Paediatric Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Regarding the effects of lateral decubitus positioning on infants compared to older children and adults:
a. During both spontaneous and controlled ventilation in the lateral decubitus position, infants have optimal V/Q matching with the “healthy” lung in the dependent position
b. Infants have a highly compressible rib cage which leads to atelectasis of the dependent lung
c. Infants have an elevated hydrostatic pressure gradient between the dependent and non-dependent lung compared to adults
d. Infants have greater risk of developing significant hypoxemia in the lateral decubitus position compared to adults - Regarding the single lumen endotracheal tube method (mainstem intubation) for achieving lung isolation in infants and children:
a. It allows the ability to provide suction and CPAP to the operative lung
b. It allows bronchoscopic visualization of the operative lung
c. It does not require additional special equipment
d. It provides a better seal of bronchus compared to bronchial blockers or double lumen endotracheal tubes - Regarding the use of bronchial blockers in pediatric patients:
a. Blockers can be placed inside or outside of the endotracheal tube
b. The smallest tube through which a bronchial blocker can be placed is a 5mm internal diameter ETT
c. It is difficult to transition back to dual lung ventilation with a bronchial blocker
d. It is very rare for a bronchial blocker to migrate out of position
Key Points
- Lung isolation provides improved surgical conditions compared to conventional dual lung ventilation for thoracic surgery
- Due to physiologic differences, infants are more prone to the development of hypoxemia in the lateral decubitus position compared to older children and adults
- Options for lung isolation in the pediatric population include mainstem intubation, bronchial blockers, Univent tubes and double lumen endobronchial tubes
- Single lung ventilation creates a large intrapulmonary shunt therefore some degree of desaturation is to be expected
INTRODUCTION
With the advancement of technology and improvements in surgical equipment over the last few decades, thoracic surgery in infants and children has progressed significantly, especially in the area of minimally invasive methods.
There have been parallel advancements in pediatric airway equipment, including the development of pediatric sized bronchial blockers, Univent tubes, and double lumen tubes, providing the ability to isolate the lung in pediatric patients as small as full term neonates. Despite this widespread ability, there remain significant regional differences in the use of lung isolation techniques, some centers utilize lung isolation for most thoracic procedures, while other centers rarely isolate the lung in infants and children. Although both open and minimally invasive thoracic surgical techniques can be successfully completed without lung isolation, the ability to deflate the ipsilateral lung provides improved surgical conditions and optimal visualization of thoracic structures. An understanding of the different techniques for lung isolation in pediatric patients is important for any anesthetist taking care of this patient population.
DISEASES OF THE CHEST THAT MAY REQUIRE LUNG ISOLATION FOR SURGERY
Neonates and Infants
There are a multitude of congenital anomalies of the thoracic cavity that present in the neonatal period and require surgical intervention. Examples include tracheal stenosis, pulmonary sequestration, congenital diaphragmatic hernia, tracheoesophageal fistula, congenital lobar emphysema, and vascular ring to name a few. Although many of these diseases may be diagnosed prenatally by ultrasound, in parturients who do not have access to prenatal care, these issues often only become apparent after the delivery of a neonate presenting in severe respiratory distress.
Children and Adolescents
In contrast to the neonate, patients who develop thoracic disease later in childhood and adolescence typically present with infection, malignancy and musculoskeletal deformities. One of the most feared presentations seen in this age group is that of an anterior mediastinal mass, due to the potential complications of complete airway compression and cardiovascular collapse under general anesthesia. Regardless of the underlying pathology, it is critical that the anesthesiologist understand the patients’ underlying anatomy and how its distortion by mass or malformation may affect respiratory and cardiac physiology (see also ATOTW tutorial 320: Anterior mediastinal masses in paediatric anaesthesia)
PHYSIOLOGY OF SINGLE LUNG VENTILATION AND THE LATERAL DECUBITUS POSITION IN INFANTS AND CHILDREN
In the spontaneously breathing patient in the upright position, ventilation and perfusion (V/Q) are greatest in the dependent regions of the lung due to gravitational forces. Ventilation and perfusion are well matched allowing for maximal oxygenation. However, during thoracic surgery there are several factors that impair this relationship resulting in significant V/Q mismatch and susceptibility to hypoxemia. These factors include:
- Decreased Functional Residual Capacity (FRC). For example general anesthesia, neuromuscular blockade or mechanical ventilation
- Surgical retraction or single lung ventilation resulting in collapse of the operative lung
- Impairment of Hypoxic Pulmonary Vasoconstriction (HPV) due to inhalational agents and other anesthetic drugs
This alteration in V/Q matching seen with thoracic surgery is similar between infants, children and adults.
