Paediatric Anaesthesia
QUESTIONS
Before continuing, try to answer the following questions. The answers can be found at the end of the article, together with an explanation. Please answer True or False:
- Which of the following statements are correct?
a. Ventricular Septal Defects (VSDs) are the most common form of congenital heart disease
b. VSDs are always associated with other cardiac abnormalities
c. Extra-cardiac anomalies may be associated with VSDs
d. VSDs can be classified according to their location on the interventricular septum
e. Subarterial defects are the most common type of VSDs - Ventricular Septal Defects may lead to:
a. Blood flow across the defect
b. Right atrium dilatation
c. Congestive heart failure
d. Excessive pulmonary blood flow
e. Pulmonary hypertension - Concerning the pathophysiology and natural history of VSDs, which are correct?
a. The haemodynamic consequences of a VSD are determined by its size
b. Pulmonary and systemic vascular resistances are important in VSD pathophysiology
c. Signs and symptoms will usually be apparent since birth
d. All VSDs will need surgical treatment
e. The likelihood of spontaneous closure depends solely on location - In the management of ventricular septal defects, which of the following are true?
a. All patients with a VSD will need medication
b. If a patients is symptomatic, diuretics are usually the first-line treatment
c. Some patients may need to be admitted to intensive care prior to surgery
d. Surgery has generally a very high morbidity and mortality
e. For uncomplicated cases therapy can be de-intensified quickly after surgery
Key Points
- VSDs are the most common form of congenital heart disease
- They can occur in isolation, or be part of a more complex congenital heart disease
- Clinical presentation depends on size, location and pulmonary and systemic vascular resistance
- Treatment may be conservative, medical and / or surgical
- Long-term prognosis is good for the vast majority of patients
INTRODUCTION
A Ventricular Septal Defect (VSD) is an abnormal communication between the two ventricles, and it is the most common form of congenital heart disease (CHD). VSDs frequently occur as isolated defects, but can also be one component of more complex cardiac abnormalities.
WHAT IS A VSD?
Definition
This type of lesion consists of an opening between the left ventricle (LV) and the right ventricle (RV), caused by a defect in the wall that separates these two chambers, the interventricular septum (IVS).
Aetiology
Aetiology of VSDs is not precisely known, and although single genetic abnormalities have been identified as causative in some cases, the majority of VSDs are believed to result from the interaction of genetic predisposition and environmental factors.
Associations
VSDs may be associated with congenital abnormalities, such as Trisomy 13, 18 and 21, as well as VACTERL association (Vertebral, vascular, Anorectal, Cardiac, Tracheo-oEsophageal fistula, Renal and Limb anomalies) and CHARGE syndrome (Coloboma, Heart defects, choanal Atresia, Retardation, Genitourinary, Ear anomalies).
Anatomy
VSDs are usually classified according to their location on the interventricular septum (IVS), as seen from the right ventricle. There are four types of VSDs:
- Inlet (also called AV canal, AV septum, endocardial cushion, juxtatricuspid, posterior)
- 5 to 10% of VSDs. Located on the area of the septum formed by endocardial cushion tissue (also called atrioventricular (AV) septum), immediately inferior to the tricuspid valve; associated with anomalies of the AV valves, considered part of atrioventricular septal defects (AVSDs);
- Muscular (can be subdivided according to location, such as apical, central, marginal)
- 5 to 20% of defects, the second most common form of VSDs; completely surrounded by muscle; can be multiple, in which case they are called “Swiss-cheese” defects;
- Perimembranous (conoventricular, infracristal, membranous, paramembranous)
- the most frequent type; they involve the membranous septum, bordered by the AV valve, and may extend into one of the other regions (confluent); the tricuspid valve is often abnormal, valve leaflets or extra tissue may herniate, prolapse or occlude the defect; aortic valve commissure abnormalities may coexist; the defects may coexist with malalignement of the outlet septum, typically as part of a more complex congenital heart disease;
- Subarterial (conal, conoseptal, doubly committed, infundibular, outlet, supracristal)
- 5 to 7% of defects; located below the pulmonary valve (subpulmonary), overlying the outlet septum; one of the aortic valve leaflets may prolapse through the defect, resulting in the development of a left ventricular outflow tract gradient, functional closure of the defect or aortic insufficiency.
