Basic Sciences
KEY POINTS
- Vasopressin is an endogenous nonapeptide neuroendocrine hormone involved in the homeostatic control of serum osmolality and blood pressure, exerting its effects via vasoconstriction and increased renal resorption of fluid.
- Vasopressin infusion is recommended by the Surviving Sepsis Guidelines (2016) as a second-line vasopressor agent to increase blood pressure or reduce noradrenaline requirements in patients with septic shock.
- Evidence of its efficacy in septic shock is unclear, but it may confer benefit in certain subgroups of patients.
- Vasopressin has 2 analogues, terlipressin and desmopressin, which are used in the treatment of bleeding oesophageal varices, cranial diabetes insipidus and in patients with haemophilia and von Willebrand disease.
INTRODUCTION
Vasopressin, also known as antidiuretic hormone or arginine vasopressin, is an endogenous hormone involved in the homeostatic control of serum osmolality and blood pressure. Vasopressin is released by the posterior pituitary gland and has actions on a number of receptor types. In health, the predominant physiological effect is to increase the absorption of water in the renal collecting duct, thereby reducing serum osmolality and increasing circulating volume.1 Figure 1 shows a diagram describing the mechanism of this effect. There are, however, a number of different receptor subtypes with differing physiological effects, including potent vasoconstriction. These actions pose a potentially important physiological system to be targeted clinically.
Septic shock is one such area of interest. Vasopressin provides a mechanism of vasoconstriction not mediated by catecholamines and as such offers an additional treatment option in this cohort. Evidence to support its use is inconclusive. The VAAST trial (2008)2 was the first randomised controlled trial to investigate the effects of vasopressin, and the results showed a significant noradrenaline sparing effect, but the trial lacked evidence of reduced mortality. Subsequent studies, including the VANISH trial (2016)3 and a large individual patient data meta-analysis by Nagendran et al (2019),4 have also failed to show improved mortality rates in this patient cohort but may demonstrate benefits in other clinical parameters.
