Covid-19
KEY POINTS
- Ventilator-associated pneumonia (VAP) is a significant cause of morbidity and mortality in critically unwell patients.
- The key risk factor to the development of VAP is a cuffed endotracheal tube or tracheostomy, both of which interfere with the normal anatomy and physiology of the respiratory tract.
- VAP only occurs in patients who have been intubated and undergone mechanical ventilation (MV) for .48 hours. The risk of VAP increases with prolonged MV.
- Early-onset VAP is usually caused by antibiotic sensitive community-acquired bacteria. VAP that develops more than 5 days after initiation of MV has an increased likelihood of being caused by multidrug–resistant bacteria.
- VAP prevention bundles provide an effective method in which to reduce individual intensive care unit rates of VAP.
- Treatment relies on high clinical suspicion, early diagnosis, and initiation of antibiotics tailored to the results of microbiological specimens.
INTRODUCTION
Ventilator-associated pneumonia (VAP) is a significant cause of morbidity and mortality in critically unwell patients within the intensive care unit (ICU) who undergo invasive mechanical ventilation (MV) via an endotracheal tube (ETT) or tracheostomy. Early diagnosis and adoption of practices known to prevent VAP can reduce mortality and decrease the development of multidrug–resistant organisms.1 Throughout this article we will discuss the incidence, scoring systems, pathophysiology, prevention, risk factors, diagnosis, and treatment of VAP.
Definition
VAP is a type of hospital-acquired pneumonia that occurs more than 48 hours after endotracheal intubation. This can be further classified into early onset (within the first 96 hours of MV) and late onset (more than 96 hours after the initiation of MV), which is more commonly attributable to multidrug–resistant pathogens.2
Incidence
VAP is common in critical care patients and is responsible for around half of all antibiotics given to patients in ICUs.3 Figures quoted by the International Nosocomial Infection Control Consortium suggest that the overall rate of VAP is 13.6 per 1000 ventilator days.4 However, the individual rate varies according to patient group, risk factors, and hospital setting. The average time taken to develop VAP from the initiation of MV is around 5 to 7 days, with a mortality rate quoted as between 24% and 76%.4

Table 1. Risk Factors for the Development of Ventilator-associated Pneumonia 6
PATHOPHYSIOLOGY
The key to the development of VAP is the presence of an ETT or tracheostomy, both of which interfere with the normal anatomy and physiology of the respiratory tract, specifically the functional mechanisms involved in clearing secretions (cough and mucociliary action).5 Intubated patients have a reduced level of consciousness that impairs voluntary clearance of secretions, which may then pool in the oropharynx.4 This leads to the macroaspiration and microaspiration of contaminated oropharyngeal secretions that are rich in harmful pathogens.
Normal oral flora start to proliferate and are able to pass along the tracheal tube, forming an antibiotic-resistant biofilm which eventually reaches the lower airways.5 Critically unwell patients exhibit an impaired ability to mount an immune response to these pathogens, leading to the development of a pneumonia.2 The presence of additional predisposing factors such as pulmonary oedema in these patients can also accelerate the process4 (See Table 1).
Early-onset VAP, occurring within the first four days of MV, is usually caused by antibiotic-sensitive community-acquired bacteria such as Haemophilus and Streptococcus. VAP developing more than 5 days after initiation of MV is usually caused by multidrug–resistant bacteria such as Pseudomonas aeruginosa.4 Table 2 highlights some of the common risk factors for the development of a multidrug–resistant pathogen.6
PREVENTION
VAP prolongs the duration of stay in the ICU, thereby increasing the cost of patient management.4 It therefore makes the prevention of VAP a priority in the management of critically unwell patients. Basic preventative measures include minimising excessive time on a ventilator via the implementation of an early weaning protocol with regular sedation breaks and the avoidance of routine or scheduled ventilator circuit changes.7
Appropriate semirecumbent positioning of patients, with a 30- to 45-degree head-up approach, reduces the incidence of microaspiration of gastric contents when compared with patients nursed in a supine position.7 Stress ulcer prophylaxis raises gastric pH, which is detrimental to the innate immunological protection provided by gastric acid. Stopping stress ulcer prophylaxis in low-risk patients (those patients absorbing feed without a history of gastrointestinal bleeding) is recommended.