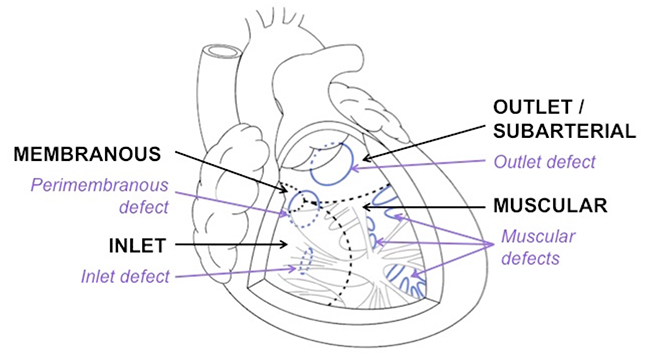
Figure 1: Diagrammatic representation of VSDs classified according to location on the interventricular septum, as seen from a cross-section through the right ventricle.
PATHOPHYSIOLOGY
VSDs occur in 1.5 to 3.5 in 1,000 live births, representing approximately 20% of all cases of CHD. Pathophysiological consequences of a VSD are influenced by its size (see below). VSDs may result in:
- shunting – blood flow across the defect;
- pulmonary hypertension;
- congestive heart failure (CHF).
The major determinants of the haemodynamic state of a patient with VSD are:
- VSD size;
- systemic vascular resistance (SVR);
- pulmonary vascular resistance (PVR);
- associated anomalies (aortic insufficiency, obstruction to LV or RV outflow tracts).
If the VSD is small enough to limit shunting by creating resistance to blood flow, it is called restrictive. When there is no resistance to blood flow across the VSD, it is an unrestrictive defect.
Classification by size
Based on maximum diameter of VSD when compared to normal size of aortic valve annulus
- Small – less than 1/3 of normal aortic valve annulus diameter;
- Moderate – 1/3 to 2/3 of the diameter of the aortic valve annulus;
- Large – more than 2/3 of normal aortic valve annulus size.
Based on amount of pulmonary blood flow relative to systemic blood flow (Qs) – Qp:Qs ratio
- Small – Qp:Qs ratio lower than 1.5;
- Moderate – Qp:Qs ratio of 1.5 to 2.0;
- Large – Qp:Qs ratio higher than 2.0.

Table 1: Pathophysiology of VSDs according to size
NATURAL HISTORY
Natural history of VSDs is related to size and location, both determining the rate of spontaneous closure, and may also be influenced by any associated anomalies. Children with small (restrictive) VSDs usually do not require any medical or surgical therapy, since they are usually asymptomatic and rate of spontaneous closure is high.
Babies with moderate or large defects will frequently need medical therapy and surgery, as they will likely develop congestive heart failure. Symptoms most often become apparent at 2 to 6 weeks of life, after the expected decrease in pulmonary vascular resistance, due to increased pulmonary blood flow and Qp:Qs ratio.
Spontaneous closure of VSDs occurs in up to 80% of muscular defects, especially if they are small; perimembranous defects will often close spontaneously; spontaneous closure of subarterial VSDs may occur, and it is rare in inlet (or malaligned) defects.
Children with large defects, if left untreated, will gradually develop pulmonary hypertension and pulmonary vascular obstructive disease (PVOD). This will eventually result in Eisenmenger syndrome, which is characterized by:
- pulmonary artery (PA) pressures higher than systemic values (supra-systemic PA pressures);
- reversal of the shunt across the VSD from left-to-right to right-to-left;
- cyanosis (and its complications).
DIAGNOSIS
History
A VSD can be diagnosed antenatally by ultrasound. Many VSDs, however, are undetected until the first 2 to 6 weeks of life. The presence of a murmur can then suggest the diagnosis.
Examination
Small VSD
- pansystolic low-intensity or high-pitched murmur on upper or lower left sternal area;
- precordial thrill may be palpable;
- normal growth and development, child remains asymptomatic.
Moderate VSD
- harsh pansystolic murmur on mid sternal border, radiating to the precordial area;
- soft thrill and prominent RV impulse (and possibly also LV impulse) may be present;
- growth and development may be affected;
- signs and symptoms of congestive heart failure.