This tutorial will explore the basic science of vasopressin and its effects on health and disease. Its pharmacokinetics and clinical uses will be explored, including the current evidence underpinning its use.
PHYSIOLOGY IN HEALTH
The body maintains tight control of serum osmolality, between 275 to 295 mmol/kg. Osmolality reflects the total number of particles dissolved in a kilogram of solute, measured in mmol/kg and importantly controls total body water due to permeability of the cell membrane to water. Important mechanisms to maintain this include thirst and subsequent water intake, excretion of osmotically active particles and vasopressin.
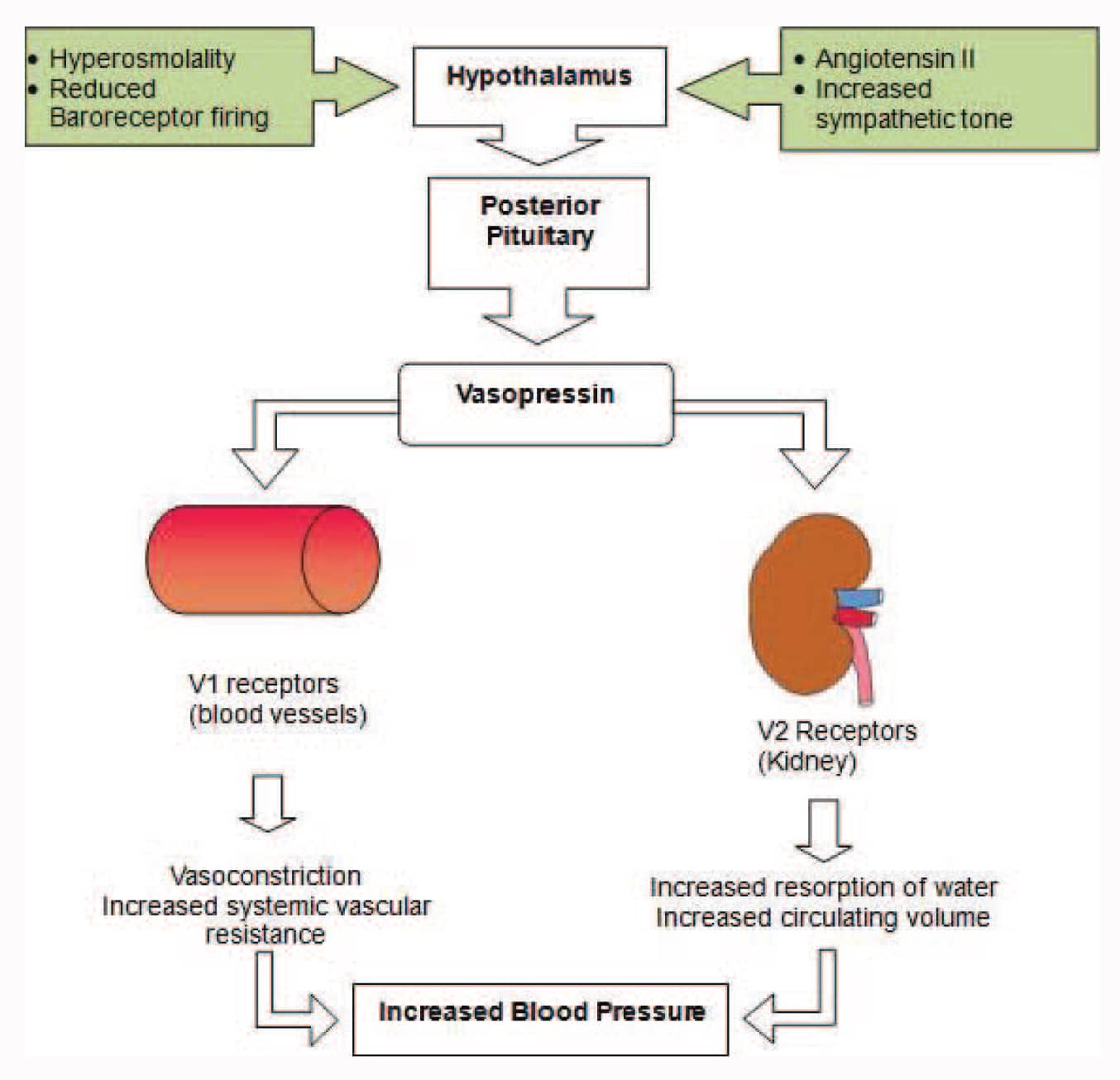
Figure 1. Vasopressin mechanism of action.
Vasopressin is a small nonapeptide neuroendocrine hormone produced in the supraoptic nucleus and paraventricular nucleus of the hypothalamus by magnocellular neurons. It is transported down the infundibulum via the nerve axons to the cell body to be stored, located in the posterior pituitary gland.
In response to raised serum osmolality, vasopressin is released from the posterior pituitary into the systemic circulation, whereby it exerts its effects. Its release is also stimulated by sympathetic stimulation, reduced baroreceptor firing and angiotensin II.1,5 The receptors by which it exerts its effect and their mechanism of action are shown in Table 1.
PHARMACOKINETICS
Vasopressin is available in 3 synthetic analogue forms. Table 2 shows the uses of these analogues.
Absorption
Desmopressin is the only vasopressin analogue that does not require intravenous administration. Absorption is 0.25% sublingual, 0.08% to 0.16% oral, or 10% intranasally.
Distribution
Vasopressin and its analogues have small volumes of distribution: vasopressin, 0.14 L/kg; terlipressin, 0.5 L/kg; desmopressin, 0.2 to 0.32 L/kg. Vasopressin is not protein bound.
Metabolism
Endogenous vasopressin is metabolized by hepatic vasopressinases, conferring a half-life of 10 to 35 minutes. Terlipressin half-life is longer at 50 to 70 minutes, and that of desmopressin is longer still at 2 to 3 hours, because of minimal enzymatic degradation.
