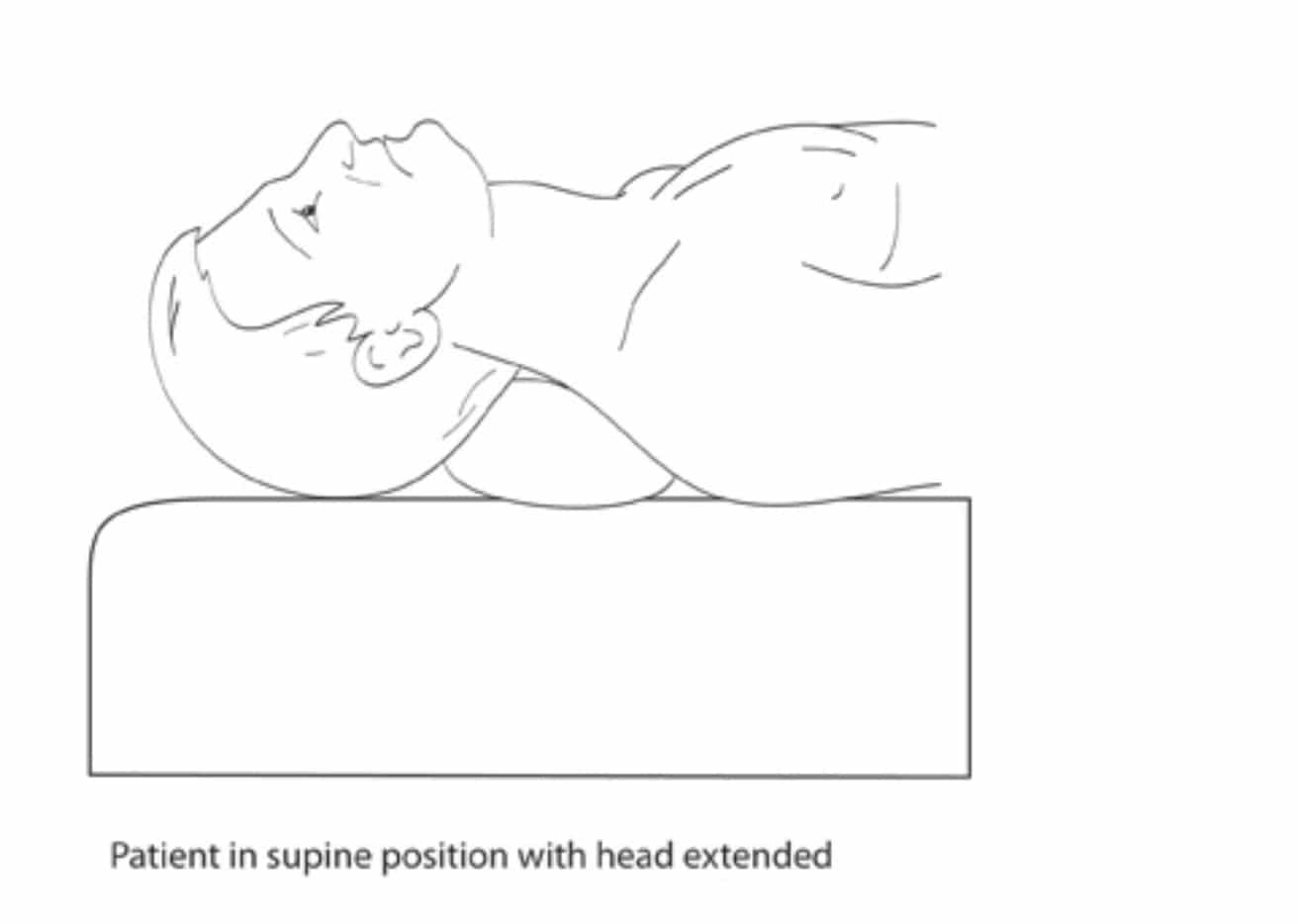
Figure 2. Patient position for performance of stellate ganglion block
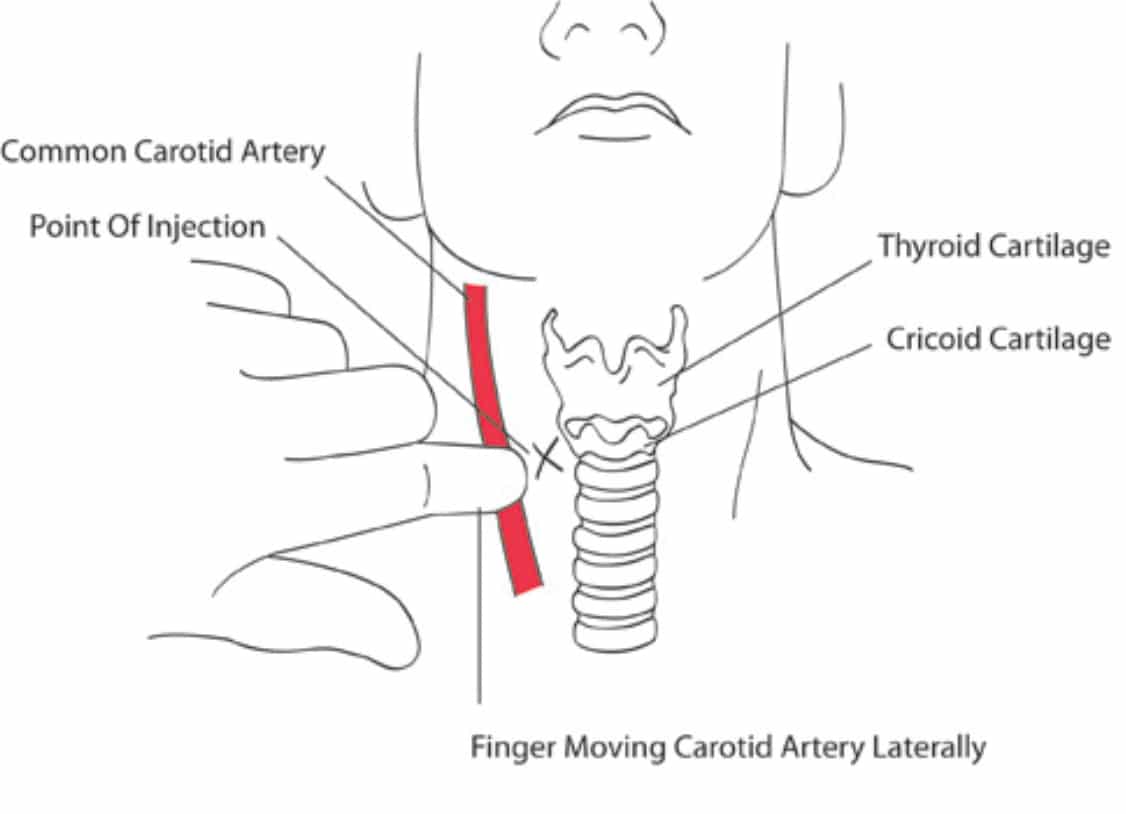
Figure 3. Stellate ganglion block using landmark technique
2) Fluoroscopy assisted
The anatomical landmarks are used to guide the approach and direction of the needle and then fluoroscopy is used to confirm its position. Radioopaque contrast is injected and the spread is visualised using anteroposterior and lateral views. Injection into the longus colli muscle is indicated by inability of the contrast medium to spread in-between the tissue planes while instantaneous disappearance indicates the presence of the needle in a vessel.
3) CT guided
The patient is supine with chin turned away from the injection site. The head of the first rib, adjacent vertebral artery and vein are identified and 25-gauge spinal needle is directed onto the head of the first rib, as close to the vertebral body as possible.
4) Ultrasound guided
The patient is in a supine position with slight extension of the neck. After cleaning and draping the site, the transducer is placed on the neck at the level of C6 to enable cross sectional visualization of anatomical structures. At this level, the carotid artery, internal jugular vein, thyroid gland, trachea, longus colli muscle, root of C6, and transverse process of C6 are identified. To retract the carotid artery laterally and to position the transducer close to the longus colli, the transducer is then gently pressed between the carotid artery and trachea. Using an in-plane approach, a 1.0-inch, 25-gauge long-bevel needle is inserted paratracheally toward the middle of the longus colli, The endpoint for injection is the ultrasound image demonstrating the tip of needle penetrating the prevertebral fascia in the longus colli. Following a negative aspiration test for blood or CSF, local anaesthetic is injected and visualised spreading in real time.
COMPLICATIONS
1) Horners syndrome : Is caused by sympathetic blockade and produces the following features on the ipsilateral side of the face :
- drooping of the eyelid (ptosis)
- constriction of the pupil (meiosis)
- decreased sweating of the face on the same side (anhydrosis)
- redness of the conjunctiva of the eye
- impression of an apparently sunken eyeball (enophthalmos)
This may also lead to increased amplitude of accommodation, paradoxical contralateral eyelid retraction, transient decrease in intraocular pressure and changes in tear viscosity. Although it may be considered a complication, the presence of Horner’s syndrome is a confirmatory sign of successful stellate ganglion blockade.
