Patient Safety
KEY POINTS
• Asleep tracheal intubation of critically ill patients is associated with a small but significant mortality. Peri-intubation cardiac arrest occurs in ~ 3% of patients of whom 30% to 45% fail to be resuscitated.
• Awake intubation offers theoretical physiologic and safety benefits over asleep approaches including avoiding pharmacologic induction agents and abrupt shifts in intrathoracic pressure, while maintaining respiratory drive.
• Hypoxemic patients are at increased risk of intubation-related complications. Optimal preoxygenation, apneic oxygenation, and awake intubation are important optimization strategies.
• Hypotensive patients are at risk of peri-intubation hemodynamic collapse. Optimization strategies include physiologic fluid resuscitation, careful pharmacologic agent selection, and readily available vasopressors and inotropes.
• Metabolic acidosis may result in apnea intolerance and peri-intubation hemodynamic collapse.
• Many other pathophysiologic processes result in a physiologically difficult airway. Awake, spontaneously breathing intubation should be considered in the physiologically difficult airway.
INTRODUCTION
The term “difficult airway” has traditionally been used in reference to anatomic airway characteristics rendering the successful placement of an endotracheal tube difficult for experienced practitioners. Although anatomy certainly plays a role in difficult airway management, contextual and physiologic factors such as time pressure, environment, and patient’s hemodynamic status and/or respi– ratory reserve all contribute to the complexity of safely securing an airway and optimizing gas exchange; the ultimate goals of tracheal intubation (TI). These contextual factors are most exaggerated in critically ill patients, who are at high risk of peri-intubation complica– tions including hypoxemia, hemodynamic instability,¹ and cardiac arrest. Cardiac arrest occurs in as many as 3.1% of TI patients, of whom up to 47.3% do not achieve return of spontaneous circulation.²,³ Patients at high risk of peri-TI hypoxemia and/or hemodynamic collapse can be thought of as having a ‘physiologically difficult airway’, where despite reassuring (or not) airway anatomy, safely securing the airway remains difficult due to ongoing physiologic derangements. In this tutorial, we will discuss specific conditions and examples of physiologically difficult airways—hypoxemia, hypotension, severe metabolic acidosis, and more—along with strategies to mitigate the risk of complications associated with airway management.
HYPOXEMIA
Hypoxemic respiratory failure is a commonly encountered indication for TI.³ This state is associated with either ventilation-perfusion mismatch, impaired gas transfer and diffusion, and/or physiologic or anatomic shunts, whereby blood does not adequately participate in alveolar gas exchange before entering systemic circulation. Common culprit conditions in the critically ill include pneumonia, atelectasis, acute respiratory distress syndrome, pulmonary oedema, and mucous plugging.⁴ Comorbidities such as obesity and impaired level of consciousness are commonly encountered in critically ill patients, and are associated with reduced functional residual capacity (FRC), further reducing respiratory reserve. These factors are acutely worsened with the induction of anaesthesia for TI,⁵ resulting in an increased risk of potentially life threatening TI-related complications,² especially when $ 2 attempts are required to secure the airway.³,⁶ Table 1 summarizes peri-TI recommendations for hypoxemic patients.

