General Topics
KEY POINTS
- The posterior cranial fossa is a rigid, noncompliant compartment that houses important structures (brain stem, cerebellum, cranial nerves).
- Preoperative assessment should be extended to establishing the patient’s premorbid status, including level of consciousness, neurologic deficit and cranial nerve
- Primary goals of anaesthesia are to avoid increase in intracranial pressure, maintain haemodynamic stability and closely monitor for potential complications.
- Pain is more severe in posterior fossa surgery as compared with supratentorial
- Delayed extubation should be considered in patients with preexisting bulbar palsy, suspected airway oedema and prolonged
INTRODUCTION
The posterior cranial fossa is a rigid, confined compartment in the cranial vault.1 Due to its noncompliant nature, even a small space-occupying lesion (SOL) can result in a substantial increase in intracranial pressure (ICP) and life-threatening consequences. Surgeries involving the posterior fossa pose unique challenges for anaesthetists, primarily due to the vital structures contained within and the need for patients to remain in extreme positions intraoperatively for a prolonged period of time.
ANATOMY
As part of the intracranial cavity, the posterior cranial fossa, also known as the infratentorial fossa, is the largest and deepest fossa among all intracranial compartments (Figure 1). The boundaries of the posterior fossa are summarised in the Table. Housed within the posterior cranial fossa are several vital structures, including the brain stem (midbrain, pons, upper medulla oblongata), cerebellum, nuclei of cranial nerves III through XII, ascending and descending tracts of the spinal cord, as well as the fourth ventricle. Circulation of the posterior cranial fossa is formed by several cerebral and cerebellar arteries, comprising the posterior cerebral artery, basilar artery, superior cerebellar artery, anterior inferior cerebellar artery and posterior inferior cerebellar artery.
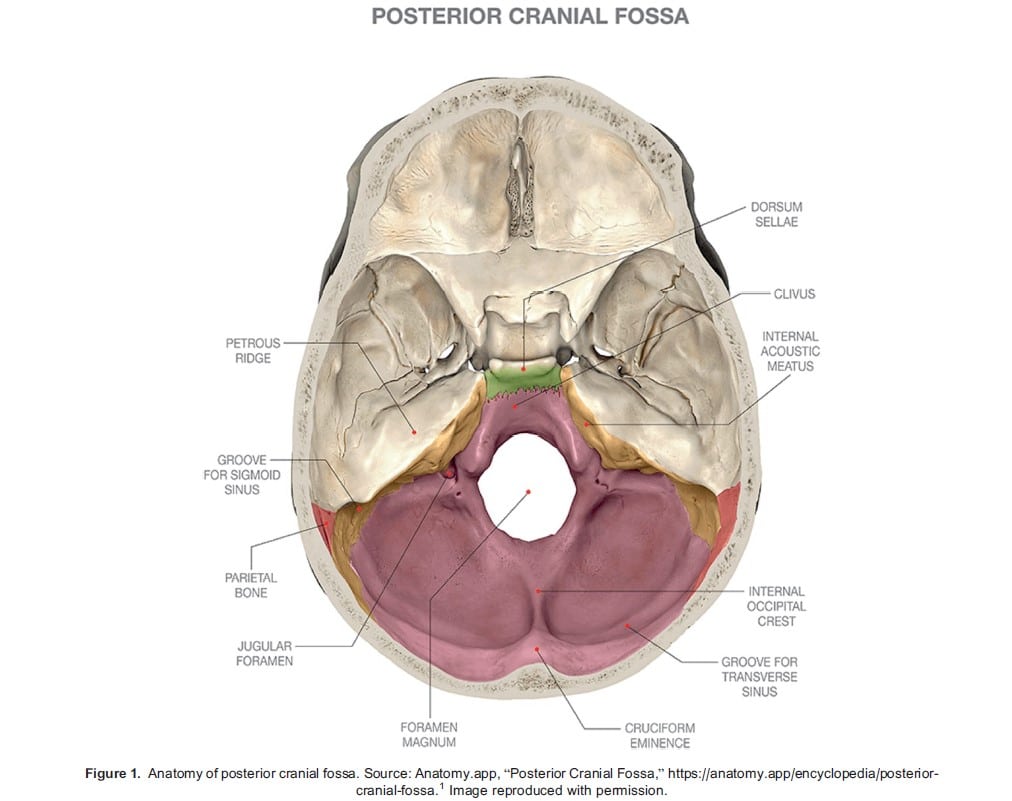
The posterior cranial fossa contains 2 dural folds: the falx cerebelli and tentorium cerebelli. The falx cerebelli lies over the floor of the fossa and partially separates the 2 cerebellar hemispheres. The tentorium cerebelli lies superior to falx cerebelli and separates it from the supratentorial fossa. Contained within the dural folds are large venous sinuses: the transverse sinuses on bilateral sides continue into the sigmoid sinuses, which further continue into the internal jugular veins before exiting the fossa. These structures are important because they play a role in the increased risks of bleeding and air entrapment during posterior fossa surgery.