2) Misplaced needle puncturing important adjacent structures
- Vascular (which may lead to local haematoma or haemothorax)
- Carotid artery puncture
- Internal jugular vein puncture
- Inferior thyroid artery (serpentine artery) puncture during ultrasound guided approach
- Neurological
- Vagus nerve injury
- Brachial plexus root injury
- Others
- Pulmonary injury, pneumothorax
- Chylothorax (thoracic duct injury)
- Oesophageal perforation
3) Inadvertent spread of local anaesthetic
- Intravascular injection into Carotid artery, Vertebral artery, Internal jugular vein or Inferior thyroid artery
- Neuraxial/brachial plexus spread
- Localised spread
- Hoarseness due to recurrent laryngeal nerve injury
- Elevated hemidiaphragm from phrenic nerve blockade
4) Local anaesthetic toxicity
5) Infection
- Soft tissue abscess
- Meningitis
- Osteitis
Role of radiofrequency and pulsed radiofrequency
Pulsed radiofrequency (PRF), a variation of conventional continuous radiofrequency (CRF) is useful in the management of neuropathic pain. It seems to alter the synaptic transmission, producing a neuromodulatory effect, thus providing analgesia without causing any tissue destruction or painful after effects of CRF such as neuritis-like reactions, motor deficits, and the risk of deafferentation pain. A 2011 review of mechanisms of action and indications for pulsed radiofrequency as a treatment modality has provided us with strong evidence for its use in cervical radicular pain with a high safety margin in the treatment of a number of conditions such as discogenic pain, chronic inguinal herniorrhaphy pain, and chronic testicular pain. However, further clinical studies and randomized controlled trials are needed to substantiate its efficacy and appropriate treatment dose11.
CURRENT OPINION
Current preclinical evidence suggests the involvement of the sympathetic nervous system in the pathophysiology of CRPS. However, there is growing debate regarding its contribution to the clinical syndrome.
A 2002 meta-analysis on the effect of sympathetic blockade in patients with CRPS reported that up to 44% of patients having sympathetic blockade were expected to have no pain relief and only 29% reported complete pain relief5.
A 2008 literature review on sympathetic blocks graded stellate ganglion block as 1C evidence (strong recommendation, low-quality, or very low-quality evidence)3. The clinician needs to balance the benefits and risks in each individual case to justify its use.
A 2010 systematic review on local anaesthetic sympathetic blockade for CRPS revealed the scarcity of published evidence to support the use of sympathetic blockade as the ‘gold standard’ treatment for CRPS and raises questions as to its efficacy10.
Stellate ganglion block continues to be used as a treatment modality in the management of CRPS but further randomized controlled trials would be required to more confidently validate its usefulness.
ANSWERS TO QUESTIONS
- a, b, c and d are true. e is false – The vertebral artery lies medial to the ganglion.
- All answers are correct. Stellate ganglion block is indicated in the treatment of a number of chronic pain pathologies and vascular disorders of the upper limb by providing sympathetic blockade.
- The possible complications of stellate ganglion block : a, b and c are true while d and e are false. Stellate ganglion block causes sympathetic blockade leading to possible bradycardia. It also produces meiosis as part of Horner’s syndrome.
REFERENCES
- Gibbs GF, Drummond PD, Finch PM, Phillips JK. Unravelling the pathophysiology of complex regional pain syndrome: focus on sympathetically maintained pain. Clin Exp Pharmacol Physiol. 2008;35:717Y724.
- Schattschneider J, Binder A, Siebrecht D, Wasner G, Baron R. Complex regional pain syndromes: the influence of cutaneous and deep somatic sympathetic innervation on pain. Clin J Pain. 2006;22:240Y244.
- Day M. Sympathetic blocks: the evidence [review] [published correction appears in Pain Pract 2008;18(4):335Y336]. Pain Pract. 2008;8:98Y109.
- Boas RA. Sympathetic nerve blocks: in search of a role. Reg Anesth Pain Med 1998; 23: 292–305.
- Cepeda MS, Lau J, Carr DB. Defining the therapeutic role of local anesthetic sympathetic blockade in complex regional pain syndrome: a narrative and systematic review. Clinical Journal of Pain 2002;18(4):216-33.
- Wang JK, Johnson KA, Ilstrup DM. Sympathetic blocks for reflex sympathetic dystrophy. Pain 1985;23(1):13-17.
- Kozin F. Reflex sympathetic dystrophy: a review. Clinical and Experimental Rheumatology 1992; 10:401- 409.
- Price DD, Long S, Wilsey B. et al. Analysis of peak magnitude and duration of analgesia produced by local anaesthetics injected into sympathetic ganglia of complex regional pain syndrome patients. Clin J Pain 1998; 14:216-226.
- Fields HL, Baron R, Rowbotham MC. Peripheral neuropathic pain : an approach to management. In: Wall PD, Melzack R, eds. Textbook of Pain. London: Churchill Livingstone, 1999; 1523–9.
- M Soledad Cepeda, Daniel B Carr, Joseph Lau. Local anaesthetic sympathetic blockade for complex regional pain syndrome (Cochrane review), January 2010.
- Nicholas H. L.Chua, Kris C.Vissers, Menno E.Sluijter. Pulsed radiofrequency treatment in interventional pain management: mechanisms and potential indications—a review, Acta Neurochir (2011) 153:763–771.