Preoxygenation
Optimal preoxygenation extends the time prior to oxyhemoglobin desaturation in patients undergoing TI, reducing the frequency of desaturation and allowing more time for airway manipulation, thereby potentially increasing the likelihood of first-pass success and reducing the risk of complications.⁶ Preoxygenation with a non-rebreather mask, both with an effective seal and set at flush flow rates (50-70 L/min), has been shown to be noninferior to preoxygenation with bag-mask-valve ventilation (BMV) at flush flow rates of 15 L/min.⁷ In the case of ineffective seal resulting in dilution of inspired oxygen with room air, the addition of a nasal cannula (NC) at 10 L/min to the primary method of preoxygenation will increase end-tidal oxygen.⁸ Preoxygenation in the head-up position at 20 to 30 degrees will increase FRC, thus optimizing the effectiveness of preoxygenation and prolonging time to desaturation.⁹ This intervention must be balanced against the patient’s hemodynamic conditions and their ability to tolerate this position. Preoxygen- ation with noninvasive positive pressure ventilation (NIPPV) is recommended for patients with shunt physiology,⁵,¹⁰ and has been associated with reduced peri-TI hypoxia compared to non-rebreather mask.¹¹ Preoxygenation with high-flow nasal oxygen (HFNO) cannula has been shown to be noninferior to NIPPV in preventing desaturation when left in place for apneic oxygenation.¹²
Patients with oxyhemoglobin saturation < 93% not responsive to the above techniques are likely to have significant shunt physiology. In such instances, NIPPV, using either a mechanical ventilator or manually assisted BVM using 5 to 10 cm H2O of positive end-expiratory pressure, is the logical next step in escalating preoxygenation efforts as it reduces ventilation-perfusion mismatch and improves FRC. Additionally, inhaled pulmonary vasodilators (eg, nitric oxide) have been recommended to reduce ventilation-perfusion mismatch and improve oxygenation.⁵
A logical approach to optimize preoxygenation is to select an oxygen delivery method based on the severity of the patient’s hypoxemia. Hypoxemia can be graded by the ratio of arterial oxygen partial pressure to the fraction of inspired oxygen (PaO2/FiO2), with a lower value representing more severe hypoxemia. BMV or HFNO have been recommended for a baseline PaO2/FiO2 of 300 to 200, NIPPV for 200 to100, and consideration of an awake, spontaneously breathing, intubation approach for PaO2/FiO2 < 100.¹³
Apneic Oxygenation
Apneic oxygenation is the application of supplemental oxygen without variable positive pressure, typically via nasal cannula, to an apneic patient. Some (unpredictable) amount of gas exchange is achieved via dead-space flushing and turbulent gaseous mixing. This can be achieved using a standard NC at 10 to 15 L/min and is effective at prolonging time to desaturation.¹⁴ Newer HFNO cannulae deliver heated and humidified oxygen at rates of 40 to 70 L/min, and have been shown to increase apnea time without desaturation and reduce the rate of carbon dioxide accumulation via the same mechanisms.¹⁵ HFNO reduces the rate of carbon dioxide accumulation to a greater extent than standard NC. The combination of NIPPV for preoxygenation and HFNO for apneic oxygenation has been shown to maintain oxygen saturation better than NIPPV alone for patients with severe hypoxia.¹⁶ In the critically ill population, HFNO and NC have not been directly compared. It may be implied that NC would underperform as HFNC has been shown to significantly prolong time to desaturation in the operating room, and the reduced rate of carbon dioxide accumulation compared to NC reflects an increase in gas exchange.¹⁵
Refractory Hypoxemia
Anxious or delirious patients may be unable to tolerate preoxygenation. In such cases, non–respiratory depressant sedatives (eg, ketamine) can be considered prior to preoxygenation to improve patient compliance and the effectiveness of preoxygenation.⁵,¹⁰ Cautious pharmacologic titration is warranted with this approach to avoid apnea that can occur with even small sedative doses, and the provider should be prepared to intubate urgently in case of desaturation.
In patients with dramatically altered gas exchange and FRC, rapid desaturation may occur the moment supplemental oxygenation and ventilation are removed. In these cases, several guidelines (including the American Society of Anesthesiologists¹⁷ recently updated difficult airway guidelines), recommend practitioners to strongly consider an “awake” TI approach, maintaining oxygenation and spontaneous ventilation during the intubation process.⁵,¹⁴,¹⁷
Rapid Sequence Intubation versus BMV
A recent study compared the use of BMV during the apneic interval between induction of anesthesia and TI to traditional rapid sequence intubation (RSI) without BMV in critically ill adults.¹⁸ BMV resulted in higher oxygen saturations and reduced the incidence of desaturations to < 80%. This trial also suggests that BMV may be more effective than apneic oxygenation at maintaining oxygen saturation in the critically ill. The study, however, was not designed to evaluate safety outcomes, notably tracheobronchial aspiration. Patients with obvious contraindications to BMV (eg, bowel obstruction) were excluded. Within these limitations, the results challenge the dogma that BMV should be avoided in critically ill patients.