Subglottic suction ports have been shown to reduce the incidence of VAP and to significantly reduce the use of antibiotics5 (Figure 1). If it is anticipated that a patient will be mechanically ventilated for more than 72 hours, consideration for insertion of a tube with subglottic drainage should be made.
Microaspiration can also be reduced by the maintenance of endotracheal tube airway cuff pressure at 20 to 30 cm H2O and the use of positive end-expiratory pressure.5
Decontamination of the digestive tract has been studied as a method of reducing the incidence of VAP by decreasing colonisation of the upper respiratory tract. Methods used include antiseptics, such as chlorhexidine in the oropharynx, and

Table 2. Risk Factors for Multidrug–resistant Ventilator-associated Pneumonia (VAP)6
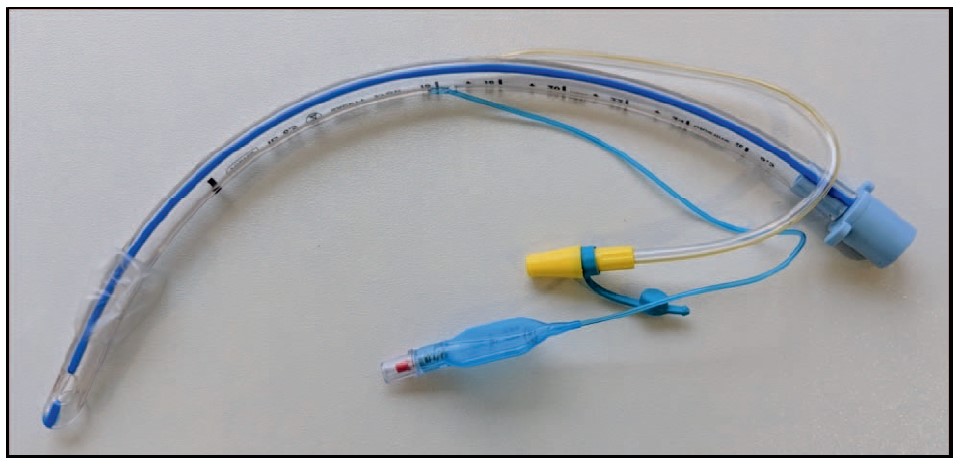
Figure 1. Endotracheal tube with the yellow subglottic suction line (manufactured by Smiths Medical).
nonabsorbable antibiotics, which can be applied to the oropharynx (selective oropharyngeal decontamination [SOD]) or administered enterally (selective digestive-tract decontamination [SDD]). The aim of this method is to eradicate the oropharyngeal or gastrointestinal carriage of potentially harmful pathogens, such as aerobic gram-negative microorganisms and methicillin-sensitive Staphylococcus aureus. These methods were shown to reduce the mortality of ICU patients in hospitals with low antibiotic resistance rates.8 However, SDD has never become mainstream practice, largely due to concerns of long-term microbial ecology within the ICU and selection of drug-resistant organisms. SOD using chlorhexidine mouthwash did become routine practice, although it has more recently been restricted to VAP prevention care bundles for patients who have undergone cardiac surgery. Recent reviews have shown a nonsignificant trend towards worse outcomes in noncardiac patients, although the reason for this is unclear.9 Therefore in noncardiac patients, chlorhexidine mouth care has been removed from many mainstream care bundles.
Regular oral care with basic hygiene aims to reduce dental plaque colonisation with aerobic pathogens. Although there is limited evidence for its use, it is unlikely to cause harm. Probiotic use has not conferred any significant impact on mortality rates.7
Although not yet established in clinical practice, silver-coated ETTs are showing promising results for reducing the relative risk for the development of VAP due to the broad-spectrum antibiotic properties of silver.7
VAP prevention bundles provide an effective method by which to reduce individual unit rates of VAP10 (Table 3).
