Critical Care
KEY POINTS
- Pulmonary artery catheters (PACs) may be used to measure key physiological variables, including cardiac output, and provide an estimate of left ventricular preload.
- Insertion of a PAC is not without risk and requires knowledge of right heart pressures.
- Evidence for the use of PACs in terms of mortality benefit has been conflicting, but in the correct clinical context, they can provide essential information to guide changes in management of vasoactive supports and fluid therapies.
INTRODUCTION
The pulmonary artery catheter (PAC) is a versatile monitoring device capable of direct, simultaneous measurement of pulmonary and cardiac pressures and oxygen saturation, and indirect calculation of a wide array of haemodynamic parameters including cardiac output (CO) and systemic and pulmonary vascular resistance.
HISTORY
Right heart catheterisation was first described in humans in 1929 by Dr Werner Forssman, who performed it on himself using a 4 French ureteric catheter via his antecubital fossa. Subsequent investigators described the use of right heart catheterisation to measure intracardiac pressures, including pulmonary capillary wedge pressure (PCWP), which was found to correlate closely with pressure in the left atrium (LA). The 1956 Nobel Prize for Medicine was shared between Werner Forssmann and Columbia University’s Cournand and Richards who estimated CO using mixed venous and arterial blood oxygen content, employing right heart catheters for sampling. Dye and thermodilution techniques using the Fick principle were subsequently employed during the 1960s to approximate CO.1
Catheterisation of the right heart was mostly limited to the cardiac catheterisation lab as it required fluoroscopic assistance until the development of the balloon-tipped, flow-directed PAC by Swan and Ganz in 1970.2 This catheter could be rapidly inserted at the bedside, and advanced into the wedge position for estimation of LA pressure. Furthermore, it could be left in situ for serial measurements allowing physicians to measure and adjust preload and vascular resistance and immediately evaluate the response to therapy.
Unfortunately, the benefits of the PAC as a monitoring device have not translated into improved mortality in the majority of patients. Although the popularity of PACs has declined significantly in recent years, there still exist several important indications for their use. A knowledge of their insertion and potential complications, as well as interpretation of PAC measurements, is still essential for the anaesthetist and intensivist.
STRUCTURE OF THE PAC CATHETER
Standard PACs for use in adults (Figure 1) are 5 to 8 French and 100 to 110 cm long. They are usually inserted via a short, wide-bore PAC introducer sheath. The catheters are marked at 10-cm intervals to facilitate identification of catheter depth. A plastic sheath with a locking mechanism allows for the catheter to remain sterile as it is moved in and out during wedging.
PACs have between 3 and 5 lumens depending on the model.
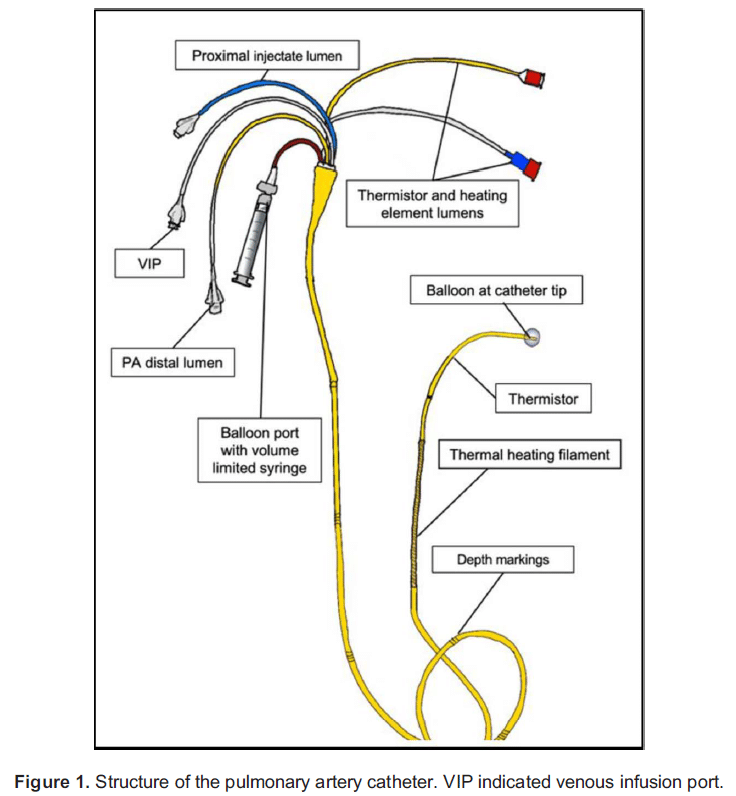
Lumens
Pulmonary Artery (PA) Pressure Transducer/Sampling Lumen (Yellow)
The tip of the catheter has a single open lumen which is used to transduce pulmonary artery (PA) pressures and to take PA blood samples.