The fourth ventricle is also contained within the confines of the posterior cranial fossa. The cerebrospinal fluid (CSF) flow pathway is narrow here, particularly at the cerebral aqueduct (of Sylvius). An SOL would easily obstruct the flow causing hydrocephalus.
PATHOLOGY AND INDICATION FOR SURGERIES
Indications for posterior cranial fossa surgeries can be broadly divided into 4 categories2-4:
- Excision of tumours
- Repair of vascular lesions
- Decompression of cranial nerves
- Procedures for craniocervical abnormalities
Primary tumours in the posterior cranial fossa are more common in children than in the adult population, among whom metastatic lesions are more prevalent.5,6 Tumours in the posterior fossa can be further subdivided into intra-axial and extra-axial (ie, rooted in and external to brain parenchyma, respectively). Examples of intra-axial tumours include medulloblastoma, astrocytoma, glioma and ependymoma. Extra-axial tumours commonly arise from the cerebello-pontine angle and include schwannoma and meningioma. Patients with an SOL in the posterior fossa typically present with a classic triad of vomiting, headache and ataxia.2 Seizures are uncommon, unlike in supratentorial SOL.2
Vascular lesions are usually aneurysmal, with a small percentage of arteriovenous malformations. The most common clinical presentation for a vascular lesion is following an acute haemorrhage.
Surgical decompression is carried out to relieve cranial nerve compression, be it by a mass or a vascular lesion. The most common nerve compression syndrome in the posterior cranial fossa is trigeminal neuralgia.7 Posterior fossa surgery is also carried out to correct craniocervical abnormalities, including decompression surgery for Arnold-Chiari malformation and corrective surgeries for craniovertebral junction abnormalities.2,4
PREOPERATIVE ASSESSMENT
A thorough preoperative assessment for posterior cranial fossa surgeries entails paying attention to specific factors in addition to the routine history and physical examination. Physical examination should be focused on determining the patient’s level of consciousness (LOC). A reduced LOC may indicate elevated ICP and may warrant external ventricular drainage (EVD) to address obstructive hydrocephalus either before or as part of definitive surgery.
Volaemic status of the patient should also be established. Patients with SOL in the posterior fossa are prone to dehydration for several reasons: excessive vomiting secondary to increased ICP, reduced intake with decreased level of consciousness and diabetes insipidus.2
Patients with posterior fossa pathologies may present with loss of cough or gag reflex due to mass effect on the cranial nerves.2 This can increase the risk of aspiration and may warrant a delayed extubation with early swallowing assessment. Visual as well as auditory function can sometimes be compromised with certain tumours and should be evaluated and documented prior to surgery.2 Patients with craniocervical anomalies will need a thorough cervical spine assessment with anticipation of difficult airway due to limited neck movement.
Specific to positioning intraoperatively, if the patient is anticipated to be placed in a sitting position, a patent foramen ovale (PFO) or intracardiac shunt would need to be excluded as they can lead to a paradoxical air embolism (PAE). Conventionally, transthoracic echocardiogram is used due to its accessibility and noninvasive nature8-10; however, some studies have questioned its value as a rule-out test.10,11
ANAESTHETIC TECHNIQUE
The primary goals of the anaesthetic management of posterior fossa surgery are to maintain haemodynamic stability, maintain cerebral perfusion and closely monitor for potential complications.9
INTRAOPERATIVE MONITORING
Similar to other surgeries, routine noninvasive monitoring in keeping with Association of Anaesthetists of Great Britain and Ireland guidelines should be instituted. In addition, invasive arterial monitoring is useful in allowing prompt treatment of hypotension and hypertension, especially during induction for those with critically raised ICP.2-4 The arterial line transducer should be zeroed at the level of the tragus to correlate with cerebral perfusion pressure.2,4 Often, a central venous catheter is placed in the right internal jugular vein, which can help with assessing central venous pressure as well as aspirating air in the event of a venous air embolism (VAE).2,4 If one is inserted, care should be taken with neck positioning to avoid kinking the central venous catheter, as this can cause venous congestion and lead to an increase in ICP. Urine output measurement with a urinary catheter is also essential.