Large VSD
- physical findings are identical to moderate-sized defects;
- prominent thrill and apical impulse, hyperactive precordium, increased intensity of S2;
- RV dilatation and hypertrophy and rise in PVR are more marked in large VSDs;
- if increased PVR and right-to-left shunt, murmur may be decrescendo, minimal or absent;
- signs and symptoms of marked congestive heart failure – poor growth and development, excessive sweating, tachypnoea, dyspnoea, respiratory distress (particularly with feeding), recurrent pulmonary infections, liver enlargement.
Special Investigations
Oxygen saturation
Usually normal, can be low with: intercurrent respiratory tract infection and pulmonary oedema – in which case it improves with oxygen therapy; or Eisenmenger syndrome (with right-to-left shunt) – not significantly improved by oxygen administration.
Chest X-ray (CXR)
Normal in small defects, cardiomegaly and increased pulmonary vascular markings in moderate defects, marked cardiomegaly and pulmonary oedema seen in large defects.
Electrocardiogram (ECG)
ECG is normal in small VSDs, shows signs of LA dilatation and LV hypertrophy in moderate VSDs, and evidence of biventricular hypertrophy in large VSDs.
Haematology
Typically normal, except for patients with Eisenmenger syndrome, who may develop polycythaemia, thrombocytopenia and coagulopathy as complications of cyanosis.
Echocardiography
It is usually the only imaging modality required to determine the diagnosis, size and location of a VSD, and can also provide information about chamber size, wall thickness, estimates on right ventricular and pulmonary artery pressures, as well as any associated anomalies.
Cardiac catheterisation
Not routinely necessary, can be performed when there are remaining questions about anatomy and pulmonary vascular resistance and its response to therapy.
MEDICAL MANAGEMENT
Medical management of VSDs will depend on clinical symptoms and natural history. Asymptomatic children with small defects do not usually require any medical therapy. Patients with moderate or large defects with signs and symptoms of congestive heart failure are frequently prescribed diuretics. Angiotensin conversion enzyme (ACE) inhibitors may also be used. For a small number of children with severe CHF and pulmonary oedema, pre-operative admission to the Intensive Care Unit (ICU), intravenous diuretics and inotropes may be necessary. Electrolyte imbalances should be carefully monitored and treated, with special attention in children treated with diuretics or ACE inhibitors.
SURGICAL MANAGEMENT
Indications for surgery
- congestive heart failure and / or failure to thrive (refractory to medical therapy);
- moderate or large defects, unlikely to close spontaneously (with or without symptoms);
- development (or progression) of aortic valve leaflet prolapse and / or aortic insufficiency;
- asymptomatic (older) children with Qp:Qs ratio higher than 2.0.
Contraindications for surgery
A small proportion of patients with pulmonary hypertension are not suitable candidates for surgery. If pulmonary vascular resistance studies show a good response to pulmonary vasodilators, then some may be apt for surgery after a period of medical therapy (usually VSD closure with a fenestrated patch, which will allow blood to shunt right-to-left during episodes of increased pulmonary vascular resistance, thus preserving cardiac output at the expense of lower oxygen saturations).
Corrective surgery
The vast majority of children are good candidates for corrective surgical repair. This consists of surgical patch closure of the VSD, requiring open-heart surgery, cardiopulmonary bypass (CPB) and cardioplegic arrest. Most defects can be closed successfully by an incision made on the right atrium (transatrial approach), while some need to be closed through the right ventricle, pulmonary artery or infundibulum.
Pulmonary artery banding
Previously employed as a two-stage strategy to allow for growth until definitive surgery could be performed, it is now usually reserved for critically ill neonates, or those with associated anomalies – who may be too ill for early corrective repair – and some with multiple VSDs.
Transcatheter closure
Even though VSDs have been successfully closed by devices inserted through a major blood vessel, equipment size is a major limitation to their use as a therapy in children.
ANAESTHETIC MANAGEMENT
General anaesthetic management
Anaesthetic management for children with VSDs will be influenced by the individual child’s clinical condition: presence and severity of signs and symptoms of congestive heart failure, excessive pulmonary blood flow (Qp:Qs ratio) and possible elevation of pulmonary vascular resistances.