Excretion
A total of 65% of the given dose of both vasopressin and desmopressin is excreted unchanged in the urine.6
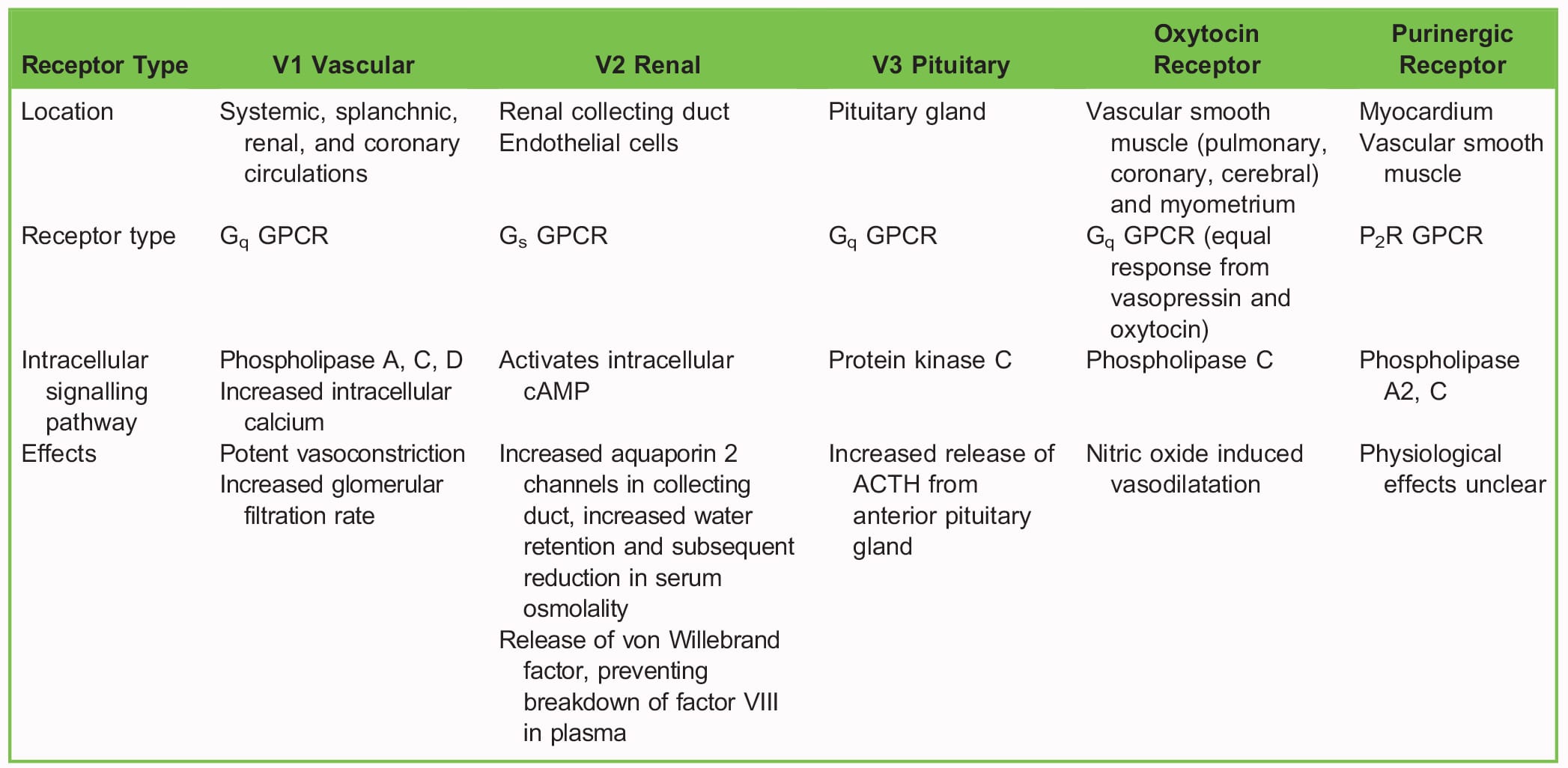
Table 1. Vasopressin Receptor Physiology1 (ACTH, adrenocorticotropic hormone; GPCR, G protein–coupled receptor)
SIDE EFFECTS
Table 3 displays common side effects observed with the use of vasopressin and its analogues.