NEUROPHYSIOLOGIC MONITORING
Neurophysiologic monitoring is sometimes used intraoperatively to avoid injury to neural structures. Various modalities are available, with somatosensory evoked potential, motor evoked potential, brain stem auditory evoked potential, electroencepha- logram and cranial nerve electromyography (EMG) being the most commonly used.12 Facial nerve EMG, for instance, is particularly relevant in procedures involving vestibular schwannomas. Each of these modalities monitors a different neural pathway. The use of these monitors potentially warrants a modification to the anaesthetic technique to minimise interference. Different neuromonitoring modalities and the anaesthetic implications are discussed separately in ATOTW 397.12
MONITORING FOR VENOUS AIR EMBOLISM
Posterior fossa surgery carries a risk of venous air embolism (VAE), especially when the sitting position is used. It occurs when a vein is exposed to the atmosphere and the pressure in the vein is more negative than atmospheric pressure, leading to aspiration of air into the venous circulation. While many VAEs are small, if they are large enough they can become haemodynamically significant once they reach the heart, impeding blood flow. If the patient has a PFO, a VAE can result in PAE.2-4,9 Hence, early detection is vital in posterior fossa surgery. There are several monitoring modalities available that can be used to detect VAE, each with its own benefits and disadvantages. More than one modality is recommended during posterior fossa surgery as no single monitor is completely reliable.2 The various monitoring modalities are listed below2,4 and discussed in ATOTW 34013:
- Transoesophageal echocardiogram and Doppler: Remains the gold standard, but is invasive, has risk of oesophageal injury
- Precordial Doppler ultrasound: Most sensitive noninvasive modality, can detect as little as 02 mL/kg of air in atria
- End-tidal carbon dioxide level (ETCO2): Most convenient as it is part of routine standard monitoring but is insensitive and nonspecific
- Pulmonary artery catheter: Invasive, not suitable to insert for detection of VAE alone
- Electrocardiogram, pulse oximetry, clinical vigilance
INDUCTION AND MAINTENANCE OF ANAESTHESIA
Induction of anaesthesia can be achieved by administering a judicious dose of opioid, hypnotic agent and a muscle relaxant. A reinforced endotracheal tube (ETT), alongside a bite block, is often used due to a common need for neck flexion or rotation (eg, in suboccipital craniotomy, resection of vestibular schwannoma), which confers a higher risk of ETT kinking.4,14 Laryngoscopy is done with care, and additional doses of induction agent and/or opioid can be used to avoid a hypertensive response, which can raise ICP.3
The maintenance of anaesthesia and considerations relating to cranial fixation with pins in posterior fossa surgery are similar to those in supratentorial craniotomies and are further discussed in ATOTW 466.15
Another important consideration in posterior fossa surgery is the provision of a “relaxed” brain. With the surgical window often being narrow, excessive cerebellar retraction can result in complications such as venous congestion, brain swelling and postoperative obstructive hydrocephalus. Optimal brain relaxation helps protect the brain from retraction injury and compression-induced ischaemia.16 To achieve this, it is important to avoid excessive doses of volatile agent due to their inherent vasodilatory effects. Furthermore, proper patient positioning, meticulous fluid management, avoiding hypotonic solutions and maintaining sufficient cerebral perfusion are essential in preventing brain congestion. Depending on the individual patient, osmotic agents (eg, hypertonic saline, mannitol) are sometimes used.
INTRAOPERATIVE POSITIONING AND PRECAUTIONS
Depending on the location of the lesions, posterior cranial fossa surgeries can be carried out in different positions. The most common positions are supine with head rotated, lateral or park bench, semirecumbent, prone and sitting position.