Most children presenting for VSD closure will be stable, with no or mild CHF and no clinically significant excessive pulmonary blood flow or PVR elevation. Management of anaesthesia for these children includes avoiding the presence of air in any lines and precipitating an increase in left-to-right shunt by lowering PVR (excessive oxygen administration, hyperventilation / hypocapnia, alkalosis, anemia).
Management of CPB for VSD closure in our institution can generally be described as follows:
- inhalation induction with sevoflurane;
- maintenance with isoflurane, including during CPB (delivered to the CPB circuit);
- intraoperative analgesia with fentanyl (total dose during the case 30 to 50mcg/Kg);
- muscle relaxation with pancuronium;
- invasive monitoring, including central venous and arterial access as well as a monitoring line;
- transoesophageal echocardiography;
- anticoagulation with heparin 400 iunits/Kg;
- availability of previously cross-matched blood products (red blood cells, platelets);
- prophylactic antifibrinolytic drugs;
- mild hypothermia during CPB (usually 34ºC);
- haematocrit higher than 21% during CPB and 30% immediately before separation from CPB;
- modified ultrafiltration (MUF); this technique consists of removal of free water and low-molecular-weight substances by convection forces, using a modification of the CPB circuit with a semi-permeable membrane filter, immediately before separation of cardiopulmonary bypass.
Children with severe CHF or pulmonary hypertension
Some children will have severe congestive heart failure and a few will present with high pulmonary vascular resistances. In these cases, it is important to avoid excessive myocardial depression from anaesthetic agents with a carefully balanced technique, and support myocardial contractility with inotropes – milrinone, loading dose 25 to 75mcg/Kg over 30min, followed by continuous infusion at a rate of 0.25 to 0.75mcg/Kg/min. Nitric oxide may also be considered on separation from CPB, to decrease PVR.
Fast-track procedure
Asymptomatic children with a predictable uncomplicated VSD closure should be considered for a “fast-track” procedure, aiming for early tracheal extubation in the operating room or within 4 hours after admission to ICU. In our institution, we use moderate doses of fentanyl (total 10 to 30mcg/Kg during the case), bolus dose atracurium, intravenous paracetamol, local anaesthetic wound infiltration and antiemetic administration in operating theatres to facilitate extubation.
Immediate post-operative care
Separation from cardiopulmonary bypass is usually smooth and uneventful, but it is important in the immediate postoperative period to monitor and correct possible electrolyte imbalances, maintain normothermia and control possible surgical sources of bleeding. Thromboelastography may be a useful tool to guide further protamine or product administration to treat any coagulation abnormalities.
Complications
Although the majority of VSD closure procedures are uneventful, post-operative complications can occur. The most common complications and their respective treatment are briefly described below:
- residual VSD(s) – treatment is surgical or transcatheter closure; size and clinical significance might not be worth the risk of returning to cardiopulmonary bypass or later reintervention;
- heart block – treated by atrioventricular synchronous pacing using epicardial pacing wires;
- arrhythmias – these include junctional ectopic tachycardia (JET), which can occur in children after procedures that involve VSD repair, most often in Tetralogy of Fallot; JET should be treated aggressively, by minimising catecholamine levels (reducing inotropes if possible), cooling to 34 to 35ºC, sedation, muscle relaxation and amiodarone.
Anaesthetic management for non-cardiac surgery in patients with VSDs
Safe conduct of anesthesia for non-cardiac surgery in patients with VSDs depends on multiple factors, such as the individual patient’s clinical condition, the nature of the surgical procedure and the local resources available. For all but the simplest procedures in compensated, stable patients, careful consideration should be given to referral to a specialist centre, if feasible.
General principles for anaesthetic management of patients with a VSD who undergo non-cardiac procedures overlap with those described previously for VSD closure. Inhalation induction with sevoflurane can usually be safely performed, albeit slower than for patients without left-to-right shunting; opioid-based techniques are useful, as these drugs have minimal interference with pulmonary or systemic vascular resistances, and no significant cardiovascular depressant effects.