Balloon Inflation Lumen (Red)
This lumen is used to inflate and deflate the balloon found at the tip of the catheter with 1.5 ml of air to allow wedging. Usually, a specialised volume-limited syringe is supplied which prevents inflation above this volume to avoid trauma to the PA.
Thermistor Lumen/Connector
The third lumen contains two insulated wires leading to a thermistor that is approximately 3.7 cm from the catheter tip. This measures the change in temperature of the blood as it flows past during thermodilution measurement of CO. Some PACs may have a thermal heating filament to allow for continuous thermodilution CO monitoring. These models have an additional connector to power the heating filament.
Proximal Lumens(s) (Blue/White)
The proximal lumen should open in the right atrium (RA), being positioned about 26 to 30 cm from the catheter tip. It is used to administer the cold injectate during cardiac output measurement. A second, more proximal, lumen designated as the ‘venous infusion port’ (VIP) may be present for drug infusions depending on the catheter design.
Additional Features
Some catheters also have an optical sensor for continuous mixed venous oxygen saturation readings and pacing wires.
RIGHT HEART PRESSURES AND PCWP
The pressure waveform transduced from the tip of the PAC changes in a predictable manner as the catheter is advanced through the chambers of the right heart and into the PA (Figure 2). The PCWP is obtained by inflating the balloon on the tip of the catheter and advancing until the pulsatile PA wave disappears, being replaced by an atrial waveform.
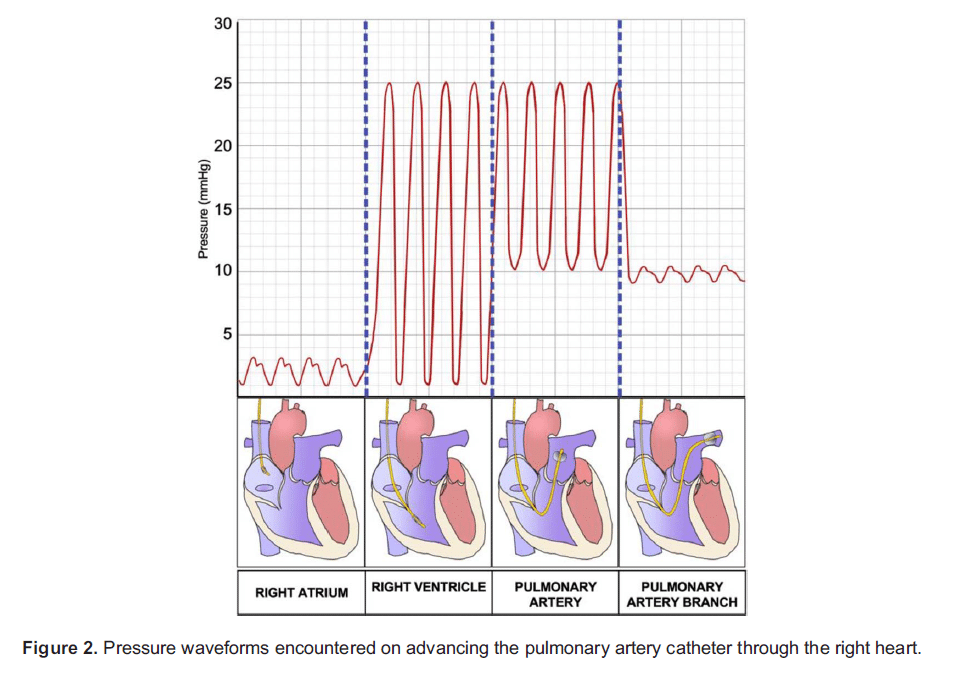
When the balloon is wedged, flow through the pulmonary circulation distal to the balloon is stopped. Thus, there is a static column of blood between the catheter tip, through the pulmonary capillary bed and into the LA, and PCWP should therefore be equivalent to LA pressure. Under normal conditions (and in particular normal transmitral flow), the LA pressure should equal the left ventricu- lar end diastolic pressure (LVEDP), which may act as a surrogate measure of left ventricular end diastolic volume (LVEDV) and thus left ventricular preload.