Effects of the Lateral Decubitus Position
The physiological changes that occur with lateral decubitus positioning are significantly different in infants (< 12 months of age) when compared to older children and adults. For older children and adults with unilateral lung disease, V/Q matching is optimal when the patient is placed in the lateral decubitus position with the “healthy” lung in a dependent position. This holds true for both spontaneous and controlled ventilation. Due to gravitational forces, this position results in increased perfusion to the “healthy”, dependent lung and decreased perfusion to the diseased, non-dependent lung. As the diseased lung has impaired ventilation at baseline, this preferential distribution of blood flow away from the diseased lung results in optimal V/Q matching.
However, in the infant the lateral decubitus position has significantly different effects on pulmonary physiology. For infants with asymmetric lung disease, the position which provides optimal oxygenation is with the “healthy” lung in the non-dependent position, which is unfortunately opposite of the positioning required for surgery. There are three main reasons for the observed difference between infants and older children/adults:
- Infants have an easily compressible rib cage, which cannot fully support the dependent lung. This anatomical feature predisposes infants to the development of atelectasis of the dependent lung at normal tidal breathing.
- Due to their small size, infants have a reduced hydrostatic pressure gradient between the dependent and nondependent lungs as compared to older children and adults; therefore, the beneficial increase in perfusion to the dependent, ventilated lung is diminished.
- In older children and adults, the dependent diaphragm has a mechanical advantage as it is “loaded” by the abdominal hydrostatic pressure gradient. This pressure gradient is significantly decreased in infants, thereby reducing the functional advantage of the dependent diaphragm.
For the above reasons, the infant is at significantly higher risk of developing hypoxemia during surgery in the lateral decubitus position compared to older children and adults.3,6
TECHNIQUES FOR LUNG ISOLATION IN INFANTS AND CHILDREN
Manual Retraction
A common method employed to obtain adequate exposure for thoracic surgery in infants and small children is by open thoracotomy with manual retraction of the operative lung performed by the surgeon. This method allows for conventional dual lung ventilation with the use of a standard endotracheal tube. Manual retraction can also be used during Video Assisted Thoracoscopic Surgery (VATS) allowing for two lung ventilation; however single lung ventilation is more desirable as it is associated with better visualization of thoracic structures, a greater amount of space for the safe placement of cameras and equipment, and a decrease in the incidence of lung injury from the use of retractors7.
Single Lung Ventilation Techniques
Single Lumen Endotracheal Tubes
The use of a single lumen endotracheal tube (ETT) is viewed as the simplest method of obtaining lung isolation. The technique for placement is straightforward and can be accomplished with standard intubating equipment. With this technique, the ETT is advanced into the mainstem bronchus of the lung to be ventilated until breath sounds on the operative side disappear. The ETT can be advanced blindly, or a fiberoptic bronchoscope can be used to guide or confirm placement. The angulation of the right mainstem bronchus is significantly less acute than that of the left (25 degrees versus 45 degrees respectively), making the right side technically easier for mainstem intubation. When attempting to advance the ETT into the left mainstem bronchus, the bevel of the ETT should be rotated 90-180 degrees to the left and the patient’s head should be turned to the right, which helps to align the trachea and the left mainstem bronchus. When using a cuffed ETT, the distance from the tip of the tube to the proximal cuff must be shorter than the length of the bronchus in order to ensure the entire cuff is within the bronchus and the upper lobe orifice is unobstructed.
Balloon Tipped Bronchial Blockers
There are multiple balloon-tipped bronchial blockers available including the Arndt endobronchial blocker (Figure 1), Fogarty embolectomy catheter, Cohen endobronchial blocker, Coopdech bronchial blocker, and EZ-blocker. Although each blocker has minor differences, the overall structure and technique for lung isolation is similar. The blocker is placed within or next to a single lumen endotracheal tube and advanced under direct visualization utilizing fiberoptic bronchoscopy into the mainstem bronchus of the operative lung. The cuff is then inflated under direct vision to ensure adequate positioning and seal of the bronchus.