Point-of-Care Ultrasound
Time permitting, point-of-care ultrasound (POCUS) may help clarify the etiology of hypoxemia and guide therapy.¹⁹
HYPOTENSION
Patients requiring mechanical ventilation often present with hypotension. Distributive or hypovolemic shock states are associated with decreased venous return, which is exacerbated by vasodilation secondary to anaesthetic induction agents, muscle relaxation, and the abrupt transition to positive pressure ventilation. Pre-TI hypotension, defined as systolic blood pressure < 90 mmHg, has been associated with a 12% to 15% incidence of peri-TI cardiac arrest in emergency intubations,²⁰ of which return of spontaneous circulation was achieved in only 53% of patients. Peri-TI cardiac arrest carries a 14-fold increased risk of in-hospital mortality. A shock index (heart rate divided by systolic blood pressure) > 0.8 has been independently associated with increased risk of peri-TI cardiac arrest.¹⁹ Even 10 minutes of hypotension can result in poor outcomes for high-risk patients.⁵ Given the severity and frequency of these complications, hypotensive patients require aggressive hemodynamic optimization prior to TI. Table 2 summarizes peri-TI recommendations for hypotensive patients.

Volume Administration
Ad hoc administration of a 500-mL crystalloid bolus prior to TI has been recently shown to not significantly alter the rate of peri-TI cardiovascular collapse.²¹ A more physiologic approach is to administer fluids to patients who are volume responsive.⁵,¹⁰ Volume responsiveness can be evaluated using bedside techniques including arterial waveform analysis, POCUS assessment of inferior vena cava respiratory variation (which has variable accuracy), and passive leg raise.¹⁰,¹⁸
Vasopressor and Inotropic Support
In hypotensive patients unresponsive or intolerant to fluid administration, prophylactic vasopressor and/or inotrope infusions are recommended.⁵,¹⁰ When unable to initiate infusions, bolus-dosed vasopressors or inotropes should be immediately available and used to support hemodynamics during TI. The agent(s) choice should be tailored to the individual patient and the suspected hypotension etiology.
Choice of Induction Agent
The majority of anesthesia induction agents may acutely worsen hypotension in critically ill patients. Propofol and benzodiazepines lead to myocardial depression and vasodilation,⁵ and propofol administration has been independently associated with hemodynamic instability in emergency TI.³ Alternatives include ketamine and etomidate, though each have potential caveats. Literature is mixed regarding the safety of etomidate. There are concerns regarding adrenal suppression in septic patients; however, some studies have shown no harm when used solely for induction.⁵ Ketamine and etomidate have been directly compared, with some studies showing similar overall complication rates. The Etomidate Versus Ketamine trial²² showed that patients induced with etomidate had a higher risk of 7-day mortality compared to ketamine, but the risk converged to similar overall mortality at day 28. Interestingly, ketamine had a greater association with postinduction cardiovascular collapse (25%) compared to etomidate (17.4%), a difference of 7.6% (95% confidence interval: 2%-13%). Ketamine is also associated with laryngospasm, airway secretions, myocardial depression, myocardial ischemia, and cardiac arrest.⁵ Ultimately, given the lack of definitive evidence, the choice of induction agent should be based on the clinician’s comfort level and the individual patient’s conditions.
POINT-OF-CARE ULTRASOUND
POCUS can be useful in identifying important physiological variables in the hypotensive patient, including the presence of left ventricle (LV) and/or right ventricle (RV) dysfunction, significant valvular abnormalities, pericardial effusion, and fluid responsiveness/tolerance. This information can assist in guiding peri-TI resuscitative efforts.¹⁹
SEVERE METABOLIC ACIDOSIS
Severe acidosis can cause seizure, coma, arrhythmia, and cardiac arrest. The risk is particularly elevated at arterial pH < 7.00.²³ While respiratory acidosis may be improved with increased ventilation, metabolic acidosis can be severely worsened during the peri-TI period. Patients with metabolic acidosis often hyperventilate in an effort to normalize their pH; hence, even a brief apneic period can cause carbon dioxide to accumulate, further lowering the pH, thereby potentially triggering cardiac arrest.¹⁰,²⁴ Notably, hemodynamic instability induced by induction agents may also contribute to worsened acidosis through reduced peripheral perfusion. Table 3 summarizes peri-TI recommendations for acidemic patients.