Limitations of PCWP
PCWP may not reliably represent LVEDP in the following situations:
Non-West Zone 3 Catheter Tip Position
PCWP may only represent LA pressure (and thus LVEDP) when there is an uninterrupted column of blood between the wedged catheter and the LA. This will only occur in the West zone 3 where pulmonary capillary pressure is greater than alveolar pressure. Practically speaking, this is below the level of the LA or behind it in the supine position. Of note, high intrathoracic pressures (eg high positive end-expiratory pressure) and hypovolaemia may produce a similar non–zone 3 effect, with an artificially low PCWP.
Mitral Regurgitation and Poor Left Ventricular Compliance
Large regurgitant pressure waves (visible on the PCWP trace as ‘v’ waves), or high pressures generated during atrial systole to fill a poorly compliant left ventricle may result in high mean atrial pressures above actual LVEDP.
Mechanical Obstruction
Pulmonary venous obstruction, mechanical atrial obstruction or a poorly compliant atrium can result in PCWP in excess of actual LA (and thus LVEDP) pressure.
It is also important to note that LVEDP is not always a reliable estimate of LVEDV. LVEDV may be reduced with deceptively normal LVEDPs in the context of cardiac tamponade or a poorly compliant left ventricle.
ESTIMATING CO
CO is estimated with a PAC using thermodilution, which can be classified as an indicator-dilution method.3 Indicator-dilution methods are based on the premise that when an indicator is added to flowing liquid in a closed circuit, the flow is related the average change in concentration measured at a distal site. Specifically, the flow in the system is inversely proportional to the area under the concentration-time curve, which is described by the Stewart-Hamilton equation (Figure 3).
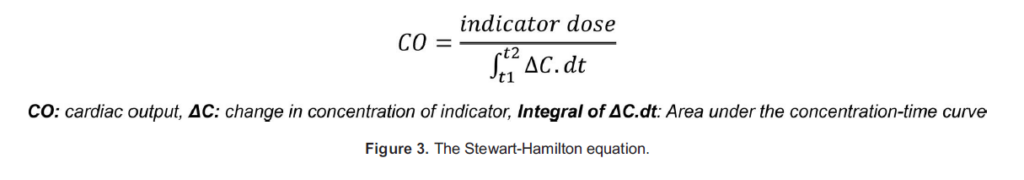
In thermodilution, the indicator used is a bolus of cold fluid of known temperature and volume, while the concentration change is a change in temperature from baseline. A 5- to 10-mL bolus of cold liquid (usually 5% dextrose at 58C; saline may also be used depending on the density constant used to calculate CO) is injected into the RA, causing a drop in temperature of the blood enter- ing the PA. The rate of blood flow is inversely proportional to the mean change in temperature (represented by the area of the temperature time curve, the AUC). The Stewart-Hamilton equation also describes this relationship; however, it is modified to take the temperature, specific heat, and specific gravity of the injectate and blood into account (Figure 4).
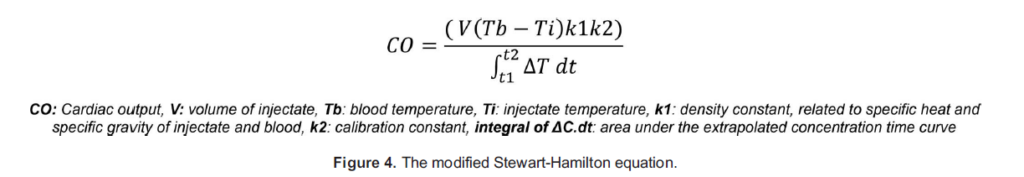
Both indicator-dilution and thermodilution methods produce a similar curve, with an initial sharp deflection followed by a slower return to baseline as the blood dilutes and carries out the cold indicator. The CO is inversely proportional to the area under this curve, thus the shape of these curves is altered with varying cardiac output (Figure 5).