CLINICAL USES
Vasopressin in Catecholamine-Refractory Septic Shock
The 2016 Surviving Sepsis Guidelines8 provide guidance for evidence-based best practice management in treating sepsis, including early intravenous fluid therapy, antimicrobial agents, and vasopressor support. Noradrenaline is the current first-line vasopressor in these guidelines; however, vasopressin is recommended second line in catecholamine-resistant states. The guidance states:
We suggest adding either vasopressin (up to 0.03 U/min) (weak recommendation, moderate quality of evidence) or epinephrine (weak recommendation, low quality of evidence) to norepinephrine with the intent of raising MAP to target, or adding vasopressin (up to 0.03 U/min) (weak recommendation, moderate quality of evidence) to decrease norepinephrine dosage.
In clinical practice, a dose range of 0.01 to 0.04 U/min is frequently observed.6 Higher infusion rates have been associated with increased rates of limb, digital and mesenteric ischaemia. There is no clear recommendation or threshold of when to start vasopressin in catecholamine-refractory septic shock, and initiation of its therapy is dependent on the clinician’s interpretation of a catecholamine refractory state.
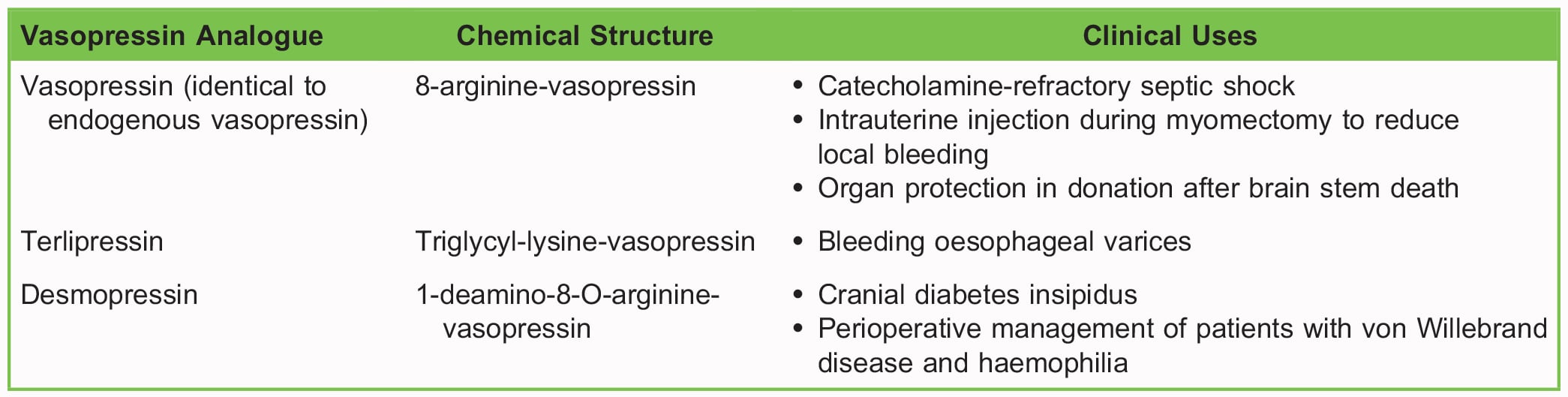
Table 2. Analogues of Vasopressin and Their Clinical Uses6

Table 3. Side Effects of Vasopressin and its Analogues6,7
Vasopressin levels in septic shock have been shown to be lower than anticipated, and this has been termed relative vasopressin deficiency.9 The reason for this and its significance is unclear. The VASST trial, a randomised controlled trial comparing noradrenaline with noradrenaline and vasopressin at 0.03 U/min in patients with septic shock, showed no difference in outcomes.2 In a subgroup analysis, however, patients on noradrenaline infusions at a rate less than 15 μg/min showed increased survival with the addition of vasopressin.
The VANISH trial3 randomised 409 patients treated for septic shock to receive either vasopressin (with hydrocortisone or placebo) or noradrenaline (with hydrocortisone or placebo). Again, this showed no improvement in mortality but did show a reduction in the use of renal replacement therapy in the vasopressin arm. This may be explained by its property of preferentially constricting the efferent renal arteriole over the afferent renal arteriole, increasing glomerular pressure.