Optimization of Metabolic Acidosis
In patients with severe metabolic acidosis, NIPPV may be used to assist ventilation and preoxygenation, and to measure respiratory parameters, predicting the required minute ventilation. In severe cases, it is possible that a high minute ventilation requirement may not be achievable by a mechanical ventilator. In these cases, intubation should be avoided or delayed when possible, and NIPPV used to support hyperventilation until treatment of the underlying cause of acidosis is initiated.⁵,¹⁰
When intubation cannot be delayed, maintenance of spontaneous respiration during and after intubation should be considered.⁵,¹⁰ Some authors recommend avoiding RSI, or using short-acting neuromuscular blocking agents (eg, succinylcholine) when RSI is required.⁵ HFNO used for apneic oxygenation can slow accumulation of carbon dioxide via dead-space flushing.¹⁵ Induction agents should be chosen and carefully titrated to avoid further hemodynamic deterioration.⁵,¹⁰ After intubation, a supported spontaneous ventilation strategy should be considered to allow the patient to set their minute ventilation and maintain respiratory compensation. Care should be taken to avoid air trapping given the high tidal volumes and respiratory rates these patients often require.
Pre-TI bicarbonate boluses in patients with very high minute ventilation remain a subject of debate.⁵
OTHER PHYSIOLOGICALLY DIFFICULT AIRWAYS
Hypoxia, hypotension, and acidosis are common presentations associated with a physiologically difficult airway; however, there are many more pathologies associated with high-risk airway management. Detailed discussion of all possible physiologically difficult airways is beyond the scope of this tutorial; however, key considerations in some critical conditions are presented below.
RV Failure
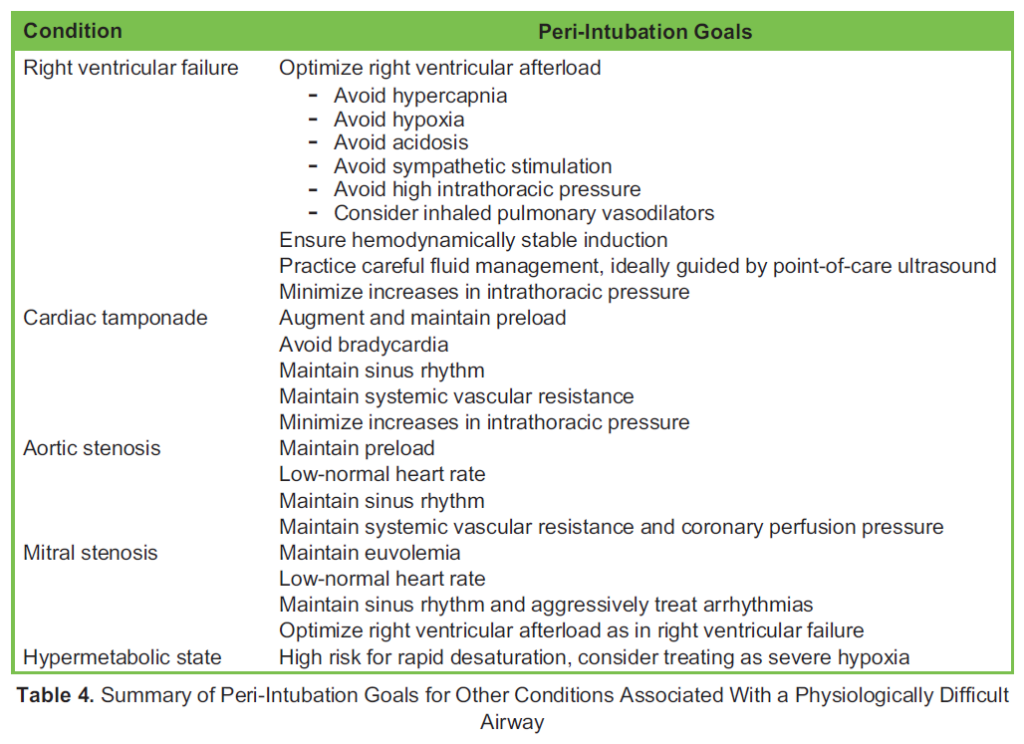
A failing RV is exquisitely sensitive to changes in preload and afterload occurring during TI. Hypotension secondary to induc- tion agents and increased thoracic pressure from mechanical ventilation can both lead to reduced venous return and RV preload. Hypoxia, hypercarbia, increased sympathetic tone, and TI-induced increased intrathoracic pressure can increase RV afterload. Patients with suspected RV failure warrant POCUS evaluation whenever possible, careful fluid management, and consideration of inhaled pulmonary vasodilators. Intubation strategies (awake or asleep) should be chosen to avoid hypoxia, hypercapnia, acidosis, sympathetic stimulation, and hemodynamic instability.⁵,¹⁰,¹⁸
Cardiac Tamponade
Cardiac tamponade results in elevated LV filling pressures, impairing diastolic filling, and resulting in a fixed stroke volume, and thus rate-dependent cardiac output, which may deteriorate further with the reduced preload, systemic vascular resistance, and increased intrathoracic pressure occurring during TI. When possible, pericardial drainage should occur prior to TI in hemodynamically unstable patients. When TI is required, strategies (awake or asleep) should be tailored to preserve spontaneous ventilation, augment and maintain preload, avoid bradycardia, maintain sinus rhythm, maintain systemic vascular resistance, maintain myocardial contractility, and minimize increases in intrathoracic pressure.²⁵
Aortic Stenosis