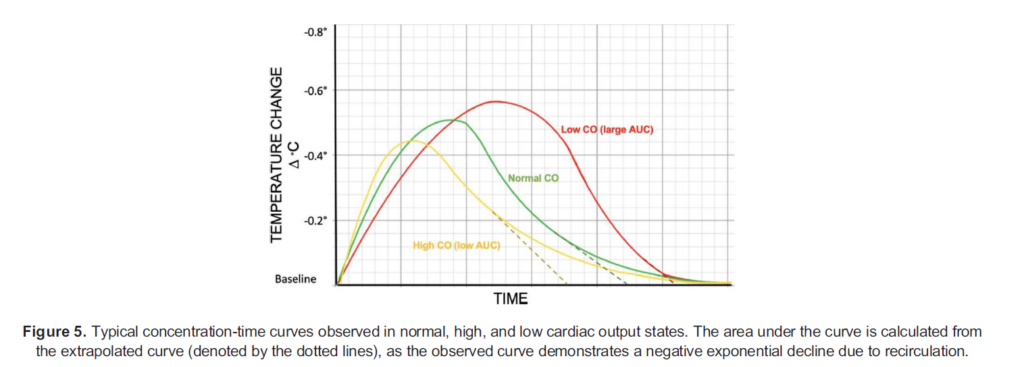
Limitations
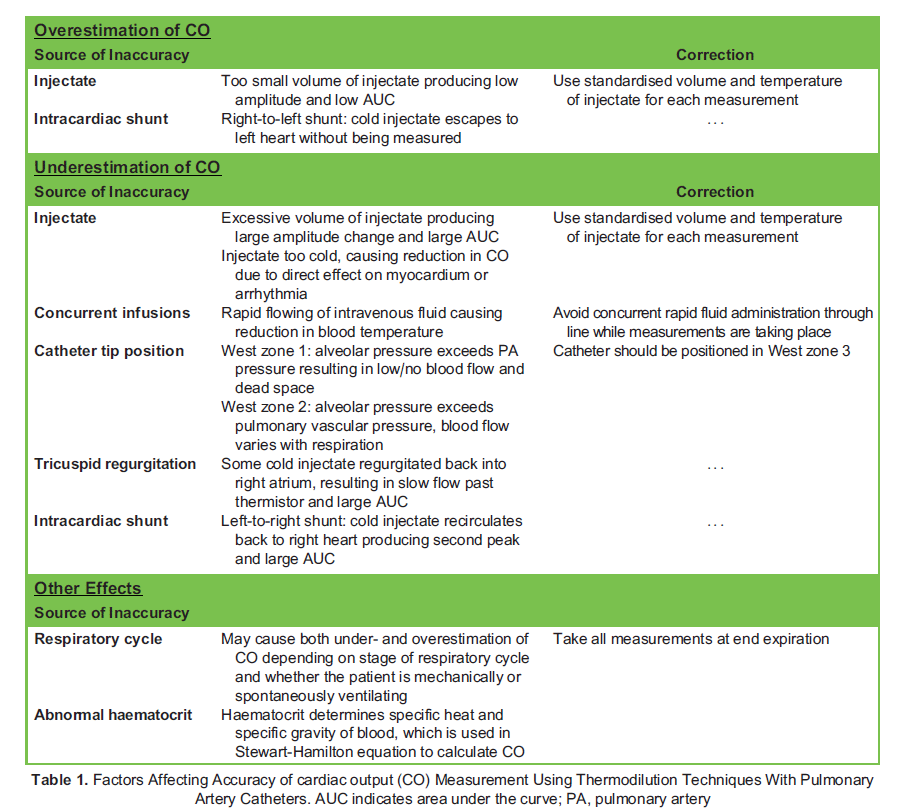
Thermodilution CO measurement accuracy can be adversely affected by several factors listed in Table 1.