One of the most common blockers used in the pediatric population in the United States is the Arndt endobronchial blocker; the stepwise technique for its placement is described below:
- Single lumen endotracheal tube placed at midtracheal position.
- Blocker placed through multi-port adapter.
- Fiberoptic bronchoscope placed through multiport adapter and advanced through wire-loop of blocker.
- Coupled bronchoscope and blocker advanced to the side of the lung to be blocked (A).
- Once in mainstem bronchus, bronchoscope is withdrawn and the cuff inflated under direct vision (B).
- Fiberoptic bronchoscope removed entirely.
- Wire loop can be retracted so that open-tipped blocker can be utilized for suction or CPAP.
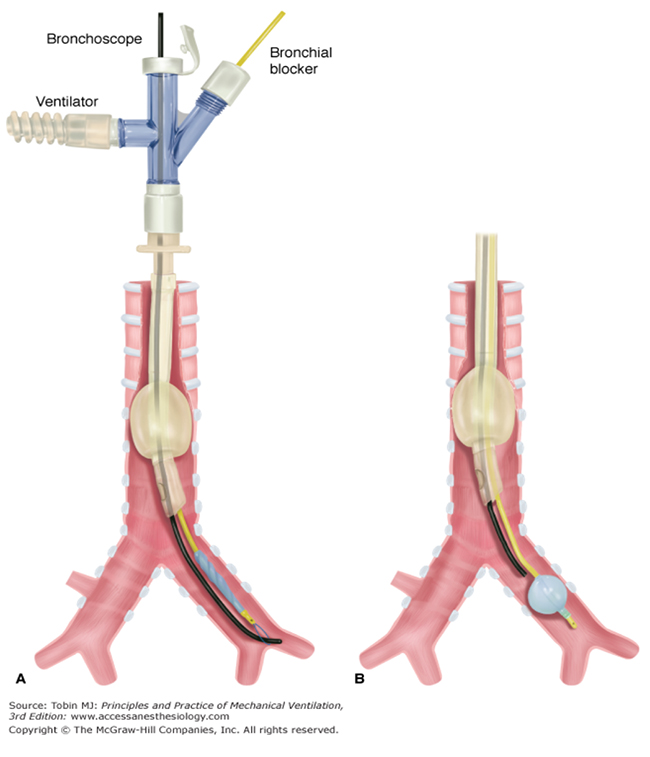
Figure 1: The Arndt endobronchial blocker showing placement with fiberoptic bronchoscope (A) followed by cuff inflation under direct vision (B)
The smallest sized Arndt endobronchial blocker available is 5 French with an outer diameter of 2.5mm. The smallest fiberoptic bronchoscope available is typically ~ 2.2mm, therefore the smallest endotracheal tube (ETT) through which the blocker can be placed is 5mm ID. For pediatric patients who require smaller ETTs, the blocker can be placed external to and alongside the single lumen ETT.

Univent Tubes
Univent tubes utilize a similar methodology for lung isolation as bronchial blockers; however the structure of the tube is slightly different. The Univent tube consists of a standard endotracheal tube with an additional lumen directly attached to the side of it (Figure 2). Within this side lumen is a balloon-tipped tube that can be advanced into a bronchus and used as a bronchial blocker. There is a large discrepancy between the internal diameter of the endotracheal tube portion and the total outer diameter of entire Univent tube (i.e.: Univent tube with 3.5mm internal diameter has an external total cross-sectional diameter of 7.5-8.0mm), which results in a high resistance to gas flow.
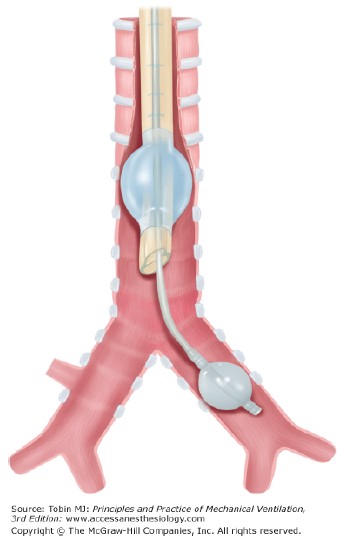
Figure 2: The Univent endotracheal tube with endobronchial blocker in situ. Note the additional lumen of the Univent tube, through which a balloontipped endobronchial blocker can be advanced.