DIAGNOSIS
VAP classically presents with symptoms such as fever, purulent respiratory secretions, rising inflammatory markers, respiratory distress, and worsening respiratory parameters (reduced tidal volume, increased minute ventilation, and hypoxia).4 Certain groups of patients are vulnerable to atypical organisms and each patient demands a full diagnostic evaluation to identify the
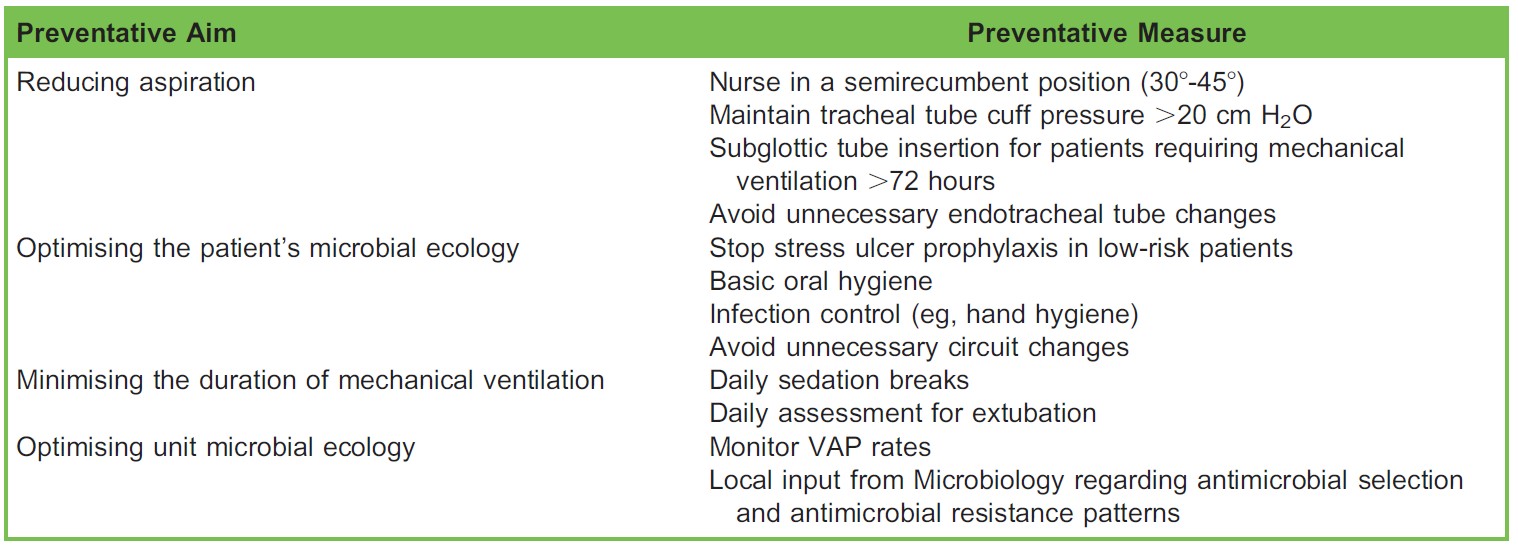
Table 3. Example of Ventilator-associated Pneumonia (VAP) Prevention Bundle
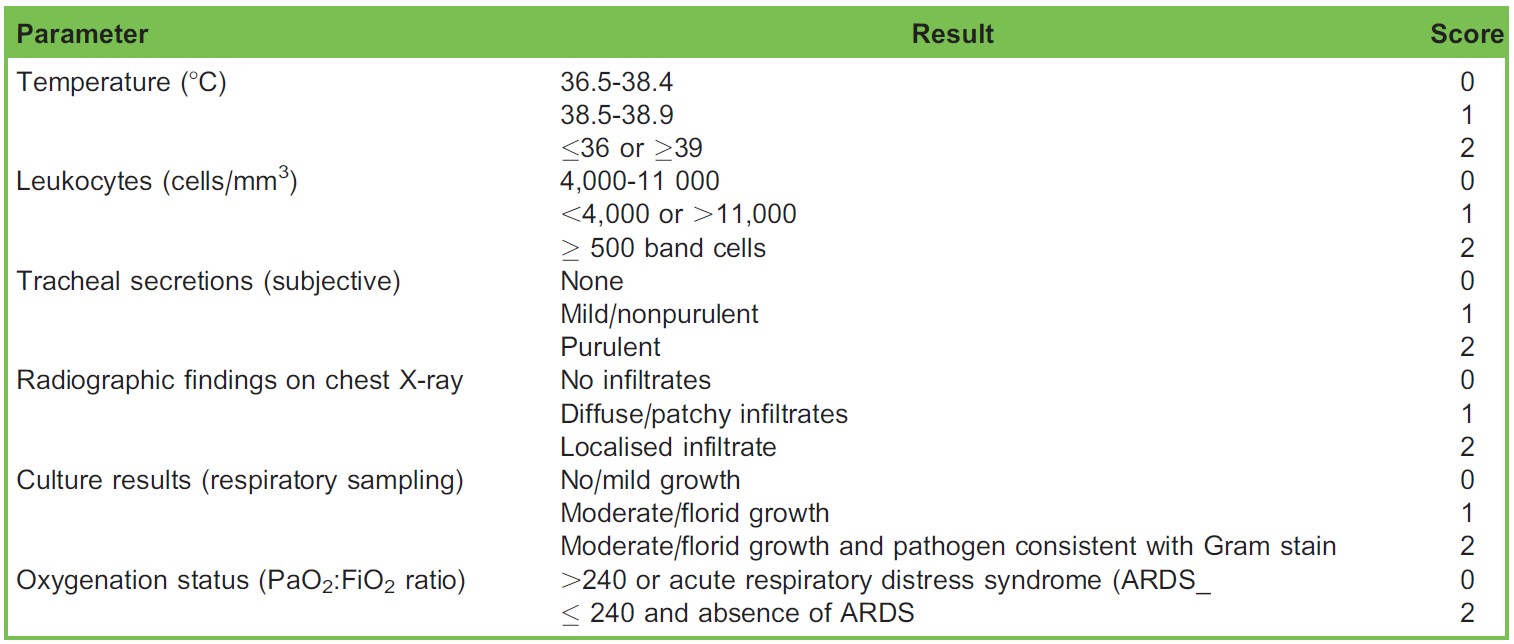
Table 4. The Clinical Pulmonary Infection Score10
likely pathogen before commencing antibiotics.6 Every patient exhibiting signs of VAP should have a chest X-ray and those patients who exhibit changes consistent with infection should have a sample of their respiratory tract secretions sent for Gram stain, culture, and sensitivity.10 A normal chest X-ray should prompt the clinician to consider an alternative diagnosis. However, one study showed that only 43% of patients who had clinical and radiographic evidence of VAP were confirmed to have a diagnosis of VAP on postmortem examination.11
Prior to commencement of broad-spectrum antibiotics, a patient must have respiratory samples sent to microbiology. This can be performed either by nonbronchoscopic sampling (tracheobronchial aspiration or mini-bronchoalveolar lavage) or via bronchoscopic sampling (bronchoalveolar lavage or protected specimen brush). These techniques have been compared and the results indicate that bronchoscopic sampling reduces respiratory tract