INSERTION AND COMPLICATIONS4
- Under strict asepsis and ultrasound guidance, the PA sheath is inserted into the internal jugular vein using the Seldinger technique. Other central veins (femoral, subclavian) may also be used.
- The distal port of the PAC is attached to a pressure transducer zeroed at the level of the patient’s heart.
- The catheter is inserted into the sheath and advanced to approximately 15 cm, after which the tip will exit the sheath and the balloon may be inflated using the volume-limited syringe.
- The catheter may be advanced to enter the RA, followed by the right ventricle (RV), pulmonary artery and finally into the wedge position (see Table 2 for typical depths and pressures). Mean RA pressure, RV/PA systolic, diastolic and mean pres- sures and finally the end expiratory PCWP should be measured.
- The balloon is deflated and a PA trace should be observed. If not, the catheter should be withdrawn until a PA trace
- The catheter depth should be noted and then may be secured in the plastic sheath.

Following the insertion of PAC, a chest radiograph should be performed to identify catheter misplacement or pneumothorax. The tip of the catheter should not be more than 1 cm lateral to the mediastinal margin, or beyond the pulmonary hilum on chest x-ray, to avoid trauma to distal branches of the PA and vascular obstruction causing pulmonary infarction. Other complications associated with PAC insertion include ventricular arrhythmias, PA or myocardial perforation, and pulmonary embolus.
PAC insertion should be avoided in the context of endocarditis, right-sided heart masses (clot or tumour) or prosthesis at risk of damage or dislodgement, high-grade right-sided valvular disease, and severe coagulopathy. Caution should be observed in patients with LBBB in whom catheter passage may induce complete heart block. The risk of myocardial perforation is also increased in patients who have a severely dilated or thinned RA or RV.
NORMAL VALUES AND INTERPRETATION
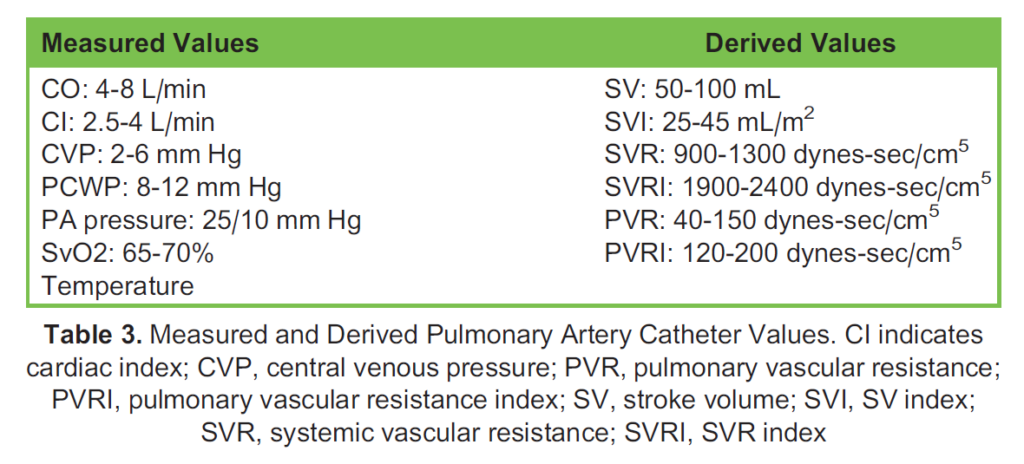
Normal measured and derived values which can be obtained from a PAC are found in Table 3.