Double Lumen Tubes (DLTs)
Double lumen tubes consist of two cuffed tubes of differing length that are molded together (Figure 3). The shorter tube ends in the trachea and the longer tube ends in either bronchus. The technique for placement of double lumen tubes is fairly simple. After the tip of the tube is passed through the vocal cords, the stylet is removed, and the tube is then rotated 90 degrees to the appropriate side and advanced further into the bronchus until resistance is met. Placement is verified with auscultation and fiberoptic bronchoscopy. In recent years, smaller sized double lumen tubes have become available and currently the smallest is a 26 French size which can be used in pediatric patients as young as 8 years old.
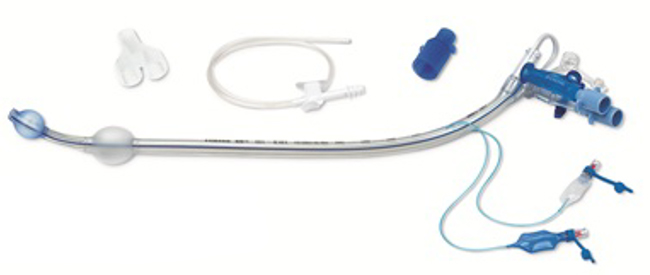
Figure 3: An example of a double lumen tube (DLT) showing both the endotracheal and endobronchial cuffs inflated (additional connectors and suction catheter also shown)

If mechanical ventilation is required postoperatively, the DLT is usually changed to a single lumen ETT. This is a significant disadvantage in patients with a known difficult airway or in prolonged operations in which the airway may be edematous making reintubation challenging.
SIZING OF TUBES USED FOR SINGLE LUNG VENTILATION
The table below shows the recommended sizing of each of the techniques described above, according to the age of the infant or child. As with all equipment, however, correct sizing will vary between patients of the same age and so it is essential to have a range of sizes available.
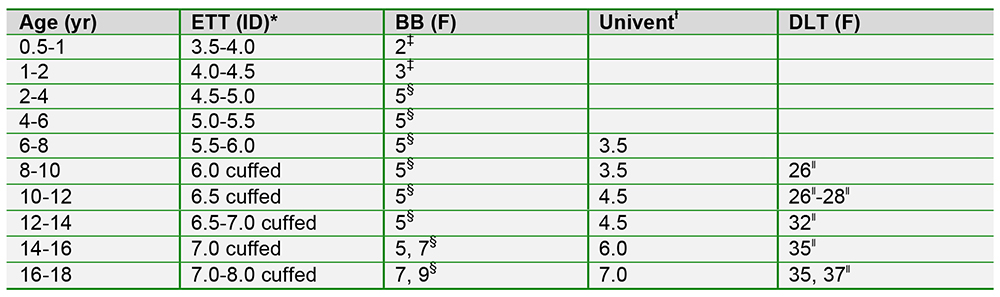
Key: BB, Bronchial blocker; DLT, double-lumen tube; ETT, endotracheal tube; F, French size; ID, internal diameter.
*Sheridan tracheal tubes, Hudson RCI, Arlington Heights, Ill.
ƚFuji Systems Corporation, Tokyo.
‡Edwards Lifesciences LLC, Irvine, Calif.
§Cook Medical, Inc., Bloomington, Ind.
॥Covidien, Mansfield, Mass.
Cote C, Lerman J, Anderson BJ. Chapter 13: Anesthesia for Thoracic Surgery. A Practice of Anesthesia for Infants and Children 5th Edition. Elsevier Saunders. 277-290. Philadelphia, PA. 2013.
TROUBLESHOOTING HYPOXEMIA DURING SINGLE LUNG VENTILATION
Single lung ventilation creates a large intrapulmonary shunt as the non-ventilated lung is still being perfused without oxygenation; therefore, some degree of desaturation is to be expected. Patients with chronic lung disease of the operative lung typically tolerate single lung ventilation better than patients with two healthy lungs. In the former situation, perfusion to the diseased lung is chronically decreased and preferentially guided to the healthy lung; therefore, when single lung ventilation is initiated there is a smaller intrapulmonary shunt fraction.