Patients with severe aortic stenosis have a fixed obstruction to LV outflow, resulting in an impaired ability to increase cardiac output to compensate for physiologic stress, particularly systemic vasodilation. LV hypertrophy and diastolic dysfunction result in increased myocardial oxygen demand and tenuous oxygen supply, which may result in myocardial ischemia and rapid hemodynamic deterioration on induction of anaesthesia and TI. Intubation strategies should be tailored to maintain adequate preload and LV filling pressures, maintain low-normal heart rate to allow diastolic filling and coronary perfusion, maintain sinus rhythm, maintain contractility, and maintain elevated systemic vascular resistance to increase coronary perfusion.²⁶
Mitral Stenosis
A stenotic mitral valve impairs LV diastolic filling and increases left atrial, pulmonary arterial, and RV pressures, eventually resulting in arrhythmia, pulmonary oedema, pulmonary hypertension, and RV failure. Intubation strategies should be tailored to maintain a low-normal heart rate to maximize LV filling time, maintain hemodynamic stability, avoid (and aggressively correct) atrial fibrillation, maintain euvolemia, and avoid precipitating RV failure as discussed above.₂₅
Hypermetabolic States
Hypermetabolic states, such as those seen in sepsis, thyrotoxicosis, acute respiratory distress syndrome, burn patients, etc, result in elevated oxygen consumption, elevated carbon dioxide accumulation, and thus impaired apnea tolerance.²³ Similar strategies to the peri-TI management of hypoxemic patients—optimal preoxygenation, apneic oxygenation, and awake intubation—may be considered.
Others
Though outside the scope of this tutorial, other conditions that deserve mention are anterior mediastinal mass, elevated intra- cranial pressure, severe cardiac valve insufficiency, hypertrophic obstructive cardiomyopathy, etc. Table 4 summarizes recommendations for the conditions discussed above.
SUMMARY
Although traditional use of the term ‘difficult airway’ generally refers to anatomical factors resulting in difficult airway management, many pathophysiologic states place patients at high risk of morbidity and mortality when undergoing TI. These patients should be considered to have a physiologically difficult airway, and require careful optimization of their condition along with consideration of alternative management strategies, most importantly awake endotracheal intubation, to mitigate the risk of intubation related complications.
REFERENCES
- Perbet S, De Jong A, Delmas J, et al. Incidence of and risk factors for severe cardiovascular collapse after endotracheal intubation in the ICU: a multicenter observational study. Crit Care. 2015;19(1):257.
- De Jong A, Rolle A, Molinari N, et al. Cardiac arrest and mortality related to intubation procedure in critically ill adult patients: a multicenter cohort study. Crit Care Med. 2018;46(4):532-539.
- Russotto V, Myatra SN, Laffey JG, et al. Intubation practices and adverse peri-intubation events in critically ill patients from 29 countries. JAMA. 2021;325(12):1164-1172.
- Bhutta BS, Alghoula F, Berim I. Hypoxia. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2022.
- Kornas RL, Owyang CG, Sakles JC, et al. Evaluation and management of the physiologically difficult airway: consensus recommendations from Society for Airway Management. Anesth Analg. 2021;132(2):395-405.
- Davis DP, Lemieux J, Serra J, et al. Preoxygenation reduces desaturation events and improves intubation success. Air Med J. 2015;34(2):82-85.