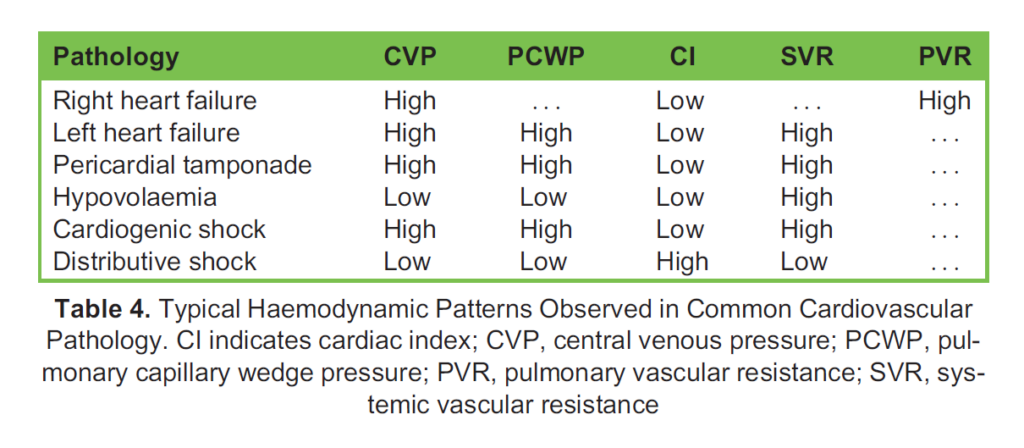
These values are deranged in distinct patterns with cardiovascular pathology, common examples of which are found in Table 4.
INDICATIONS AND EVIDENCE FOR USE
PACs may be inserted at the bedside and can yield valuable measurements of stroke volume, CO, mixed venous oxygen satura- tions, and intracardiac pressures, as well as providing data for several derived variables to guide diagnosis and management. They have been utilised extensively in the intensive care setting with the wealth of haemodynamic information provided guiding vasoactive, inotropic, and diuretic therapy. Unfortunately, evidence for a mortality benefit was not borne out in several large trials and subsequent metanalyses (Table 5) and as such most societies’ guidelines have moved away from recommending the routine use of PACs in critically ill patients.
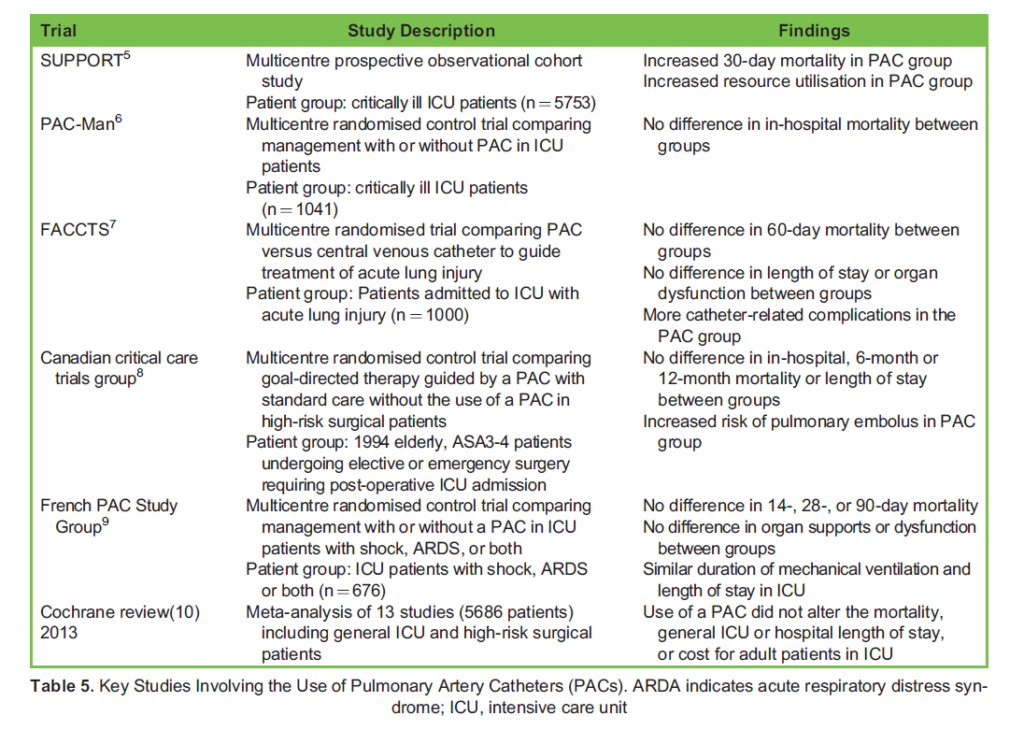
PAC insertion is still indicated in certain situations, and there have been calls in recent years to reconsider the evidence against their use, which may be less conclusive than is apparent from headline results. Many of the studies investigating PAC use in general intensive care unit (ICU) patients are subject to significant limitations. The SUPPORT study was one of the first to signal a worrying lack of mortality benefit with PAC use; however, it was not designed for this purpose.5 This observational study only included patients with an estimated 6-month mortality risk of > 50% from a heterogenous group of severe disease categories, the outcomes of which would be highly unlikely to be modified by using a PAC. The PAC-Man study, one of the largest randomised clinical trials involving ICU patients, showed no in-hospital mortality benefit; however, it is important to note that it was designed to detect a 10% absolute change in this outcome.6 Very few ICU therapies, let alone a monitoring device, are capable of changing mortality rates by 10%. A Cochrane review conducted in 2013 noted that many of the included studies were highly heterogenous in their interventions, with a lack of standardisation of therapies based on PAC information.10
PACs continue to be used regularly in cardiothoracic surgery where the evidence for benefit is mixed. Recent large observational studies suggest that routine PAC insertion in uncomplicated cardiac surgery may not confer a mortality benefit.11,12 The literature suggests that the advantages of PAC monitoring are likely to be seen in more complex patients, such as those with severe left ventricular failure, those undergoing cardiac transplant, multivalvular operations, and ventricular assist device insertion, all of which have been variously excluded from historical PAC studies. Thus PACs do not appear to add benefit for ‘routine’ or low-risk cardiac surgery, but in appropriate patients with more complex pathology may yield invaluable information for the clinician.