SITTING POSITION
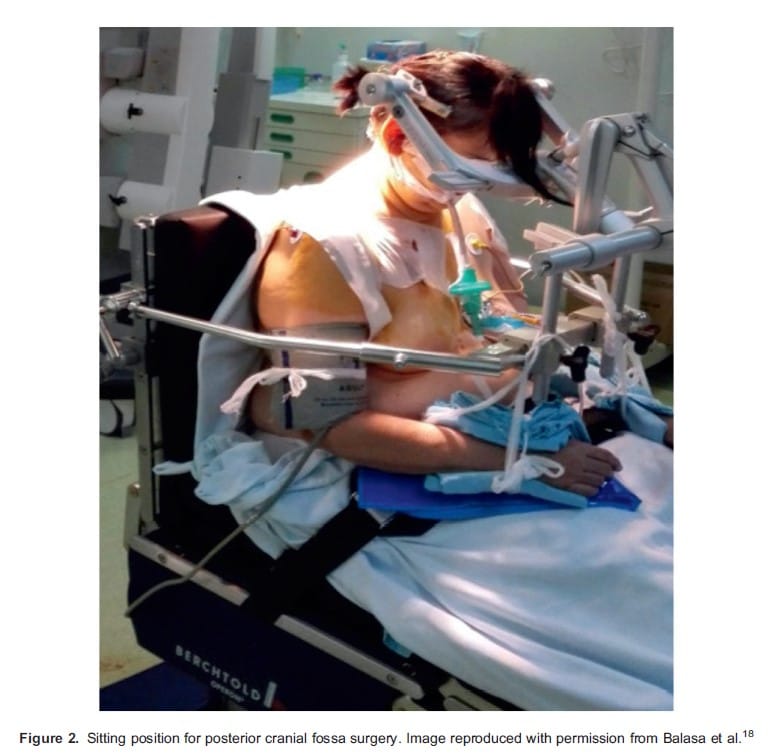
Although previously popular owing to the improved surgical orientation, the sitting position has fallen out of favour in recent years due to potential serious complications.4,8 It is now performed in only a handful of neurosurgical centres worldwide.17 Due to improved venous and CSF drainage, it provides surgeons with a better access to midline and cerebellopontine structures (Figure 2). It is, however, associated with various complications including VAE, haemodynamic instability, pneumocephalus, macroglossia and peripheral nerve injury.2-4,9 In view of the high incidence of VAE, a transoesophageal echocardiogram probe or precordial Doppler is usually placed before the start of surgery. Sitting position is also contraindicated in patients with PFO, and its exclusion is warranted in preoperative assessment.2,9
SUPINE POSITION
Supine position is usually used for lateral structures in the posterior fossa as well as acoustic neuroma.2-4 The patient’s head is rotated to the contralateral side, and a supporting pad is often placed under the ipsilateral shoulder to minimise brachial plexus injury.18 Extreme lateral rotation of the head can obstruct venous flow and lead to increased ICP.4,18
PRONE POSITION
The prone position facilitates access to the midline structures.2-4 It has gained popularity over the sitting position due to the lower incidence of VAE. However, it has several disadvantages and may not be suitable for certain patient groups. This position restricts airway access, requires repositioning for effective cardiopulmonary resuscitation, and can compromise ventilation by splinting the diaphragm.2-4 Positioning may require more time and additional staff for assistance.