- Driver BE, Klein LR, Carlson K, et al. Preoxygenation with flush rate oxygen: comparing the nonrebreather mask with the bag-valve mask. Ann Emerg Med. 2018;71(3):381-386.
- Hayes-Bradley C, Lewis A, Burns B, et al. Efficacy of nasal cannula oxygen as a preoxygenation adjunct in emergency air- way management. Ann Emerg Med. 2016;68(2):174-180.
- Dixon BJ, Dixon JB, Carden JR, et al. Preoxygenation is more effective in the 25 degrees head-up position than in the supine position in severely obese patients: a randomized controlled study. Anesthesiology. 2005;102(6):1110-1115.
- Mosier JM, Joshi R, Hypes C, et al. The physiologically difficult airway. West J Emerg Med. 2015;16(7):1109-1117.
- Baillard C, Fosse JP, Sebbane M, et al. Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med. 2006;174(2):171-177.
- Frat JP, Ricard JD, Quenot JP, et al. Non-invasive ventilation versus high-flow nasal cannula oxygen therapy with apnoeic oxygenation for preoxygenation before intubation of patients with acute hypoxaemic respiratory failure: a randomised, multicentre, open-label trial. Lancet Respir Med. 2019;7(4):303-312.
- Russotto V, Rahmani L, Parotto M, et al. Tracheal intubation in the critically ill patient. Eur. J. Anaesthesiol. 2022;39(5):463-472.
- Wimalasena Y, Burns B, Reid C, et al. Apneic oxygenation was associated with decreased desaturation rates during rapid sequence intubation by an Australian helicopter emergency medicine service. Ann Emerg Med. 2015;65(4):371-376.
- Patel A, Nouraei SA. Transnasal humidified rapid-insufflation ventilatory exchange (THRIVE): a physiological method of increasing apnoea time in patients with difficult airways. Anaesthesia. 2015;70(3):323-329.
- Jaber S, Monnin M, Girard M, et al. Apnoeic oxygenation via high-flow nasal cannula oxygen combined with non-invasive ventilation preoxygenation for intubation in hypoxaemic patients in the intensive care unit: the single-centre, blinded, rando- mised controlled OPTINIV trial. Intensive Care Med. 2016;42(12):1877-1887.
- Apfelbaum J, Hagberg C, Connis R, et al. 2022 American Society of Anesthesiologists practice guidelines for management of the difficult airway. Anesthesiology 2022; 136(1):31-81.
- Casey J, Janz D, Russell D, et al. Bag-mask ventilation during tracheal intubation of critically ill adults. N Engl J Med. 2019;380(9):811-821.
- Khorsand S, Chin J, Rice J, et al. Role of point-of-care ultrasound in emergency airway management outside the operating room. Anesth Analg. 2023;137(1):124-136.
- Heffner AC, Swords DS, Neale MN, et al. Incidence and factors associated with cardiac arrest complicating emergency airway management. Resuscitation. 2013;84(11):1500-1504.
- Russell DW, Casey JD, Gibbs KW, et al. Effect of fluid bolus administration on cardiovascular collapse among critically ill patients undergoing tracheal intubation: a randomized clinical trial. JAMA. 2022;328(3):270-279.
- Matchett G, Gasanova I, Riccio CA, et al. Etomidate versus ketamine for emergency endotracheal intubation: a random- ized clinical trial. Intensive Care Med. 2022;48(1):78-91.
- Allyn J, Vandroux D, Jabot J, et al. Prognosis of patients presenting extreme acidosis (pH <7) on admission to intensive care unit. J Crit Care. 2016;31(1):243-248.
- Capone J, Gluncic V, Lukic A, et al. Physiologically difficult airway in the patient with severe hypotension and metabolic acidosis. Case Rep Anesthesiol. 2020;2020:8821827.
- Madhivathanan PR, Corredor C, Smith A. Perioperative implications of pericardial effusions and cardiac tamponade. BJA Educ. 2020;20(7):226-234.
- Mittnacht AJ, Fanshawe M, Konstadt S. Anesthetic considerations in the patient with valvular heart disease undergoing noncardiac surgery. Semin Cardiothorac Vasc Anesth. 2008;12(1):33-59.