Neonates usually have minimal shunting across the defect until the expected decrease in pulmonary vascular resistances occurs, and are generally asymptomatic. For this patient population, as well as for older children with small defects and only mild clinical signs and symptoms, no major changes in anaesthetic technique are necessary. Children with moderate to large VSDs, high Qp:Qs and evidence of congestive heart failure and particularly patients with longstanding uncorrected defects with pulmonary hypertension represent a higher risk group; anaesthetic techniques need to be tailored to address the individual child’s pathophysiology and would ideally be conducted in a specialist centre. Patients with successful complete (early) correction of a VSD can be thought of as being compensated and treated as patients without cardiac disease. Endocarditis prophylaxis is indicated for patients with uncorrected or residual lesions, for the first 6 months after correction with prosthetic material; it is not recommended for patients with fully repaired VSDs.
LONG-TERM PROGNOSIS
Patients with isolated VSDs have generally good long-term outcomes. Children with small defects have an excellent long-term prognosis without surgery, with high rates of spontaneous closure during the first 2 years of life, and minimal clinical consequences even if this does not occur.
Spontaneous closure is uncommon for moderate or large defects and symptoms occur more frequently, but can often be controlled medically and surgery can be performed with very low morbidity and mortality, with good long-term outcomes.
A small subset of patients with associated anomalies have poorer outcomes. A minority will have irreversible pulmonary hypertension, being candidates for heart or heart-lung transplantation.
ANSWERS TO QUESTIONS
- ..
a. True: VSDs occur in 50% of all children with CHD; incidence ranges from 1.56 to 53.2 per 1,000 live births
b. False: they occur in isolation in 20% of children with CHD
c. True: other congenital malformations may coexist with VSDs, such as renal abnormalities
d. True: according to their location, they can be inlet, muscular, perimembranous or subarterial defects
e. False: perimembranous defects are the most common type, representing up to 80% of VSDs - ..
a. True: as a communication between the two ventricles, blood can flow across VSDs, which is termed shunting
b. False: right atrium dilatation is not a characteristic feature of VSDs, since shunting occurs at ventricular level
c. True: the volume load imposed on the heart by the presence of a VSD may lead to CHF, if left untreated
d. True: most often, blood will flow across the VSD from left to right, increasing blood flow to the lungs
e. True: pulmonary hypertension will eventually result from long-standing excessive pulmonary blood flow - ..
a. True: severity of volume overload to the heart is determined by the degree of shunting, influenced by VSD size
b. True: the amount of blood flow across a VSD is also determined by the resistance to flow from both ventricles
c. False: signs and symptoms will often appear only when pulmonary vascular resistance decreases significantly
d. False: there is a high rate of spontaneous closure of ventricular septal defects
e. False: the likelihood of spontaneous closure is also influenced by size - ..
a. False: asymptomatic or mildly symptomatic patients do not need any medication
b. True: diuretics reduce symptoms of congestive heart failure in patients with VSDs
c. True: a small number of children with severe CHF and pulmonary oedema need preoperative ICU admission
d. False: both short- and long-term outcomes are generally good for surgery of patients with isolated VSDs
e. True: asymptomatic patients with a predictable uncomplicated course after surgery can be fast-tracked
REFERENCES and FURTHER READING
- Hensley Jr FA, Martin DE, Gravlee GP, A Practical Approach to Cardiac Anesthesia, 3rd Edition; Davies LK, Knauf DG, CHAPTER 14: Anesthetic Management for Patients with Congenital Heart Disease http://tele.med.ru/book/cardiac_anesthesia/text/he/he014.htm – he014p084 (accessed 30th Nov 2014)
- Mancini MC, Bove EL, Devaney EJ, Ohye RG, Willis PW et al, Ventricular Septal Defect Surgery in the Pediatric Patient. E-Medicine http://emedicine.medscape.com/article/903271-overview – a1 (accessed 30th Nov 2014)
- Penny DJ, Vick III GW, Ventricular septal defect; Lancet 2011; 377: 1103–12
- Jacobs JP, Burke RP, Quintessenza JA, Mavroudis C, Congenital Heart Surgery Nomenclature and Database Project: Ventricular Septal Defect; Ann Thorac Surg 2000; 69: S25–35