LATERAL POSITION
The lateral position provides optimal surgical access for unilateral procedures by facilitating gravity-assisted drainage of blood and CSF.2-4 It has a lower incidence of VAE and cardiovascular instability but has the risks of brachial plexus and peripheral nerve injury and may increase ventilation-perfusion mismatch of the dependent lung.2,4 Care should be taken to position the dependent arm to avoid pressure on peripheral nerves.18
SEMIRECUMBENT POSITION
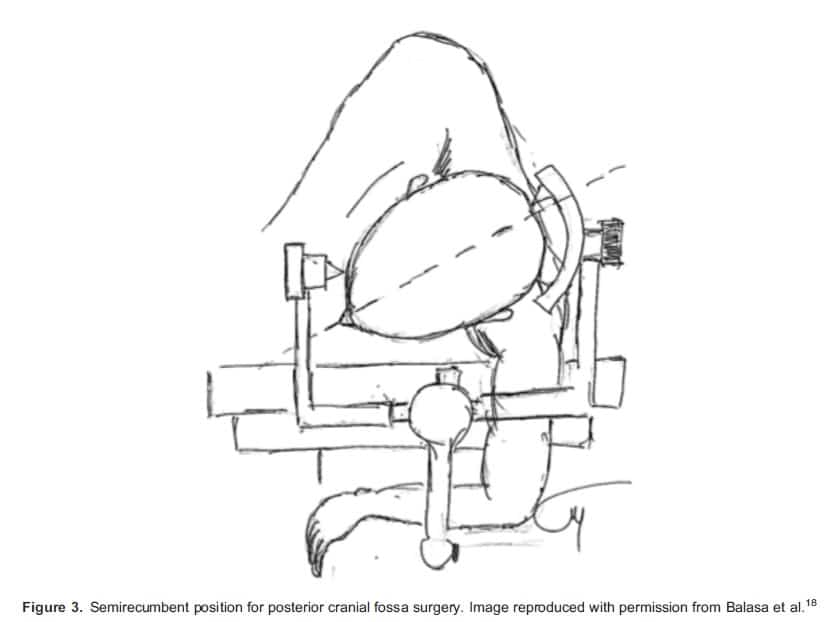
The semirecumbent position is a modification of the lateral position (Figure 3). In this position, the patient’s head is flexed at the neck, and the body is rotated into a semiprone position so the eyes face the floor. It provides better access to midline structures as compared with the lateral position, but also carries the risk of peripheral nerve injury, venous engorgement and macroglossia.2,4,18
VENOUS AIR EMBOLISM
Nitrous oxide use is not recommended given the risk of VAE and postoperative pneumocephalus, as it can cause expansion of the trapped air. The risk of VAE increases if the surgery involves opening of any dural sinuses. The sitting position is now used infrequently, and fewer cases of VAE have been reported. VAE is discussed further in ATOTW 340.13
BRAIN STEM–CARDIAC REFLEX
The brain stem–cardiac reflex can occur during posterior fossa surgery and is often related to direct brain stem manipulation or cranial nerve traction. Clinical manifestation ranges from transient hypotension or bradycardia to more severe haemodynamic instability that may result in cardiac arrest. Several brain stem–cardiac reflexes have been described, including the trigeminal- cardiac reflex and glossopharyngeal reflex.19 Although each reflex arc is distinct, all arcs culminate in cardioinhibitory vagal stimulation, leading to haemodynamic perturbations. Prompt recognition is key, and management involves informing the surgeon so that surgical stimulation is stopped immediately, and if bradycardia persists, adult cardiac life support should be followed. More detailed discussion regarding the brain stem–cardiac reflex can be found in ATOTW 452.19
POSTOPERATIVE MANAGEMENT
Emergence from anaesthesia should be smooth, with the avoidance of coughing and straining, which may raise ICP. Care in a neurosurgical intensive care unit postoperatively is essential to allow frequent neurologic monitoring. Delayed extubation after posterior fossa surgeries is common, and there are several factors to take into account when deciding whether to delay extubation.2
- Preexisting bulbar palsy: Patients with preexisting dysphagia, dysarthria or respiratory compromise may not be able to adequately protect their airway and may require controlled ventilation postoperatively
- Location and extent of surgery: Surgeries in close proximity to the brain stem and cranial nerves, or those involving excessive handling of the brain stem and lower cranial nerves (as evident from intraoperative neuromonitoring), could increase aspiration risk and airway compromise postoperatively
- Airway oedema: Can occur as a result of intraoperative positioning (eg, prone, lateral, park bench) with prolonged surgery
- Intraoperative events: Patients with clinical PAE or significant intracranial bleeding may warrant further stabilisation before extubation
POSTOPERATIVE NAUSEA AND VOMITING
Neurosurgery is an isolated surgery-related risk factor for postoperative nausea and vomiting (PONV), more so with posterior fossa surgery due to the proximity to the vomiting centre and vestibular structures, with an incidence up to 45% to 70% if left untreated.20 A multimodal antiemetic regimen remains the mainstay of prophylaxis and often comprises a combination of 2 to 3 different medications. Dexamethasone, often given to neurosurgical patients with vasogenic oedema undergoing tumoural surgery, also serves as an antiemetic. 5-hydroxytryptamine antagonists and neurokinin-1 receptor antagonists are among other popular drugs of choice.21
PAIN
Postoperative pain is common in posterior fossa surgery and more severe when compared with supratentorial surgery, as it involves surgical disruption of the overlying posterior cervical muscles, resulting in muscle spasm.22 While multimodal approach to pain management is ideal, opioid use is often necessary but has to be weighed against the risk of PONV and oversedation.