CONCLUSION
Despite the overall decline in their use, PACs and the variables which they measure underpin core physiological principles central to the practice of intensive care and anaesthesia. In the appropriate clinical context, the PAC may provide essential information to guide changes in management of vasoactive supports and fluid therapies.
REFERENCES
- Thakkar AB, Desai SP. Swan, Ganz, and their catheter: its evolution over the past half century. Ann Intern Med. 2018; 169(9):636-642.
- Swan HJ, Ganz W, Forrester J, Marcus H, Diamond G, Chonette Catheterization of the heart in man with use of a flow- directed balloon-tipped catheter. N Engl J Med. 1970;283(9):447-451.
- Argueta EE, Paniagua Thermodilution cardiac output: a concept over 250 years in the making. Cardiol Rev. 2019; 27(3):138-144.
- Kelly CR, Rabbani Videos in clinical medicine. Pulmonary-artery catheterization. N Engl J Med. 2013;369(25):e35.
- Connors AF, Speroff T, Dawson NV, et al. The effectiveness of right heart catheterization in the initial care of critically ill patients. SUPPORT Investigators. JAMA. 1996;276(11):889-897.
- Harvey S, Harrison DA, Singer M, et Assessment of the clinical effectiveness of pulmonary artery catheters in manage- ment of patients in intensive care (PAC-Man): a randomised controlled trial. Lancet. 2005;366(9484):472-477.
- Wheeler AP, Bernard GR, Thompson BT, et al. Pulmonary-artery versus central venous catheter to guide treatment of acute lung injury. N Engl J Med. 2006;354(21):2213-2224.
- Sandham JD, Hull RD, Brant RF, et A randomized, controlled trial of the use of pulmonary-artery catheters in high-risk surgical patients. N Engl J Med. 2003;348(1):5-14.
- Richard C, Warszawski J, Anguel N, et Early use of the pulmonary artery catheter and outcomes in patients with shock and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2003;290(20):2713-2720.
- Rajaram SS, Desai NK, Kalra A, et Pulmonary artery catheters for adult patients in intensive care. Cochrane Database Syst Rev. 2013;2013(2):CD003408.
- Shaw AD, Mythen MG, Shook D, et Pulmonary artery catheter use in adult patients undergoing cardiac surgery: a retro- spective, cohort study. Perioper Med (Lond). 2018;7:24.
- Brown JA, Aranda-Michel E, Kilic A, et The impact of pulmonary artery catheter use in cardiac surgery. J Thorac Cardiovasc Surg. 2022;164(6):1965-1973.e6.