Subsequent meta-analysis, including 9 trials with 1324 patients, has substantiated the lack of certainty in data around mortality benefit.4 Although most studies show efficacy in demonstrating a catecholamine sparing effect, beneficial effects on morbidity and mortality have not been proven. For these reasons, the guidelines are weak recommendations, with considerable variation seen in clinical practice.
Vasopressin in Cardiac Arrest
There has been interest in vasopressin use in cases of cardiac arrest, leading to several randomised control trials. A Cochrane review of 26 such trials, involving 21thinsp;704 participants, examined the use of adrenaline, vasopressin or a combination in patients suffering from cardiac arrest in or out of hospital. In comparison with high-dose adrenaline as a single agent, singleagent vasopressin or in combination with adrenaline conferred no benefit in terms of return of spontaneous circulation, survival to admission or survival with a favourable neurological outcome. As such, the use of vasopressin is not supported by current evidence.10 Vasopressin was removed from the Advanced Cardiac Life Support (ACLS) guidelines for cardiac arrest in 2015 as a result of this evidence.
Vasopressin in Surgical Interventions
The von Willebrand factor is a large glycoprotein involved in haemostasis. It is required for platelet adhesion and acts as a bridge between a damaged subendothelium and activated platelets. It also acts to inhibit the breakdown of Factor VIII, enhancing haemostasis.11 Vasopressin and desmopressin act via endothelial V2 receptors, resulting in cyclic AMP–mediated enhancement of von Willebrand factor release, up to 5-fold.12 For this reason, desmopressin is indicated in the perioperative management of von Willebrand disease and haemophilia A. It is important to know that the platelet count may paradoxically fall with desmopressin treatment in certain types of von Willebrand disease (type IIB), and thus controlled, monitored administration prior to surgery to assess response may be indicated. The dose required in this setting is up to 15 times higher than that required for diabetes insipidus (DI).
Vasopressin is also used in cases of myomectomy and other surgical interventions on the uterus. The vasoconstrictive properties observed following direct intrauterine injection reduce the rates of local bleeding but frequently cause a corresponding rise in blood pressure that the anaesthetist must be aware of.
Vasopressin Use in Organ Donation
Patients who suffer from brain stem death undergo extensive pathophysiological changes, with profound hormonal dysregulation. Raised levels of catecholamines and reduced levels of cortisol, vasopressin, insulin and thyroid hormones are observed. In those who are to donate their organs following brain stem death, there is therefore a desire to prevent the deleterious effects these changes may provoke. Organ donation specialists may recommend vasopressin administration in these patients with a hope to improve organ function after transplant and thus patient outcomes. Vasopressin use is frequently advised, including when DI is not observed. Despite the underlying logic underpinning its use, the supporting evidence is weak and largely relies on expert opinion. For this reason, its recommendation varies according to local policies, and input from specialists in organ donation should be sought.13
Terlipressin in Bleeding Oesophageal Varices
Oesophageal variceal bleeding is a serious, life-threatening complication of portal venous hypertension. Submucosal veins in the distal third of the oesophagus become grossly dilated in response to increased portal pressure and can bleed profusely into the oesophagus. Terlipressin exerts its benefit in this patient group via V1 receptor agonism.14 Splanchnic vasoconstriction reduces portal venous pressure, thus reducing the venous pressure of the oesophageal varices. By reducing the rate of bleeding observed through this effect, additional time is created to facilitate adequate resuscitation and definitive management via endoscopic intervention.
A large 2018 meta-analysis15 of 3344 patients within 34 randomised control trials demonstrated that terlipressin significantly improved the control of bleeding within 48 hours, reduced in-hospital mortality, lowered the risk of complications compared with vasopressin, and further improved outcomes when combined with endoscopic ligation therapy.
In the United Kingdom, a dose of 2 mg every 4 hours intravenously until bleeding is controlled is recommended, decreasing to 1 mg every 4 hours if required for a maximum of 48 hours.7 Manufacturers recommend dose reduction in those with lower body weights; therefore, local guidelines should be sought.