contamination of samples and provides a more accurate representation of likely pathogens; however, impact on overall morbidity or mortality has not been demonstrated.12 This may allow a more targeted antibiotic regime and earlier de-escalation and cessation of antibiotic therapy.
Inflammatory markers such as procalcitonin and C-reactive protein lack sensitivity and specificity for the diagnosis of pneumonia but they can help decision making and reduce the overuse of antibiotics.13 Current research aiming to improve diagnosis in VAP include novel biomarkers and fibre-optic microbial staining.14,15
Scoring Systems
One scoring system described is the clinical pulmonary infection score (CPIS), which takes a number of different investigations into account.10

Table 5. Hospitals in Europe Link for Infection Control through Surveillance definition of ventilator-associated pneumonia16
A CPIS score of 6 or higher out of a maximum score of 12 indicates a likely diagnosis of VAP.10 There have been concerns raised, however, regarding the diagnostic validity of CPIS, with one meta-analysis reporting the sensitivity and specificity for CPIS as 65% and 64% respectively.10 Furthermore, there is significant user variability in CPIS calculation despite its seemingly simple calculation.10
The Hospitals in Europe Link for Infection Control through Surveillance (HELICS) criteria shown below are commonly used for monitoring VAP rates.16 A diagnosis of VAP is made using the HELICS criteria when each of the radiological, systemic, and pulmonary criteria have been met.