PNEUMOCEPHALUS
Pneumocephalus is the entrapment of air in the cranial vault. Surgery in the sitting position is associated with a higher occurrence of pneumocephalus, with an incidence of up to 3%.23 Measures that lead to diminution of brain volume intraoperatively promotes the entrainment of air in the intracranial space, and when the cranial vault is closed, it can cause mass effect with the reexpansion of brain leading to life-threatening tension pneumocephalus. This can present in the immediate postoperative period as delayed emergence, confusion, headache, neurologic deficit or seizure, the presence of which should prompt a computed tomography (CT) scan. Treatment includes surgical evacuation (eg, via a burr hole) in an urgent situation or conservative treatment promoting absorption of air by delivering high oxygen concentration.
OBSTRUCTIVE HYDROCEPHALUS
Obstructive hydrocephalus is a common perioperative complication of posterior fossa surgery, particularly in those involving tumours infiltrating the ventricular system.24 Postoperative hydrocephalus can be caused by local cerebral oedema, cerebral haemorrhage or mass effect by residual tumours that obstruct CSF flow.24 The presentation mimics that of elevated ICP, and suspicion should warrant a CT scan. Management of acute hydrocephalus include ventriculostomy, EVD and insertion of ventriculoperitoneal shunt.
CRANIAL NERVE DYSFUNCTION
Postoperative cranial nerve dysfunction is a recognised complication of posterior fossa surgery, with a reported incidence of 4.8%.25 Depending on the extent of pathology and the surgical approach, cranial nerves III to XII can be at risk. Cranial nerve dysfunction can be temporary or permanent and is typically caused by mechanical and thermal disruption of the nerves. Dysfunction of the lower cranial nerves, in particular, can lead to loss of gag reflex, potentially necessitating mechanical ventilation postoperatively.2,3 Intraoperative neurophysiologic monitoring helps reduce the risk.12
POSTERIOR FOSSA SYNDROME
Posterior fossa syndrome is characterised by a collection of symptoms including mutism, ataxia, hypotonia and behavioural- affective symptoms that may develop after posterior fossa surgery, usually following a short symptom-free interval.26,27 It occurs almost exclusively in the paediatric population, although a few cases are reported in adults.28 Speech and language therapy remains the mainstay of treatment.
SUMMARY
Posterior fossa surgery can pose many distinct challenges to anaesthetists. It can be performed in various positions, and a good understanding of the intraoperative position and the associated complications helps mitigate the risk.
REFERENCES
- app. Posterior cranial fossa. Accessed May 17, 2023. https://anatomy.app/encyclopedia/posterior-cranial-fossa
- Sandhu K, Gupta Chapter 14: anaesthesia for posterior fossa surgery. In: Prabhakar H, ed. Essentials of Neuroanaesthesia. Cambridge, UK: Academic Press, Elsevier; 2017.
- Jagannathan S, Krovvidi Anaesthetic considerations for posterior fossa surgery. Continu Educ Anaesth Criti Care Pain. 2014;14:202-206.
- Veenith T, Absalom Chapter 16: anaesthetic management of posterior fossa surgery. In: Matta BF, Menon DK, Smith M, eds. Core Topics in Neuroanaesthesia and Neurointensive Care. Cambridge, UK: Cambridge University Press; 2011.
- Krane EJ, Phillip BM, Yeh KK, et Anaesthesia for paediatric neurosurgery. In: Smith RM, Mototyama EK, Davis PJ, eds. Smith’s Anaesthesia for Infants and Children. 7th ed. Philadelphia, PA: Mosby; 2006:651-684.
- Grossman R, Ram Posterior fossa intra-axial tumors in adults. World Neurosurg. 2016;88:140-145.