In the setting of hypoxemia, a methodical evaluation of potential causes should be sought. The following are basic steps that can be taken sequentially to evaluate and treat hypoxemia during single lung ventilation:
- Place patient on 100% FIO2.
- Evaluate position of tube or blocker by auscultation for breath sounds or via fiberoptic bronchoscopy if available. The placement of the tube or blocker should be verified after every change in patient positioning.
- Apply CPAP to the non-dependent lung. A level of CPAP between 5-10mmHg typically does not interfere with surgical conditions and will help to decrease shunt fraction.
- Apply low levels of PEEP to the dependent lung. Avoid high levels of PEEP as this can divert blood flow away from the dependent lung due to increases in intrathoracic pressure.
- Pass soft suction catheter down tube to ensure there is no obstruction from secretions.
- Lastly, if hypoxemia remains after the above steps have been taken, the surgeon should be informed and the operative lung should be re-inflated.
Answers to MCQ questions
- ..
a) False: In the lateral decubitus position V/Q matching is optimized in infants with the “healthy” lung in the non- dependent position
b) True
c) False: Infants have a lower hydrostatic pressure gradient between the dependent and non-dependent lungs compared
to adults
d) True - ..
a) False: With the single lumen ETT technique, one cannot provide CPAP or suction to the operative lung
b) False: With the single lumen ETT technique, there is no method to visualize the operative lung
c) True
d) False: The single lumen ETT technique typically provides a less adequate seal of the bronchus compared to bronchial blockers/Univent tube/DLT, especially when smaller, uncuffed ETTs are used - ..
a) True
b) True
c) False: It is simple to transition from single lung ventilation to dual lung ventilation with a bronchial blocker. The balloon of the blocker is deflated and the blocker can be easily removed from the airway resulting in ventilation of both lungs
d) False: It is common for bronchial blockers to become dislodged or migrate after placement. If they migrate proximally this can result in complete airway obstruction and the inability to ventilate as the blocker balloon will be inflated in the trachea and impede ventilation
REFERENCES and FURTHER READING
- Bair AE, Doherty MJ, Harper R, Albertson TE. An Evaluation of a Blind Rotational Technique for Selective Mainstem Intubation; Academy of Emergency Medicine; 2004; Vol 11; 1105-1107.
- Bird GT, Hall M, Nel L, Davies E, Ross O. Effectiveness of Arndt endobronchial blockers in pediatric scoliosis surgery: a case series, Pediatric Anesthesia; Vol 17; 289-294.
- Cote C, Lerman J, Anderson BJ. A Practice of Anesthesia for Infants and Children 5th Edition. Elsevier Saunders. 277-290. Philadelphia, PA. 2013.
- Cohen DE, McCloskey JJ, Motas D, Archer J, Flake AW. Fluoroscopic-assisted endobronchial intubation for single-lung ventilation in infants; Pediatric Anesthesia; 2011; Vol 21; 681-684.
- Fabila TS, Menghraj SJ. One lung ventilation strategies for infants and children undergoing video assisted thoracoscopic surgery; Indian Journal of Anaesthesia; 2013; Vol 57 Issue 4; 339-344.
- Hammer GB. Single-lung ventilation in infants and children. Pediatric Anesthesia; 2004; Vol 14; 98-102.
- Hammer GB. Pediatric Thoracic Anesthesia. Anesthesia & Analgesia; 2001; Vol 92; Issue 6; 1449-1464.
- Hammer GB, Fitzmaurice BG, Brodsky JB. Methods for Single-Lung Ventilation in Pediatric Patients. Anesthesia and Analgesia; 1999; Vol 89; 1426-1429.
- Shah R, Reddy AS, Dhende NP. Video assisted thoracic surgery in children. Journal of Minimal Access Surgery; 2007; 3(4); 161-167.
- Tobin, M. Principles and Practice of Mechanical Ventilation. McGraw Hill. 2013.
- Scrace B, McGregor K. Anterior mediastinal masses in paediatric anaesthesia. Anaesthesia Tutorial of the Week; Tutorial 320; 2015. Available at http://resources.wfsahq.org/resources/virtual-library
Images in this tutorial have been reproduced with kind permissions from Smiths Medical ASD and McGraw-Hill ducational Materials