TREATMENT
Treatment of VAP relies on knowledge of common pathogens, patient risk factors (for example immunosuppression and underlying respiratory condition), and previous microbiology specimens. Empirical treatment for VAP should include antibiotics with cover against Pseudomonas aeruginosa, Staphylococcus aureus, and gram-negative bacilli, with antibiotics administered in a timely fashion.2 Delaying treatment and failing to select a suitable antibiotic regimen in accordance with local policy has been shown to result in higher mortality rates.1 Antibiotics should be safely de-escalated once microbiology results are available, with cessation after 7 days based on improving clinical and biochemical markers.6
SUMMARY
VAP remains a significant risk to the critically ill ventilated patient. The risk of developing VAP can be mitigated by VAP prevention care bundles. There is no single diagnostic test for VAP and therefore scoring systems based on multiple parameters are used. Timely diagnosis is required to instigate appropriate antibiotics for improved outcomes. Both patients and units are at risk of developing multidrug–resistant organisms and therefore appropriate antibiotic stewardship is also required. Current research aims to improve diagnostics for VAP, which may lead to improved certainty with when to start antibiotics. However, prevention remains the best cure.
REFERENCES
- Muscedere JG, Shorr AF, Jiang X, et al. The adequacy and timely empiric antibiotic therapy for ventilator-associated pneumonia and blood stream infection: a meta-analysis. J Crit Care. 2012;27:322e7.
- Hunter JD. Ventilator associated pneumonia. Postgrad Med. 2006;82:172-178.
- Vincent JL, Bihari DJ, Suter PM, et al. The prevalence of nosocomial infection in intensive care units in Europe. Results of the European Prevalence of infection in Intensive Care (EPIC) Study. EPIC International Advisory Committee. JAMA. 1995;274:639-644.
- Charles P, Kali A, Easow JM, et al. Ventilator-associated pneumonia. Australas Med J. 2014;7(8):334-344.
- American Thoracic Society, Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171:388.
- Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61.
- Klompas M, Branson R, Eichenwald ED, et al. Strategies to prevent ventilator-associated pneumonia in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014;35:915.
- Silvestri L, Van Saene HK, Casarin A, et al. Impact of selective decontamination of the digestive tract on carriage and infection due to gram-negative and gram-positive bacteria: a systematic review of randomised controlled trials. Anaesth Intensive Care. 2008;36:324.
- National UK guidelines for provision of intensive care version 1.1 (2016). https://www.ficm.ac.uk/standards-and-guide lines/gpics. Access Jun 15 2018.
- Kalanuria AA, Zai W, Mirski M. Ventilator-associated pneumonia in the ICU. Crit Care. 2014;18:208.
- Wunderink RG, Woldenberg LS, Zeiss J, et al. The radiologic diagnosis of autopsy-proven ventilator-associated pneumonia. Chest. 1992;101:458.
- Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med. 2002;165(7):867-903.
- Seligman R, Meisner M, Lisboa TC, et al. Decreases in procalcitonin and C-reactive protein are strong predictors of survival in ventilator-associated pneumonia. Crit Care. 2006;10(5):R125.
- Mills B, Bradley M, Dhaliwal K. Optical imaging of bacterial infections. Clin Transl Imaging. 2016;4:163-174.
- Hellyer TP, Anderson NH, Parker J, et al. Effectiveness of biomarker-based exclusion of ventilator-acquired pneumonia to reduce antibiotic use (VAPrapid-2): study protocol for a randomised controlled trial. Trials. 2016 Jul 16;17(1):318.
- Browne E, Hellyer TP, Baudouin SV, et al. A national survey of the diagnosis and management of suspected ventilatorassociated pneumonia. BMJ Open Respiratory Research. 2014;1(1):e000066.