- Baldauf J, Rosenstengel C, Schroeder Nerve compression syndromes in the posterior cranial fossa. Dtsch Arztebl Int. 2019;116(4):54-60.
- Feigl GC, Decker K, Wurms M, et Neurosurgical procedures in the semisitting position: evaluation of the risk of paradoxical venous air embolism in patients with a patent foramen ovale. World Neurosurg. 2014;81(1):159-164.
- Welch Anaesthesia for posterior fossa surgery. In: Post TW, ed. UpToDate. Waltham, MA: Wolters Kluwer; 2022.
- Teshome MK, Najib K, Nwagbara CC, et al. Patent foramen ovale: a comprehensive review. Curr Probl Cardiol. 2020;45(2):100392.
- Mojadidi MK, Winoker JS, Roberts SC, et Accuracy of conventional transthoracic echocardiography for the diagnosis of intracardiac right-to-left shunt: a meta-analysis of prospective studies. Echocardiography. 2014;31(9):1036-1048.
- Yu WH, Chung CK. ATOTW 397—introduction to intraoperative neurophysiological monitoring for anaesthetists. ATOTW. 2019. Accessed June 10, 2023. https://resources.wfsahq.org/atotw/introduction-to-intraoperative-neurophysiological-monitoring- for-anaesthetists/
- Low A, Krivvidi ATOTW 340—air embolism and anaesthesia. ATOTW. 2016. Accessed June 10, 2023. https://resources. resources.wfsahq.org/atotw/air-embolism-and-anaesthesia/
- Hubler M, Petrasch Intraoperative kinking of polyvinyl endotracheal tubes. Anesth Analg. 2006;103:1601-1602.
- Keown T, Bhangu S, Solanki S. ATOTW 466—anaesthesia for craniotomy and brain tumour resection. ATOTW. 2022. Accessed June 28, 2023. https://resources.wfsahq.org/atotw/anaesthesia-for-craniotomy-and-brain-tumour-resection/
- Li J, Gelb AW, Flexman AM, et al. Definition, evaluation, and management of brain relaxation during Br J Anaesth. 2016;116:759-769.
- Muzumdar D, Mehta S, Jadhav Sitting position for posterior fossa tumor in infants: a technical report. J Pediatr Neurosci. 2020;15(3):169-170.
- Balasa A, Hurghis C, Tamas F, Chinezu Patient positioning in neurosurgery, principles and complications. Acta Marisiensis – Seria Medica. 2020;66(1):9-14.
- Ijaz MK. ATOTW 452—brainstem reflexes during neurosurgery: an overview. ATOTW. 2021. Accessed June 10, 2023. https://resources.wfsahq.org/atotw/brainstem-reflexes-during-neurosurgery-an-overview/
- Latz B, Mordhorst C, Kerz T, et al. Postoperative nausea and vomiting in patients after craniotomy: incidence and risk J Neurosurg. 2011;114:491-496.
- Gan TJ, Diemunsch P, Habib AS, et Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2014;118:85-113.
- Thibault M, Girard F, Moumdjian R, et Craniotomy site influences postoperative pain following neurosurgical procedures: a retrospective study. Can J Anaesth. 2007;54(7):544.
- Standefer M, Bay JW, Trusso The sitting position in neurosurgery: a retrospective analysis of 488 cases. Neurosurgery. 1984;14(6):649-658.
- Zhang C, Zhang T, Ge L, et Management of posterior fossa tumors in adults based on the predictors of postoperative hydrocephalus. Front Surg. 2022;9:886438.
- Dubey A, Sung WS, Shaya M, et al. Complications of posterior cranial fossa surgery—an institutional experience of 500 patients. Surg Neurol. 2009;72:369-375.
- Lanier JC, Abrams Posterior fossa syndrome: review of the behavioral and emotional aspects in pediatric cancer patients. Cancer. 2017;123(4):551-559.
- Fabozzi F, Margoni S, Andreozzi B, et Cerebellar mutism syndrome: from pathophysiology to rehabilitation. Front Cell Dev Biol. 2022;10:1082947.
- Wibroe M, Cappelen J, Castor C, et Cerebellar mutism syndrome in children with brain tumours of the posterior fossa. BMC Cancer 2017;17:439.