Desmopressin in Cranial DI
Cranial DI is a condition characterised by an inability to secrete endogenous vasopressin, resulting in polyuria, polydipsia and nocturia. Hypovolaemia, dehydration and electrolyte derangement follow if left untreated because of the body’s inability to maintain appropriate fluid balance. Causes include surgery, brain injury, infection, tumours and hereditary conditions.16
In 1913, vasopressin was first used in the treatment of DI through administration of extracts of the posterior pituitary containing both vasopressin and oxytocin. It was not until the 1970s that desmopressin was introduced, which conferred many advantages over vasopressin. Desmopressin has a less potent vasoconstrictive profile compared with vasopressin and has a more pronounced antidiuretic action. These factors, in addition to its longer duration of action, availability of oral preparations, and reduced side effect profile, make desmopressin a more desirable treatment option.16 In the adult population, oral dosage can range from 0.2 to 1.2 mg daily, in 3 divided doses. Intravenous, sublingual, and intranasal doses vary because of their altered absorption profiles.7
SUMMARY
Vasopressin is an endogenous hormone with important roles in fluid and blood pressure homeostasis. It has a number of clinical uses because of its broad range of physiological roles including those on the cardiovascular system, fluid balance and haemostasis. Its use in catecholamine-resistant septic shock is recognised, but evidence of benefit is limited. Nevertheless, it offers a novel pharmacologic option in this cohort as it does not rely on the sympathetic nervous system to exert its effects.
REFERENCES
- Holmes CL, Landry DW, Granton JT. Science review: vasopressin and the cardiovascular system part 1—receptor physiology. Crit Care. 2003;7(6):427-434.
- Russell JA, Walley KR, Singer J, et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med. 2008;358(9):877-887.
- Gordon AC, Mason AJ, Thirunavukkarasu N, et al. Effect of early vasopressin vs norepinephrine on kidney failure in patients with septic shock: the VANISH randomized clinical trial. JAMA. 2016;316(5):509-518.
- Nagendran M, Russell JA, Walley KR, et al. Vasopressin in septic shock: an individual patient data meta-analysis of randomised controlled trials. Intensive Care Med. 2019;45:844-855.
- Danziger J, Zeidel ML. Osmotic homeostasis. Clin J Am Soc Nephrol. 2015;10(5):852-862.
- Smith S, Edwards C, Sasada M. Drugs Used in Anaesthesia and Critical Care. 4th ed. Oxford, UK: Oxford University Press; 2011.
- Kaufmann JE, Oksche A, Wollheim CB, et al. Vasopressin-induced von Willebrand factor secretion from endothelial cells involves V2 receptors and cAMP. J Clin Invest. 2000;106(1):107-116.
- Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45:486-552.
- Landry DW, Levin HR, Gallant EM, et al. Vasopressin deficiency contributes to the vasodilation of septic shock. Circulation. 1997;95(5):1122-1125.
- Kalra S, Zargar AH, Jain SM, et al. Diabetes insipidus: the other diabetes. Indian J Endocrinol Metab. 2016;20(1):9-21.
- Zhou X, Tripathi D, Song T, et al. Terlipressin for the treatment of acute variceal bleeding: a systematic review and metaanalysis of randomized controlled trials. Medicine (Baltimore). 2018;97(48):e13437.
- Joint Formulary Committee. British National Formulary. 78th ed. London, UK: BMJ Group and Pharmaceutical Press;2019.
- Hilzenrat N, Sherker AH. Esophageal varices: pathophysiology, approach, and clinical dilemmas. Int J Hepatol. 2012;2012:795063.
- Finn J, Jacobs I, Williams TA, et al. Adrenaline and vasopressin for cardiac arrest. Cochrane Database Syst Rev.2019(1):CD003179.
- Peyvandi F, Garagiola I, Baronciani L. Role of von Willebrand factor in the haemostasis. Blood Transfus. 2011;9(2):3-8.
- Hahnenkamp K, Bo¨ hler K, Wolters H, et al. Organ-protective intensive care in organ donors. Dtsch Arztebl Int. 2016;113(33-34):552-558.



